Cord Blood Collection: Safety Protocols Explained
Cord Blood Collection: Safety Protocols Explained
Cord blood collection is a safe, painless process where blood rich in stem cells is taken from the umbilical cord after childbirth. These stem cells are FDA-approved for treating over 80 conditions, including leukemia and immune disorders. Safety protocols are critical to ensure the quality and long-term viability of the sample. Here's a quick breakdown:
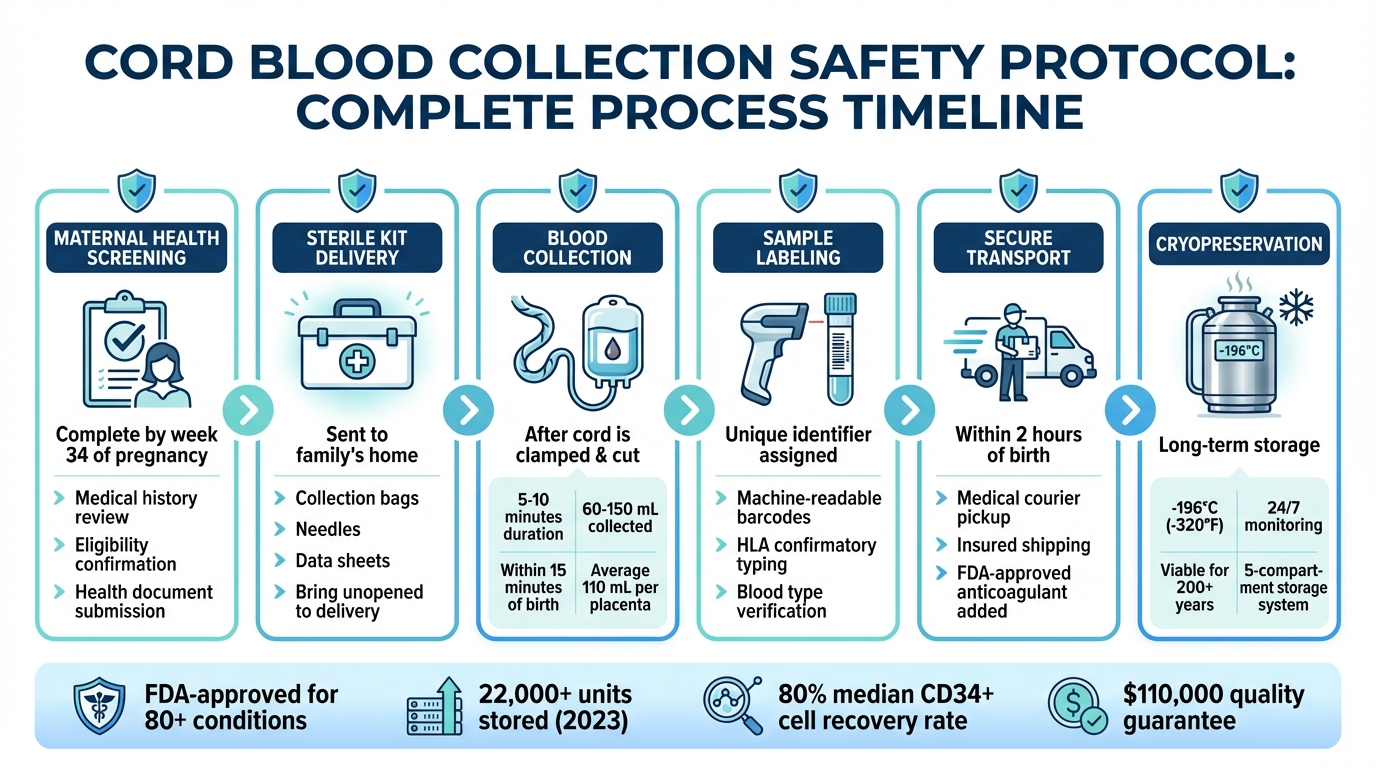
- Collection Process: Blood is collected after the cord is clamped and cut, using sterile tools to avoid contamination. It takes 5–10 minutes and collects 60–150 mL of blood.
