Not Just for Babies: Cord Blood Banking Protects Your Child Into Adulthood
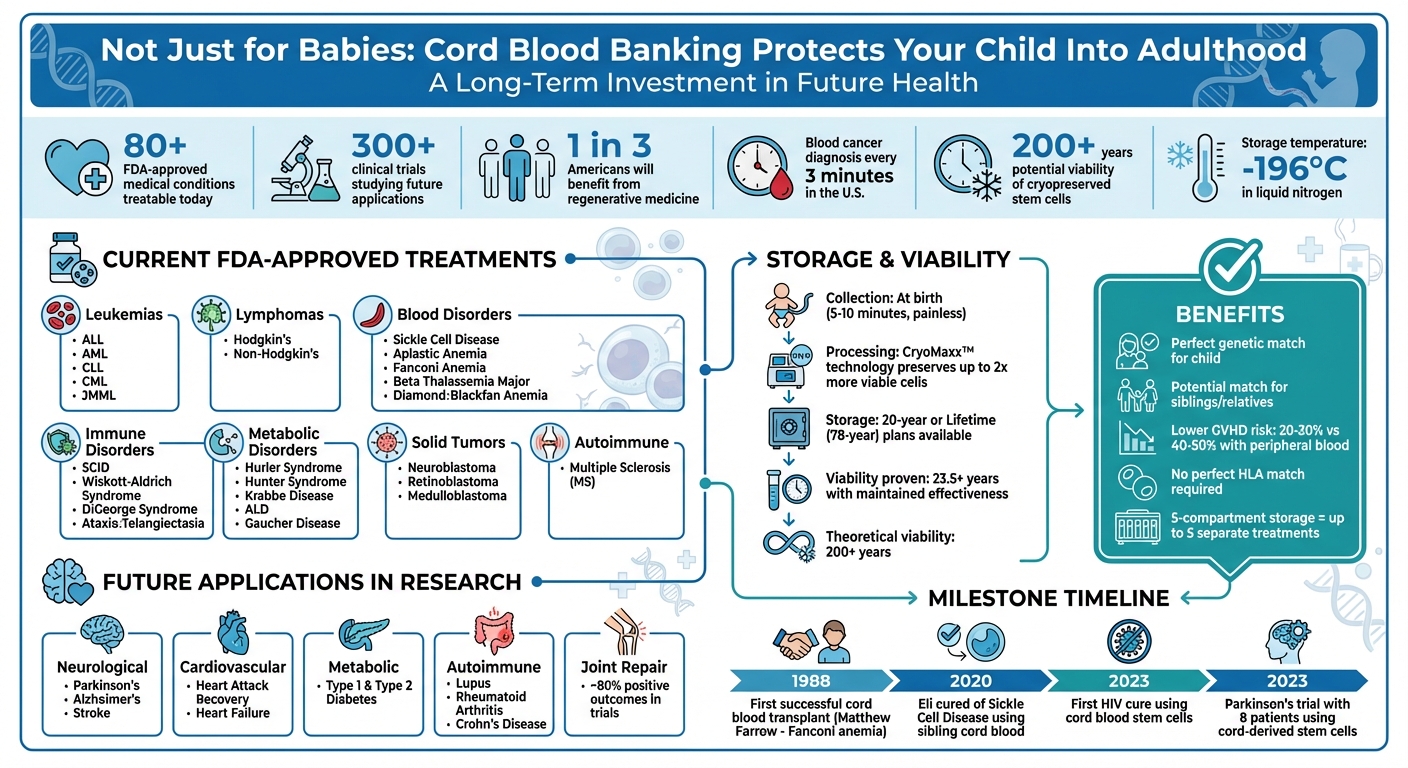
Cord blood banking isn’t just about safeguarding your baby’s health during childhood - it’s a long-term investment in their future. By storing stem cells from the umbilical cord, parents can access treatments for over 80 medical conditions today, including leukemia and sickle cell disease, with potential applications for adult diseases like Parkinson’s, heart disease, and diabetes in the future.
