Exosomes in Wound Healing: What Studies Show
Exosomes are tiny vesicles that play a critical role in wound healing by delivering molecular "instructions" to cells. These cell-free therapies, derived from sources like mesenchymal stem cells (MSCs), help repair tissue, reduce inflammation, and promote blood vessel formation. Unlike stem cell transplants, exosomes carry a lower risk of immune rejection and tumor formation, making them a safer alternative for regenerative medicine.
Key points:
- How they work: Exosomes deliver proteins, lipids, and genetic material (e.g., miRNA) to activate repair pathways like PI3K/Akt and Wnt/β-catenin.
- Healing stages: They support clotting, inflammation control, tissue growth, and remodeling by stimulating keratinocytes, fibroblasts, and angiogenesis.
- Sources: Adipose-derived exosomes are easy to produce and excel at promoting blood vessel growth. Bone marrow-derived exosomes aid cell proliferation, while newer sources like gingival MSCs show promise in diabetic wound care.
- Delivery methods: Hydrogels and nanofiber scaffolds improve exosome retention at wound sites, while direct injections provide immediate but short-lived effects.
- Challenges: Scaling production, ensuring quality, and addressing regulatory hurdles remain obstacles. Exosome banking is emerging as a solution for long-term storage and personalized treatments.
Exosomes are reshaping tissue repair, offering a safer, cell-free approach to wound healing with growing clinical applications for chronic wounds and burns.
How Exosomes Work in Wound Healing
Exosomes act as messengers, delivering microRNAs, proteins, and lipids that activate repair pathways like PI3K/Akt, Wnt/β-catenin, ERK, and STAT3. These pathways guide processes like cell movement, collagen production, and inflammation control, which are essential for effective wound healing.
Cell Growth and Movement
Exosomes play a key role in boosting cell activity during healing. For instance, microRNAs like miR-21-5p activate the Wnt/β-catenin pathway, promoting keratinocyte growth and movement. Similarly, miR-150-5p triggers the PTEN/PI3K/Akt signaling pathway to enhance HaCaT cell proliferation and migration. Another example, miR-146a, increases fibroblast mobility and growth by elevating SERPINH1 and p-ERK levels. These signals ensure that skin cells multiply and migrate efficiently to the wound site, speeding up essential processes like re-epithelialization and wound contraction.
Collagen Production and Blood Vessel Formation
Exosomes are crucial for producing collagen and forming new blood vessels during the proliferative phase of healing. They stimulate fibroblasts to produce Type I and III collagen, while also delivering VEGF, PDGF, and Angiopoietin-2 to encourage capillary growth. This new vascular network delivers oxygen and nutrients to the wound, meeting the high energy demands of healing tissue. As the wound repairs, exosomes help transition collagen types, restoring about 80% of the skin's original strength.
Reducing Inflammation and Cell Death
Controlling inflammation is another critical function of exosomes. They help shift macrophages from the pro-inflammatory M1 state, which releases IL-6, TNF-α, and IL-1β, to the anti-inflammatory M2 state, which produces IL-10 and TGF-β. This shift is essential for moving the wound from an inflammatory phase to regenerative medicine. Exosomes also suppress excessive immune responses and activate survival pathways like PI3K/Akt and ERK1/2, preventing apoptosis in keratinocytes and fibroblasts. In chronic wounds that linger in an inflammatory state for weeks, exosomes help break this cycle, creating an environment conducive to healing.
sbb-itb-df90ce4
Stem Cell Sources for Exosomes
The regenerative power of exosomes isn't just about their biological mechanisms - it also hinges on the type of stem cells they come from. The source of the stem cells shapes the exosome's composition and its ability to aid in healing. Plus, the choice of source impacts how practical it is to produce these exosomes and the specific benefits they bring to wound repair. Let’s break down how exosomes from adipose tissue, gingival/synovial tissue, and bone marrow each bring something different to the table.
Adipose-Derived Mesenchymal Stem Cells
Adipose tissue is a goldmine for therapeutic exosomes. Why? It contains about 500 times more stem cells per volume than bone marrow, making it far more efficient for large-scale production. On top of that, adipose-derived stem cells (ADSCs) multiply faster than bone marrow-derived MSCs, which means more exosomes can be harvested in less time.
ADSC exosomes are particularly good at promoting angiogenesis (the growth of new blood vessels). They do this by reducing DLL4 expression and activating PKA signaling. They also carry miRNA-451a, which helps shift macrophages from the inflammatory M1 type to the healing-focused M2 type. As wounds progress into later healing stages, these exosomes also reduce scarring by boosting the MMP3 to TIMP1 ratio, which helps the extracellular matrix remodel more effectively.