- Maternal Screening: Parents undergo health screenings to confirm eligibility before collection.
- Sterile Kits: Families receive sterile kits for use during delivery, ensuring no contamination.
- Transport & Storage: Samples are swiftly transported to labs, treated with anticoagulants, and stored at -196°C for decades.
Americord Registry stands out by following strict safety measures, offering a $110,000 quality guarantee, and using advanced storage techniques. Proper protocols ensure cord blood remains ready for future medical use.
Cord Blood Collection Safety Protocol Timeline from Screening to Storage
Safety Measures Before Collection
Maternal Health Screening
Before collecting cord blood, expectant parents need to complete a thorough medical history and eligibility screening. This process ensures both the safety of the procedure and the viability of the sample. The screening evaluates factors like active infections, cancer, autoimmune disorders, and other disqualifying conditions. It also gathers vital information about the health of both parents, including recent medical procedures and medications.
Healthcare providers advise completing this screening well in advance of the due date. For instance, registries like Americord Registry require families to finalize enrollment and submit all necessary health documents by the 34th week of pregnancy. This timeline provides enough time to review eligibility, address any potential issues, and ensure everything is ready before the baby’s arrival.
Equipment Sterilization and Setup
Once enrollment is complete, Americord Registry sends a sterile collection kit directly to the family’s home. This kit includes all the necessary items - such as collection bags, needles, and data sheets - packaged to maintain sterility. Families must bring the unopened kit to the delivery facility, where it will be used immediately after the baby is born.
Coordination with Healthcare Providers
Clear communication between families, healthcare providers, and the cord blood registry is essential for a smooth collection process. The American Medical Association highlights the importance of this collaboration, noting:
"Physicians who participate in collecting umbilical cord blood for storage should: Ensure that collection procedures do not interfere with standard delivery practices or the safety of a newborn or the mother." – American Medical Association
Parents are encouraged to discuss their cord blood banking plans during prenatal visits, ideally by the second trimester. This allows healthcare providers to document the plan in delivery records and secure informed consent. After delivery, and following a recommended 30–60-second delay in umbilical cord clamping, the remaining cord and placental blood can be collected. The sample is then placed in sterile containers and promptly transported to Americord Registry’s storage facility to maintain stem cell viability.
This careful coordination ensures that everything is in place for a safe and efficient collection process once the baby is born.
Safety Protocols During Collection
Vaginal Delivery Protocols
During a vaginal delivery, cord blood collection is carried out with strict safety measures to ensure the well-being of both the mother and the baby. The process begins only after the umbilical cord has been clamped and cut, ensuring that no blood is drawn directly from the baby. The same OB-GYN or midwife overseeing the delivery is responsible for collecting the cord blood.
Using a sterile, large-bore needle, the healthcare provider draws blood from the detached umbilical cord into a specially designed collection bag. This bag is positioned below the placenta to allow gravity to assist the flow of blood. The procedure typically takes just a few minutes and must be completed within 15 minutes after birth to preserve the sample's viability. On average, about 110 mL of cord blood is collected per placenta, although at least 40 mL is usually required for the sample to be considered useful.
As Americord Registry explains:
"The collection is completely painless and risk-free for both mother and baby. Because the blood is taken from the umbilical cord after it has been detached from the baby, there is no physical impact on either of you."
C-Section Delivery Protocols
Cord blood collection during a cesarean section integrates seamlessly into the surgical process. As with vaginal deliveries, the blood is collected from the umbilical cord only after it has been clamped and cut. The surgical procedure itself remains unchanged, and the collection is carried out alongside the standard delivery practices without interfering with the operation.
To prevent contamination or sample mix-ups, all collected materials are stored in sterile, individualized containers. Some registries, like Americord Registry, use specialized lab processing techniques to enhance stem cell yield. If complications arise during the delivery, the protocols are adjusted on the spot to ensure the safety of the mother and baby remains the top priority.
Emergency Response Procedures
In the event of unexpected complications, the focus immediately shifts to ensuring the safety of both the mother and the baby. Emergency protocols are designed to prioritize health and stabilization over the collection of cord blood. Medical teams are trained to evaluate the situation in real time and determine whether it is safe to continue with the collection process.