Key Points:
- Stem Cells: Cord blood contains hematopoietic and mesenchymal stem cells, which can regenerate blood, bone, and even organ cells.
- FDA-Approved Uses: Treats blood cancers, immune disorders, and inherited conditions.
- Future Potential: Over 300 clinical trials are studying its use for adult conditions, including neurological and cardiovascular diseases.
- Storage Longevity: Cryopreserved stem cells remain viable for decades, potentially over 200 years.
- Family Matching: Provides a perfect genetic match for the child and a potential match for siblings or relatives.
Americord Registry offers advanced processing, multi-compartment storage, and lifetime plans to ensure these cells are ready when needed, making cord blood banking a proactive step toward future healthcare solutions.
What is Cord Blood Banking?
Cord blood banking involves collecting and cryogenically preserving stem cells from the placenta, essentially serving as a long-term biological safeguard for your child's health. These stem cells aren't just useful during infancy - they hold the potential to benefit your child throughout their life.
One of the major advantages is that the stored cells are a perfect genetic match for your child, reducing the risk of rejection during treatments. Plus, there's a good chance they’ll be compatible with siblings and other family members, offering protection for more than just one person in your family.
The process itself is simple, painless, and non-invasive for both the mother and baby. It takes just 5 to 10 minutes and doesn’t disrupt the birthing process or delay cord clamping, which is often recommended for 30–60 seconds. Once collected, the stem cells are preserved at ultra-low temperatures of –196°C in liquid nitrogen. In fact, scientists suggest that these cells could remain viable for decades - or even over 200 years. Let’s dive into how the collection works and why these cells are so extraordinary.
How Cord Blood is Collected and Stored
Once the umbilical cord is clamped and cut, a needle is used to draw blood into a sterile collection bag.
From there, the collection kit is sent to a laboratory via medical courier. At the lab, technicians use manual methods to isolate hematopoietic stem cells (HSCs) from other components like plasma and platelets. For instance, Americord's CryoMaxx™ processing combines manual isolation with a cryoprotectant and controlled-rate cooling to ensure the highest possible yield of viable cells. These cells are then transferred into specialized five-compartment cryo-bags and cooled at a controlled rate.
Finally, the samples are stored in liquid nitrogen tanks at –196°C. Around-the-clock monitoring ensures the samples remain sterile and secure. Research has shown that stem cells stored for up to 23.5 years maintain their viability, with industry standards requiring at least 40 mL of blood and 100 million total nucleated cells for effective storage.
Properties of Cord Blood Stem Cells
Cord blood stem cells are younger and less exposed to harmful environmental factors, radiation, or illnesses compared to adult stem cells, making them a more reliable option for treatments.
They also have longer telomeres and a higher ability to replicate than bone marrow-derived cells. This is due to their high concentration of primitive CD34+ HSCs, typically ranging from 1 to 5 × 10^6 cells/mL. Additionally, these cells are immunologically naïve, meaning they include a larger proportion of naïve T lymphocytes (CD45RA+). This characteristic reduces the risk of Graft-versus-Host Disease (GVHD) to about 20% to 30%, compared to 40% to 50% with peripheral blood stem cell transplants.
Cord blood stem cells are multipotent, meaning they can transform into various specialized cell types. HSCs are responsible for forming blood and immune cells, while mesenchymal stem cells (MSCs) from cord tissue can develop into bone, cartilage, and nerve cells. This flexibility allows cord blood transplants to succeed even with less stringent HLA matching and stem cell transplants, unlike bone marrow transplants. These unique properties make cord blood a cornerstone of future regenerative medicine.