Gingival and Synovium MSC Exosomes
Exosomes from gingival and synovium-derived MSCs are newer players in the field, but they’re showing promise for specialized uses. For example, synovium MSC exosomes contain lncRNA HAND2-AS1, which helps suppress fibroblast-like synoviocyte activation and provides strong anti-inflammatory effects. These types of exosomes are especially useful in treating diabetic wounds. When paired with advanced delivery systems like hydrogels, they can stay active at the wound site longer - outlasting the usual 24-hour clearance period.
Bone Marrow-Derived Exosomes
Bone marrow MSCs have long been a cornerstone of stem cell research, and their exosomes are key to wound healing. They speed up granulation and release growth factors like IGF-1, VEGF, and KGF, all of which are essential for re-epithelialization and angiogenesis. These exosomes also carry specific molecules like lncRNA KLF3-AS1, which boosts VEGFA expression to enhance blood vessel formation in diabetic wounds. Additionally, they include lncRNA MEG3, which helps reduce fibrosis and lower the risk of keloid scars.
However, bone marrow harvesting is more invasive and yields fewer stem cells compared to adipose tissue extraction. This makes it a less practical option for large-scale exosome production, despite its effectiveness.
Each of these stem cell sources offers distinct benefits, making them valuable for tailoring exosome-based therapies to specific clinical needs.
Methods for Delivering Exosome Therapies
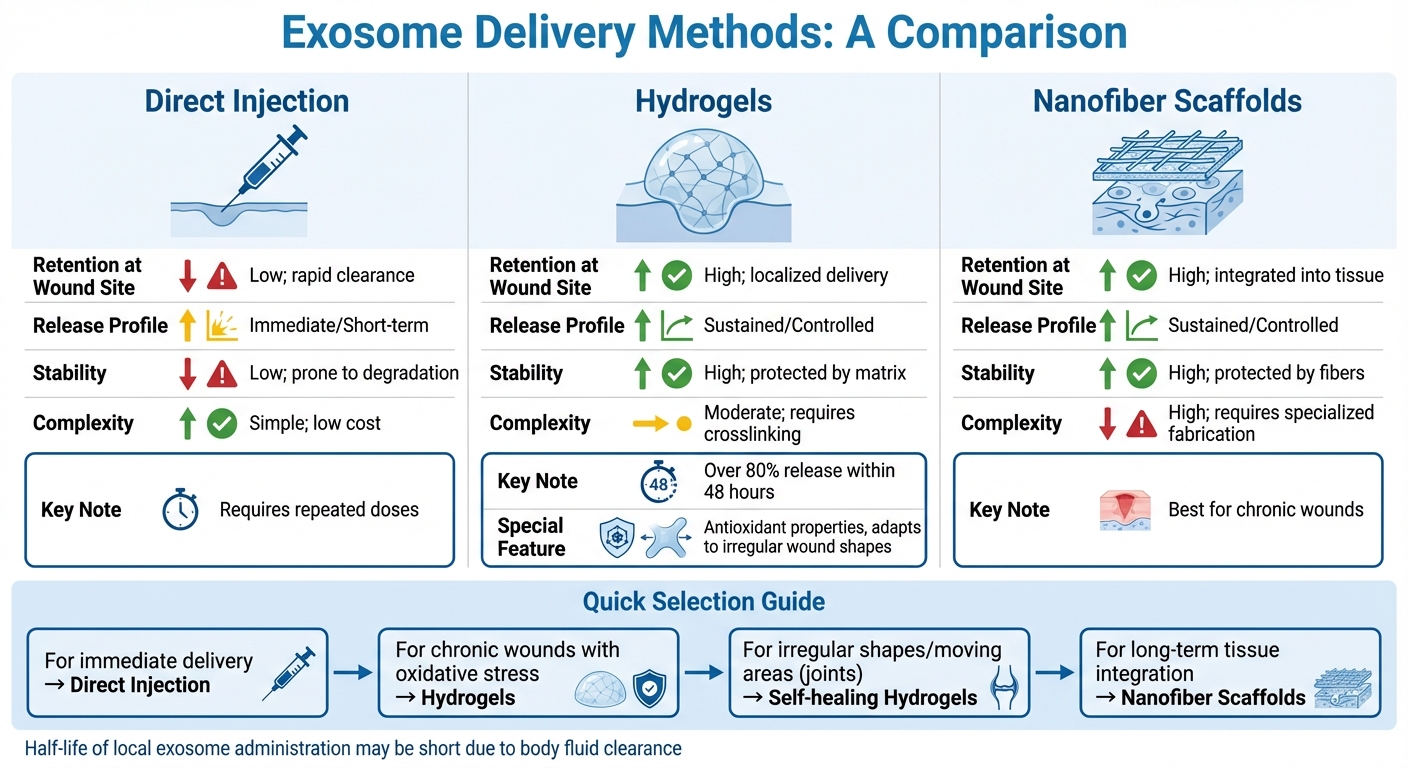
Comparison of Exosome Delivery Methods for Wound Healing
Getting exosomes to the right place and keeping them there is no easy task. The delivery method is key to ensuring these tiny vesicles stay active at the injury site. Each approach comes with its own mix of practicality, effectiveness, and how long the therapeutic effects last, often requiring advanced tissue cryostorage to maintain exosome stability before use.