If the obstetrician or medical team needs to address an urgent situation, they may delay or forgo cord blood collection altogether. Healthcare facilities follow stringent guidelines to handle errors or adverse events, ensuring that the highest standards of care are maintained even in challenging circumstances. These measures allow medical teams to respond effectively to emergencies while safeguarding patients' well-being.
Safety After Collection
Sample Labeling and Verification
Proper labeling is crucial to avoid mix-ups after collection. Each sample is assigned a unique identifier, typically using machine-readable barcodes like Codabar or ISBT 128. These barcodes carry essential details, including the facility identifier, donor lot number (which links the sample to both mother and baby), product code, and the ABO and Rh blood type.
As the AABB explains:
"Labels and bar codes - machine-readable representations of information - include critical information necessary to accurately identify, store, ship, track, and distribute blood and cellular therapy products." – AABB
Before a unit is approved for medical use, its identity is confirmed through HLA (Human Leukocyte Antigen) confirmatory typing. This verification process follows the sample throughout its journey - from collection to cryopreservation and storage. If a donor is identified as having infectious disease risks, the unit receives special labeling with a "Biohazard" legend and additional warnings to protect healthcare workers and recipients.
Transport to Storage Facility
Once verified, the sample is prepared for secure transport. A medical courier collects the kit from the hospital bedside within two hours of birth. The kit is then expedited to the laboratory using an insured shipping service to ensure the sample remains viable during transit.
Americord Registry employs a specialized 4-in-1 medical collection kit with sterile, individualized containers designed to protect samples during transportation. Parents are encouraged to contact the medical courier at 866-503-6005 immediately after birth to arrange bedside pickup. For emergency or late enrollments, AmericordRush™ provides same-day delivery of collection kits.
To prevent clotting during transport, the collected blood is treated with Citrate Phosphate Dextrose, an FDA-approved anticoagulant. Once collected, all samples are transported to Americord's specialized laboratory in New Jersey. These steps maintain a continuous safety chain, complementing the rigorous protocols followed during the pre-collection and collection phases.
Cryopreservation and Storage Standards
Upon arrival at the lab, strict chain-of-custody protocols ensure the integrity of the samples. They are processed manually using Americord's proprietary CryoMaxx™ Processing method, which is based on the Rubenstein method. This technique isolates a higher yield of hematopoietic stem cells compared to automated systems. Additionally, processing occurs in a closed system to eliminate contamination risks, reinforcing the safety measures taken earlier.
During freezing, a cryoprotectant is added to safeguard the stem cells. The samples are then stored at -196°C in nitrogen-cooled tanks that use vapor nitrogen to maintain sterility. Americord's 5-compartment storage bag allows for up to five treatments from a single collection, far exceeding the industry-standard two-compartment bags.
The facility ensures 24/7 monitoring with multi-layered security systems and has an emergency team ready to relocate samples immediately in case of natural disasters or national emergencies. Regular quality control testing is conducted to track Total Nucleated Cell (TNC) counts, with an 80% median recovery rate of CD34+ cells after thawing. As of 2023, Americord has successfully stored over 22,000 cryogenic units, and studies show that properly preserved cord blood can remain viable for more than 200 years without degradation.
Accreditation and Compliance Standards
AABB Accreditation Explained
The AABB (Association for the Advancement of Blood & Biotherapies) sets the gold standard for cord blood banking facilities. Their accreditation signifies that a facility meets stringent quality and safety requirements, verified through detailed on-site inspections by independent experts. These inspections ensure that every protocol, from donor selection to clinical use, is followed with precision.
"The main purpose for having standards and accreditation is to establish a uniform level of practice dedicated to supporting patient safety." – AABB
AABB standards encompass the entire cord blood banking process. This includes donor selection, infectious disease testing, processing, cryopreservation at ultra-low temperatures of -196°C (-320°F), storage, and eventual clinical use. The Health Resources and Services Administration (HRSA) officially recognizes AABB as an accrediting body for U.S. National Cord Blood Inventory participants. To keep pace with advancements in cellular therapies, these standards are regularly updated by expert committees.