"Umbilical cord blood, once considered medical waste, has emerged as a clinically valuable source of hematopoietic stem cells (HSCs), mesenchymal stromal cells (MSCs), immune effector cells, and bioactive molecules." - Emmanuel Ifeanyi Obeagu, Annals of Medicine & Surgery
sbb-itb-df90ce4
Current FDA-Approved Treatments Using Cord Blood
Cord blood stem cells are already saving lives. While future advancements hold immense promise, today's FDA-approved treatments highlight the immediate impact of cord blood in medicine. These therapies primarily focus on critical blood and immune system conditions, helping patients rebuild their blood and immune systems after damage caused by chemotherapy or radiation.
In the U.S., someone is diagnosed with blood cancer every three minutes. For many, cord blood transplants are a lifeline. In some cases, they are the only treatment option available. Unlike bone marrow, cord blood doesn’t require a perfect HLA match, making it easier to find compatible donors.
Conditions Treated with Cord Blood Stem Cells
Cord blood’s life-saving abilities are evident in the wide range of conditions it can treat. Among the most common are leukemias, including Acute Lymphoblastic Leukemia (ALL), Acute Myelogenous Leukemia (AML), Chronic Lymphocytic Leukemia (CLL), Chronic Myelogenous Leukemia (CML), and Juvenile Myelomonocytic Leukemia (JMML). Treatments also extend to lymphomas, like Hodgkin's Lymphoma and Non-Hodgkin's Lymphoma.
For patients with blood disorders, cord blood offers hope. Conditions such as Sickle Cell Disease, Aplastic Anemia, Fanconi Anemia, Beta Thalassemia Major (Cooley's Anemia), and Diamond-Blackfan Anemia are treatable with these stem cells. Additionally, inherited immune disorders like Severe Combined Immunodeficiency (SCID), Wiskott-Aldrich Syndrome, DiGeorge Syndrome, and Ataxia-Telangiectasia are on the list of approved conditions.
Cord blood also addresses various inherited metabolic disorders, including Hurler Syndrome (MPS-IH), Hunter Syndrome (MPS-II), Krabbe Disease, Adrenoleukodystrophy (ALD), and Gaucher Disease. Even certain solid tumors, such as Neuroblastoma, Retinoblastoma, and Medulloblastoma, can be treated using cord blood. More recently, hematopoietic stem cell transplants have become the standard-of-care for aggressive forms of multiple sclerosis (MS), as they can effectively "reset" the immune system and halt disease progression.
Patient Treatment Examples
The potential of cord blood was first proven in 1988 when Dr. Hal Broxmeyer performed the first successful transplant on Matthew Farrow, a young boy with Fanconi anemia. Using stem cells from his newborn sister’s cord blood, Matthew’s diseased bone marrow was replaced, marking a pivotal moment in medical history.
In 2020, Eli, a 10-year-old with Sickle Cell Disease, received a life-changing transplant using cord blood from his younger brother, Gus. Thanks to the family’s decision to bank Gus’s cord blood through the Americord Cares program, Eli’s condition improved dramatically - an outcome that wouldn’t have been possible otherwise.
Another groundbreaking case involved a woman of mixed race who became the first person cured of HIV using cord blood-derived stem cells. This remarkable achievement demonstrates cord blood’s ability to tackle conditions once thought untreatable, inspiring hope for more breakthroughs.
"For many, stem cells from umbilical cord blood hold the cure." – Cord For Life Foundation
These transformative cases highlight cord blood’s established role in medicine today, paving the way for even greater advancements in regenerative therapies.
Future Applications for Cord Blood in Regenerative Medicine
While current FDA-approved treatments focus on blood disorders, researchers are now exploring how cord blood could help address adult-onset conditions. Building on its success in treating childhood illnesses, cord blood is being investigated as a potential tool in regenerative medicine, where stem cells are used to repair damaged tissues or regulate immune responses in diseases that typically arise later in life.
