Mesenchymal Stem Cells for Crohn's Fistulas: Research Update
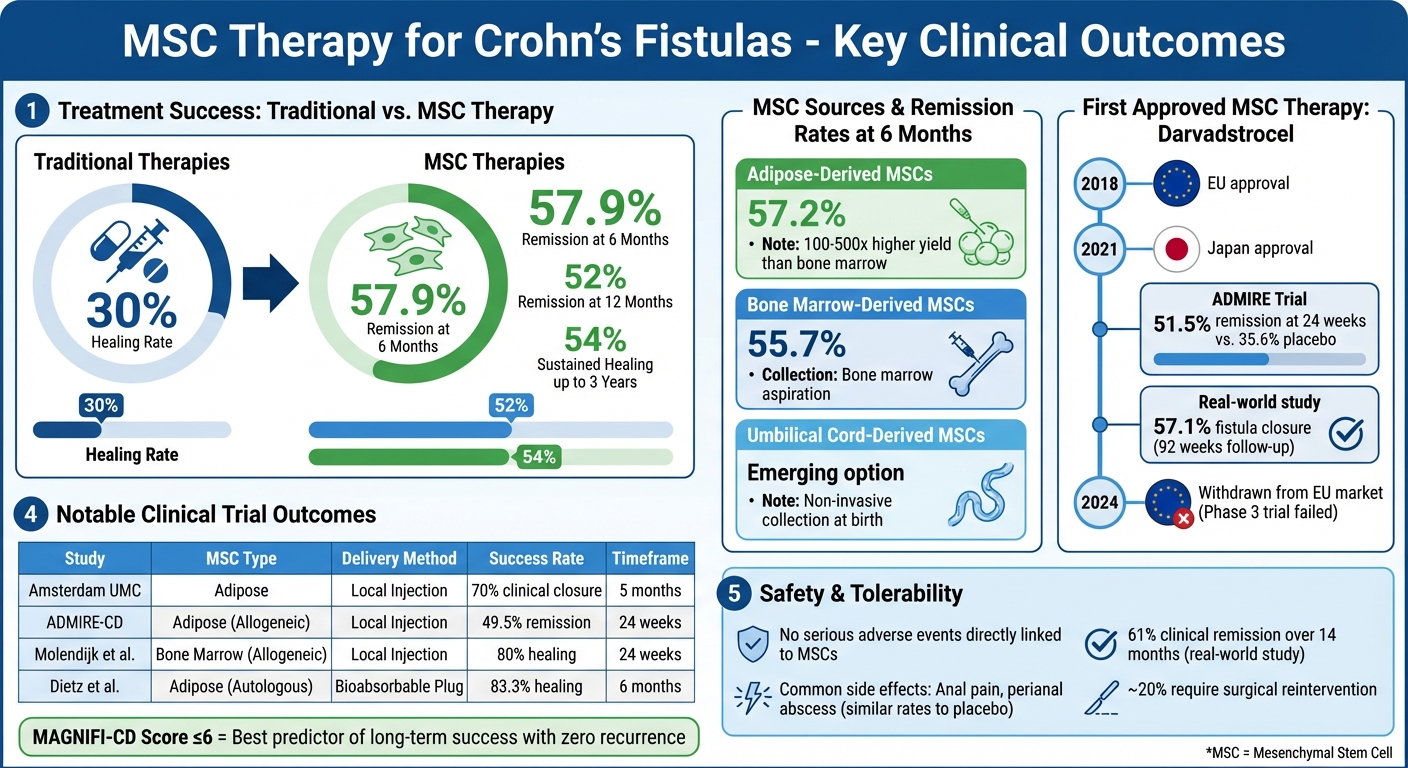
Crohn's disease often leads to fistulas, painful and complex complications that are tough to treat. Traditional therapies like antibiotics, immunosuppressants, biologics, and surgeries have limited success, with healing rates around 30% and frequent complications. Mesenchymal stem cells (MSCs) offer a new treatment option by reducing inflammation and promoting tissue repair. Clinical trials show MSC therapies achieve remission rates of 57.9% at six months and 52% at one year, surpassing traditional methods.
Key findings:
- Adipose-derived MSCs are preferred for their ease of collection and high yield.
- Darvadstrocel (Alofisel), the first approved MSC therapy, showed a 51.5% remission rate at 24 weeks but faced setbacks due to limited efficacy in later trials.
- Umbilical cord-derived MSCs are emerging as a promising alternative due to their non-invasive collection and high potency.
Safety data indicates MSC therapies are well-tolerated, with minimal adverse effects. Stem cell banking is becoming essential to ensure timely access to treatments. MSCs represent an evolving approach, offering hope for patients with refractory Crohn's fistulas.
MSC Therapy Success Rates for Crohn's Fistulas: Clinical Outcomes and Comparison
Recent Research on MSC Therapies for Crohn's Fistulas
A 2025 meta-analysis of 25 studies, involving 596 patients, revealed that mesenchymal stem cell (MSC) therapies show the highest remission rates at six months. Combined remission rates were 36.2% at 3 months, 57.9% at 6 months, and 52% at 12 months. Clinical healing was sustained in 54% of patients for up to three years (156 weeks).
MRI-based scoring systems, such as MAGNIFI-CD ≤6, are now being used to predict long-term success more effectively than relying solely on clinical closure. Below is a closer look at findings related to adipose-derived, darvadstrocel, and umbilical cord-derived MSC therapies.
Adipose-Derived MSCs in Clinical Trials
Adipose-derived MSCs are often preferred due to their ease of collection via liposuction and their significantly higher yield - 100 to 500 times greater - compared to bone marrow-derived cells. According to the 2025 meta-analysis, adipose-derived MSCs achieved a 57.2% remission rate at 6 months, compared to 55.7% for bone marrow-derived cells, with no statistically significant difference between the two.
Darvadstrocel (Cx601) Clinical Development
Darvadstrocel, sold under the name Alofisel, became the first allogeneic MSC therapy to gain regulatory approval. The pivotal ADMIRE trial demonstrated a 51.5% combined remission rate at 24 weeks, compared to 35.6% for placebo.
"Darvadstrocel is a safe, minimally invasive surgical technique without significant perioperative complications. Clinical success can be expected in about half of the treated patients."
Umbilical Cord-Derived MSCs in Development
Research is shifting toward cell-free derivatives, such as extracellular vesicles and exosomes, derived from umbilical cord MSCs. These derivatives retain the therapeutic properties of MSCs while eliminating the logistical hurdles associated with live cell therapies.
MSC Sources and Delivery Methods Compared
For delivery, local injection directly into the fistula tract has become the preferred approach. This method ensures that the MSCs are applied precisely where they are needed.
Comparison Table: MSC Sources and Delivery Methods
| MSC Source | Delivery Method | Remission Rate |
|---|---|---|
| Adipose (Allogeneic) | Local Injection | 49.5% |
| Bone Marrow (Allogeneic) | Local Injection | 80% |
Safety Data and Patient Outcomes
MSC therapies have shown to be both safe and well-tolerated. Most reported adverse events are procedural, such as anal pain and perianal abscess formation.
Stem Cell Banking and Regenerative Medicine
Stem cell banking involves preserving cells at birth for potential future use. Research indicates that MSCs collected from individuals with active inflammation might have weakened properties, making banked cells from healthy sources highly valuable.
FAQs
Why are umbilical cord-derived MSCs promising?
They regulate the immune system and support tissue repair with a low risk of rejection, offering a high regenerative potential for complex cases.
The views, statements, and pricing expressed are deemed reliable as of the published date. Articles may not reflect current pricing, offerings, or recent innovations.