Americord Registry goes beyond these strict benchmarks by implementing additional compliance measures to ensure exceptional care and safety.
Americord Registry's Compliance Practices

Americord Registry adheres to globally recognized standards, ensuring compliance with all regulatory requirements. The facility is FDA-registered and meets the legal requirements for operation across all 50 states, backed by necessary state-specific licenses.
Every cord blood sample processed at Americord undergoes mandatory maternal infectious disease testing, as outlined by FDA regulations. The facility exclusively uses FDA-approved materials, such as Citrate Phosphate Dextrose as an anticoagulant, while avoiding Heparin, which is not FDA-approved for cord blood collection. Additionally, all testing is conducted through CLIA-certified reference laboratories.
As of 2023, Americord has successfully cryogenically stored over 22,000 units. The company also maintains a strong reputation, with ratings of 4.8/5 stars on Google Reviews, 4.9/5 stars on Trustpilot, and a perfect 5/5 stars on the Better Business Bureau.
Conclusion
The safety of cord blood collection relies on a detailed and well-coordinated process. From thorough maternal screening and sterile procedures to accurate collection methods and secure transportation, each step is meticulously designed to preserve the sample's quality and ensure its long-term viability. This comprehensive process forms the backbone of maintaining the integrity of cord blood samples for years to come.
Americord Registry takes this a step further with its manual CryoMaxx™ processing and exclusive 5-compartment storage system, maintained at an ultra-cold -196°C (-320°F). Around-the-clock monitoring and an emergency response team add another layer of protection to safeguard stem cell viability.
Their proven track record includes 12 successful treatment releases since 2008, backed by a $110,000 Cord Blood Quality Guarantee. With cord blood stem cells already FDA-approved for treating over 80 conditions and regenerative medicine advancing rapidly - potentially benefiting 1 in 3 people in their lifetime - these rigorous protocols ensure that your family’s decision to bank cord blood translates into meaningful medical opportunities when needed most.
FAQs
What medical conditions can be treated with stored cord blood stem cells?
Stored cord blood stem cells have proven to be a resource in treating more than 80 medical conditions, including blood disorders, immune system deficiencies, and some forms of cancer. Beyond these established uses, researchers are exploring their potential in addressing conditions like cerebral palsy and type 1 diabetes, hinting at possibilities for future regenerative therapies.
As medical research progresses, the scope of applications for cord blood stem cells keeps growing, positioning them as a promising tool in advancing treatment options and medical discoveries.
How does Americord ensure the quality and longevity of cord blood samples during storage?
Americord takes careful steps to ensure cord blood samples remain viable for long-term storage. Once collected, these samples are stored in cryogenic nitrogen tanks. These tanks are constantly monitored to maintain stable, optimal conditions, ensuring the samples stay preserved. Advanced alarm systems are in place to detect and address any temperature changes or other potential issues immediately.
To add another layer of protection, the stem cells are stored in specially designed containers that separate them into multiple compartments. This setup not only safeguards the integrity of the cells but also allows flexibility for future treatments, should they be needed. With cutting-edge facilities and rigorous safety protocols, Americord prioritizes the quality and usability of every stored cord blood sample.
What should parents know about safely collecting cord blood during childbirth?
When getting ready for cord blood collection during childbirth, there are a few important steps parents should take to make the process smooth and safe. First, educate yourself about the potential benefits of cord blood banking. Cord blood is rich in stem cells, which have been used in treatments for a variety of medical conditions, offering hope for families dealing with certain diseases.
It’s also crucial to coordinate early with both your healthcare provider and the cord blood banking company, such as Americord Registry. This involves understanding the collection process, reviewing safety protocols, securing the collection kit, and completing any required paperwork ahead of time. By planning in advance, you can ensure the collection happens at the right moment and that the stem cells remain usable for the future.
Lastly, take time to discuss any specific medical needs or family considerations with your healthcare team. This collaboration will help you make informed decisions and ensure the process aligns with your family’s health goals. With careful planning, cord blood collection can be a meaningful investment in your family’s health journey.
The views, statements, and pricing expressed are deemed reliable as of the published date. Articles may not reflect current pricing, offerings, or recent innovations.