Research on Treatments for Adult-Onset Conditions
Clinical trials are opening new possibilities for using cord blood to treat conditions once considered untreatable. For example, neurological disorders such as Parkinson's disease, Alzheimer's disease, and stroke are being studied. In early 2023, a trial involving eight Parkinson's patients tested cord-derived stem cell therapy, aiming to promote neural regeneration while offering anti-inflammatory and neuroprotective effects through trophic factors.
In the field of cardiovascular health, researchers are examining how cord blood might repair heart tissue after events like heart attacks or heart failure. For metabolic diseases like Type 1 and Type 2 diabetes, studies are underway to see if stem cells can restore insulin production or regulate immune responses. Similarly, autoimmune diseases such as Lupus, Rheumatoid Arthritis, and Crohn's disease are being explored, with joint repair trials reporting approximately 80% positive outcomes.
"There are still significant challenges that we need to overcome, but in the long run we might even be able to create organs from stem cells taken from patients. That would enable rejection-free transplants." – Professor Janet Rossant, The Hospital for Sick Children
These advancements highlight the growing importance of ensuring the long-term usability of stored cord blood.
How Long Stored Cord Blood Remains Usable
Cord blood stem cells, when properly cryopreserved, remain viable for decades without aging or losing their effectiveness. This is particularly significant since many conditions being targeted - like Parkinson's, heart disease, and diabetes - tend to develop years or even decades after birth.
"Under well-controlled CB manufacturing and storage conditions, we do not see evidence of cell damage or product 'decay' (deterioration) in the CB units, at least for the storage times evaluated." – Andromachi Scaradavou, Department of Pediatrics Stem Cell Transplantation and Cellular Therapies, Memorial Sloan Kettering Cancer Center
With proper cryopreservation, stored cord blood retains its potency indefinitely. This makes lifetime storage plans - which can extend up to 78 years - an increasingly valuable option. The high post-thaw viability of CD34+ cells remains a key quality marker, ensuring the cells can successfully engraft and perform tissue repair when needed.
Why Choose Americord Registry for Long-Term Protection?
When it comes to long-term cord blood banking, how the cells are processed and stored makes all the difference. Americord Registry combines advanced preservation methods with flexible plans, ensuring protection that extends from infancy well into adulthood.
CryoMaxx™ Processing: Prioritizing Cell Viability
Americord’s CryoMaxx™ technology takes a manual, customized approach to processing each delivery, unlike automated systems. This method can preserve up to twice as many viable stem cells compared to the industry average. Why does this matter? Simply put, more viable cells translate to better treatment options when they’re needed - whether that’s years or decades down the road.
Their proprietary 5-compartment storage bag is a game-changer. Unlike the standard 2-compartment design, this innovation allows for up to five separate treatments from a single collection, leaving unused portions available for future needs. Stem cells are stored using controlled-rate cooling, which minimizes cell damage during freezing.
"We chose Americord because it is constantly investing in innovations in stem cell preservation and it offers cord blood products that are not available with other companies." – Jhansi P., Mother of 1
Americord also offers a $110,000 Cord Blood Quality Guarantee. If the stored cells fail to engraft, this guarantee covers finding an alternative stem cell source. Combined with their advanced processing methods, Americord ensures your family has access to both today’s treatments and emerging therapies in regenerative medicine.
Storage Plans to Fit Every Family
Americord offers five storage packages designed to meet a variety of family needs. Options range from the Essential Family Plan, which focuses on cord blood, to the Maximum Family Plan, which includes cord blood, cord tissue, placental tissue, and exosomes from both mother and newborn. Families can choose between 20-year storage or Lifetime storage (78 years), ensuring availability of stem cells well into adulthood - when conditions like heart disease, diabetes, or neurological disorders are more likely to arise.