Hydrogels and Nanofiber Scaffolds
Hydrogels are a popular choice for exosome delivery because they help tackle the issue of retention. Made from polymers like PVA and gelatin, hydrogels encapsulate exosomes, allowing for a steady release that maintains therapeutic levels. This sustained delivery sets them apart from methods prone to rapid clearance.
"Biomaterials encapsulated exosomes are used in wound healing for prolonged function." - Sumel Ashique, Researcher, ScienceDirect
Self-healing hydrogels take this a step further. Thanks to dynamic crosslinking (such as borate ester bonds), they adapt to irregular wound shapes and stick to tissues - even in areas that move a lot. Some formulations even come with antioxidant properties, as the borate bonds help neutralize hydrogen peroxide at the wound site. This reduces oxidative stress while delivering exosomes.
Another challenge in chronic wounds is hypoxia, or low oxygen levels. Under these conditions, cells often recycle exosomes instead of releasing their therapeutic cargo - a process called hypoxia-induced endocytic recycling. To address this, researchers have developed oxygen nanobubble systems (EBO). These systems coat nanobubbles with exosomes and embed them in hydrogels. In studies, EBO-Gel systems released over 80% of their contents within 48 hours, aligning with standard dressing change intervals. This approach combines sustained delivery with a solution to the oxygen deficiency often seen in chronic wounds. For patients looking to secure these resources early, options like a cord blood exosome upgrade ensure high-quality vesicles are available for future therapeutic applications.
While hydrogels excel at retention, direct injection offers a more straightforward delivery method.
Direct Injection
Direct injection - administered intravenously, subcutaneously, or directly into the wound - is the simplest way to deliver exosomes. It provides immediate delivery, but the downside is a short half-life due to rapid clearance by body fluids.
"The half-life of local administration of exosomes may be even shorter with low bioavailability owing to flushing by body fluid, resulting in a short retention rate of exosomes." - Hanxing Zhao et al.
Systemic injections present additional challenges. Most exosomes end up in the liver, lungs, or kidneys, with only a small portion reaching the intended site. This off-target distribution reduces the therapy's effectiveness and increases the risk of inflammation and rapid clearance from the bloodstream.
Comparing Delivery Approaches
| Delivery Method | Retention at Wound Site | Release Profile | Stability | Complexity |
|---|---|---|---|---|
| Direct Injection | Low; rapid clearance | Immediate/Short-term | Low; prone to degradation | Simple; low cost |
| Hydrogels | High; localized delivery | Sustained/Controlled | High; protected by matrix | Moderate; requires crosslinking |
| Nanofiber Scaffolds | High; integrated into tissue | Sustained/Controlled | High; protected by fibers | High; requires specialized fabrication |
The choice of delivery method depends on the wound's specific needs. For chronic wounds with high oxidative stress, hydrogels with antioxidant properties are a smart choice. In areas with irregular shapes or constant movement, such as joints, self-healing hydrogels provide the flexibility and adhesion needed for effective treatment. Direct injection, while offering immediate delivery, often requires repeated doses due to its rapid clearance. Each method has its strengths, and selecting the right one can make all the difference in therapeutic outcomes.
Clinical Use and Future Research
Current Clinical Trials
Exosome therapies are making their way into clinical applications, particularly for chronic wounds like diabetic ulcers, burns, and even aesthetic treatments. Early studies suggest that exosome therapy can significantly reduce scarring and chronic inflammation, especially in wounds that are resistant to traditional treatments. For example, in diabetic wound models, exosomes derived from mesenchymal stem cells have shown the ability to improve healing rates by 30% to 50%, offering a promising, cell-free alternative to direct stem cell transplantation.
"Exosomes have significant clinical and translational potential in the treatment of chronic wounds, burns, and aesthetic dermatology, meeting the demand for biocompatible, low-immunogenicity products." - Emmanouil Dandoulakis, MD
Unlike live cell therapies, exosome treatments avoid risks like immune rejection, tumor formation, or unintended differentiation. They deliver regenerative benefits through mechanisms such as promoting angiogenesis, reducing inflammation, and enhancing extracellular matrix remodeling - all without the complications associated with using live cells.
Despite these encouraging results, there are still obstacles to overcome.
Challenges and Opportunities
One of the biggest challenges is the lack of standardized methods for isolating, characterizing, and ensuring the quality of exosomes. Scaling up production to create therapeutic-grade exosomes in large quantities is another hurdle, as the process is both complex and expensive. On top of this, regulatory approval is difficult because exosomes exist in a gray area between biologics and cell-free therapies, requiring new frameworks to guide their use.
Clinical trials also face limitations, often involving small sample sizes and a lack of long-term data. Biological barriers add to the complexity: exosomes degrade quickly at wound sites and may cause unintended effects if they interact with the wrong tissues. To tackle these issues, researchers are exploring engineering solutions. For instance, CRISPR-engineered exosomes are being developed to target specific cells more effectively, while smart delivery systems aim to protect exosomal cargo from enzymatic degradation. The shift toward precision medicine is fueling interest in creating "modular" extracellular vesicles that can be customized for different stages of wound healing.
These challenges have inspired innovative approaches, including the concept of exosome banking.
Exosome Banking and Regenerative Medicine
Exosome banking is emerging as a practical long-term solution in regenerative medicine. Unlike stem cells, exosomes are stable and easier to store, making them ideal for preservation. Companies like Americord Registry now offer exosome banking services alongside cord blood, tissue, and placental storage, allowing families to save these powerful signaling molecules from newborn and maternal sources for future use.
This approach transforms exosomes into ready-to-use therapies, bypassing the logistical challenges of storing live cells. As research progresses toward engineered exosomes with specific therapeutic properties, banked exosomes could enable personalized regenerative treatments tailored to individual needs. By preserving exosomes at birth, families can capture these biological assets at their peak potency, potentially unlocking new possibilities for wound healing and other regenerative applications decades down the line.
Conclusion
Exosomes are reshaping the landscape of wound healing and skin regeneration. These tiny vesicles deliver the benefits of stem cells without the risks tied to immune rejection or tumor development. Studies confirm that exosomes derived from mesenchymal stem cells (MSCs) play a key role in all four stages of wound healing - hemostasis, inflammation, proliferation, and remodeling - making them a promising cell-free treatment option.
"MSC-derived exosomes have gathered increasing interest as a novel 'cell-free' therapeutic strategy in wound healing and skin regeneration." - Yulin Yang et al., Shenzhen Beike Biotechnology Co., Ltd.
Different exosome sources bring unique advantages. Bone marrow-derived exosomes encourage cell proliferation, umbilical cord-derived ones help regulate inflammation and enhance self-renewal, while adipose-derived exosomes promote angiogenesis. To address rapid degradation at wound sites, advanced delivery systems like hydrogels and nanofiber scaffolds are being developed. Additionally, bioengineered exosomes are improving stability and precision, further boosting their therapeutic potential.
However, challenges remain. Standardizing isolation methods, scaling production, and creating regulatory pathways are essential next steps. Exosome banking is emerging as a practical solution, offering families access to high-quality exosomes preserved at their peak. Companies like Americord Registry (https://americordblood.com) are already pioneering this space, making it easier to integrate these therapies into regenerative care strategies.
With ongoing research and innovations in delivery methods, the future of precision regenerative medicine is becoming more attainable. The development of engineered exosomes and personalized treatments could transform care for the 6 million Americans suffering from chronic wounds, signaling a major shift in how we approach tissue repair and regeneration.
FAQs
Are exosome wound treatments available in the U.S. today?
Exosome-based wound treatments are currently under investigation and have shown encouraging potential. However, as of early 2026, they are not broadly accessible as standard clinical therapies in the United States. Ongoing studies aim to better understand their role in skin repair and regeneration, but additional progress and regulatory clearances are necessary before they can be widely adopted in medical practice.
How are exosomes actually applied to a wound (gel vs injection)?
Exosomes are typically administered to wounds through injections rather than gels. Research indicates that daily local injections at the wound site can accelerate healing and reduce scarring. This method ensures exosomes are delivered directly to the damaged tissue, enhancing their regenerative potential. Although gel-based applications are being studied, current findings suggest injections remain the more effective approach for promoting wound recovery.
What safety risks or side effects are still being studied?
Researchers are diving deep into the potential safety risks and side effects of exosome therapies in wound healing. They’re focusing on critical factors like dosage, timing, delivery methods, and how often treatments should be administered to strike the right balance between safety and effectiveness.
Exosomes seem promising because they tend to have lower immunogenicity and carry fewer tumor-related risks compared to stem cell therapies. However, ongoing research is working to tackle challenges like possible immune reactions, unintended effects on surrounding tissues, and refining dosing strategies to reduce adverse outcomes.
The views, statements, and pricing expressed are deemed reliable as of the published date. Articles may not reflect current pricing, offerings, or recent innovations.