The Essential Family Plan starts at $3,499 or $145/month for 24 months, with an upfront kit and shipping fee of $280. Until February 28, 2026, Americord is offering 15% off multi-service 20-year plans and 30% off multi-service lifetime plans. Payment options include 0% interest financing for up to 24 months, and they accept FSAs, HSAs, and HRAs when medically necessary.
| Package Name | Included Services | Best For |
|---|---|---|
| Essential | Cord Blood + CryoMaxx™ | Standard FDA-approved treatments |
| Advanced | Cord Blood + Cord Tissue | Families exploring regenerative medicine |
| Complete | Cord Blood + Cord Tissue + Placental Tissue | Maximizing mesenchymal stem cell collection |
| Ultimate | Cord Blood + Cord Tissue + Placental Tissue + Newborn Exosomes | Expanding regenerative medicine potential |
| Maximum | All Ultimate services + Maternal Exosomes | Comprehensive protection for mother and child |
Commitment to Industry Standards
Americord’s services are built on a foundation of rigorous certifications and protocols. Their in-house lab is AABB accredited, ensuring compliance with strict international standards for cellular therapies. The facility is also FDA registered and meets all federal and state regulatory requirements. Additionally, their reference laboratories are CLIA certified, guaranteeing precise and reliable clinical testing.
Americord goes the extra mile with 24/7 multi-step monitoring and uses vapor nitrogen storage to avoid cross-contamination risks. A dedicated emergency team is always on standby to relocate stem cells in case of natural disasters or other emergencies. To ensure future usability, Americord maintains minimum storage thresholds of 40 mL of blood and 100 million total nucleated cells. As of 2023, they’ve cryogenically preserved over 22,000 units and facilitated 12 successful stem cell releases since 2008.
Conclusion
Cord blood banking offers a unique opportunity to secure your newborn's health potential for their entire life. These stem cells are a perfect genetic match for your child, ready to address medical conditions that could arise even decades down the road.
With over 80 FDA-approved treatments already in use and more than 300 clinical trials currently underway, the possibilities for these cells continue to expand. They act as a form of biological insurance, becoming even more valuable as advancements in medical science unlock new therapies.
This is a one-time chance - available only at birth - to store these powerful cells. Studies indicate that 1 in 3 Americans will benefit from regenerative medicine or stem cell therapies during their lifetime. While some conditions like cerebral palsy or blood disorders may appear in childhood, others, such as chronic diseases, often surface later in life.
"When you bank your baby's stem cells and perinatal tissues, you are giving them a key to unlocking and accessing new treatment options, for their entire life." – Americord Registry
Choosing lifetime storage (78 years) ensures your child will have access to these cells when new treatments for adult-onset conditions are developed. Experts suggest that properly preserved stem cells can remain viable for over 200 years, potentially enabling therapies that don't even exist yet. Americord's CryoMaxx™ processing, multi-compartment storage bags, and $110,000 engraftment guarantee provide the reliability and security families need for this long-term commitment.
Banking your baby's cord blood is more than preparing for the unexpected - it's about ensuring access to emerging treatments and medical advancements for a lifetime. This forward-thinking decision today could open the door to tomorrow's most advanced healthcare solutions.
FAQs
What are the chances my child will ever use their banked cord blood?
The chances of using stored stem cells depend on the availability of medical treatments. Currently, there are FDA-approved therapies for over 80 conditions, including leukemia and sickle cell disease. Researchers are also investigating regenerative treatments for conditions like autism and heart disease. While the likelihood varies, stem cells can stay viable for decades, making them potentially useful even into adulthood, particularly with lifetime storage plans.
Can cord blood still be collected if we do delayed cord clamping?
Yes, it’s possible to collect cord blood even with delayed cord clamping. Healthcare providers are usually able to gather an adequate amount of cord blood for storage, ensuring it remains suitable for potential future use.
What makes stored cord blood usable decades later?
Cord blood can remain usable for decades thanks to deep freezing. This preservation method keeps the stem cells intact, maintaining their ability to function and their potential to be used in medical treatments down the line.
The views, statements, and pricing expressed are deemed reliable as of the published date. Articles may not reflect current pricing, offerings, or recent innovations.




