Future of Cord Blood in Regenerative Therapy
Cord blood, once discarded, is now a vital resource in regenerative medicine. It contains stem cells that can treat over 80 conditions, including blood disorders and immune deficiencies. Its unique properties, like immune tolerance and long-term storage potential, make it a safer and faster alternative to bone marrow transplants. Advances in cell expansion and gene editing are unlocking its potential for treating neurological and cardiovascular conditions. Challenges like limited cell yield and high costs remain, but ongoing research is paving the way for broader applications. Banking cord blood offers families a one-time opportunity to secure access to emerging medical treatments.
Current Medical Uses of Cord Blood Stem Cells
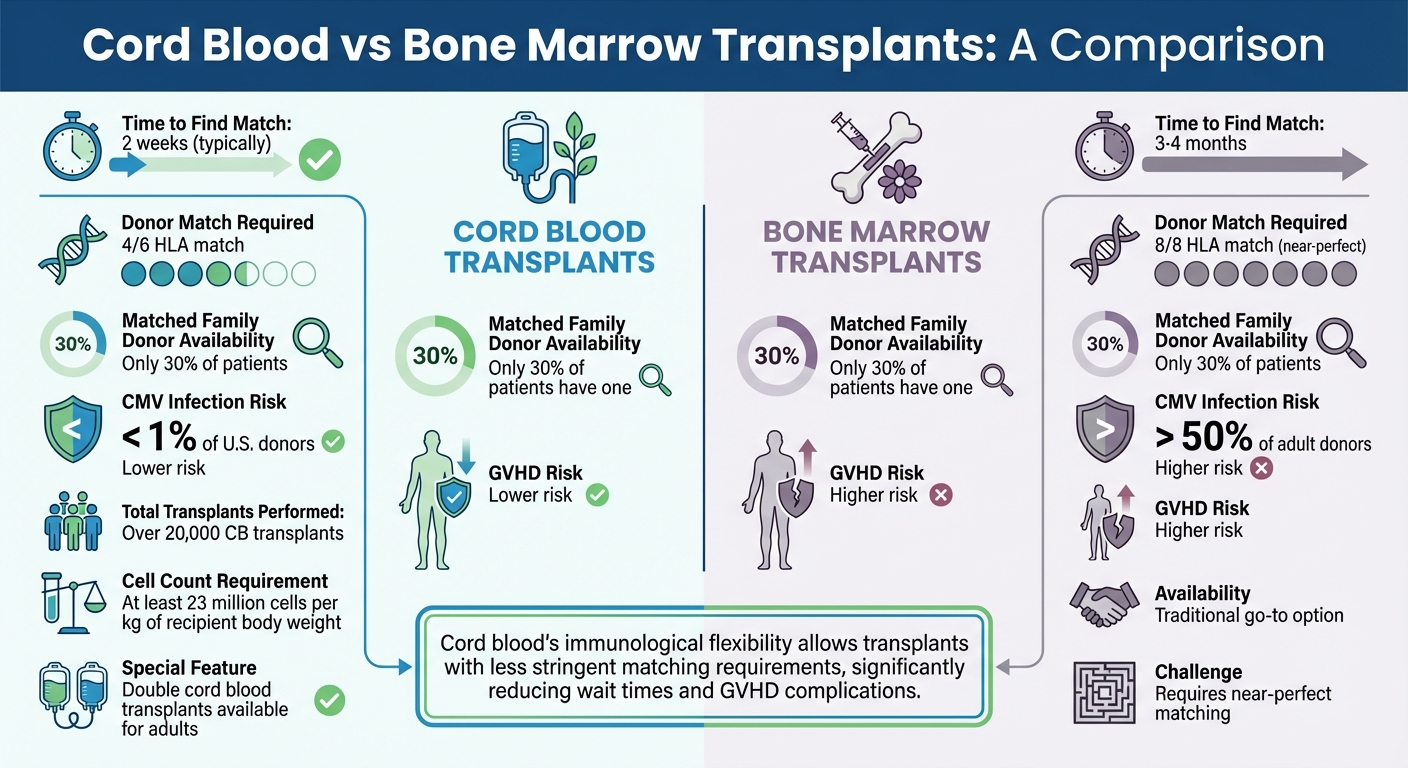
Cord Blood vs Bone Marrow Transplants: Key Differences and Advantages
Cord blood stem cells are currently approved by the FDA for treating over 80 conditions. At the heart of these therapies are hematopoietic stem cells (HSCs), which have the remarkable ability to renew themselves and transform into blood, immune, and repair cells. These stem cells play a vital role in replacing damaged or diseased bone marrow and immune systems through transplantation, offering hope for a range of serious conditions.
Treating Blood and Immune System Disorders
In many cases, patients undergo high-dose chemotherapy or radiation to clear out diseased bone marrow before receiving donor stem cells. Cord blood stem cells are then infused to rebuild the blood and immune systems. This method has proven effective for treating conditions like leukemias, lymphomas, and inherited blood disorders such as Sickle Cell Disease and Beta Thalassemia Major.
One notable example involves a 10-year-old child with Sickle Cell Disease who underwent a successful transplant using cord blood from a sibling through the Americord Cares program. The procedure led to significant recovery, demonstrating the transformative potential of these treatments. However, success often hinges on the number of cells available; doctors generally aim for a nucleated cell count of at least 23 million cells per kilogram of the recipient's body weight to ensure optimal engraftment.
Cord blood has also been used to treat immune deficiencies like SCID (Severe Combined Immunodeficiency) and Wiskott-Aldrich Syndrome, as well as metabolic disorders such as Hurler Syndrome, Krabbe Disease, and Sanfilippo Syndrome. A groundbreaking case occurred in 1988 when a patient with Fanconi Anemia received the first cord blood transplant. More than 20 years later, the patient remained healthy and free from hematological complications, showcasing the long-term benefits of this therapy.
Cord Blood in Bone Marrow Transplants
Cord blood is changing the landscape of bone marrow transplantation. It offers a viable alternative when a matched donor is unavailable or when time is of the essence. While bone marrow transplants are often the go-to option, only 30% of patients have a matched family donor. In contrast, a cord blood unit can typically be located in as little as 2 weeks, compared to the 3 to 4 months needed to find a suitable bone marrow or peripheral blood stem cell donor.
Dr. Hal E. Broxmeyer of Indiana University School of Medicine highlights the importance of cord blood:
"Cord blood has proven to be a legitimate source of clinically useful HSCs and HPCs, with a number of already noted advantages that have resulted in over 20,000 CB transplants."
One of cord blood’s key strengths lies in its immunological flexibility. Unlike bone marrow transplants, which require a near-perfect 8/8 HLA match, cord blood transplants can proceed with just a 4/6 match. This flexibility reduces the time spent searching for a donor and significantly lowers the risk of Graft-versus-Host Disease (GVHD). Additionally, cord blood carries a lower risk of viral contamination, with CMV infection present in less than 1% of U.S. cord blood donors, compared to over 50% of adult bone marrow donors.
To address the challenge of limited cell doses in single cord blood units, clinicians have developed techniques like double cord blood transplants for adult patients. This approach has improved engraftment rates, further cementing cord blood’s role as a valuable resource in regenerative medicine and transplantation.
Recent Research and Developments in Cord Blood Therapy
The field of cord blood therapy continues to evolve, with breakthroughs aimed at improving cell availability, correcting genetic disorders, and utilizing stem cells from cord tissue for regenerative purposes. These advancements are reshaping how cord blood is used in medical treatments.
Expanding Stem Cell Availability
One of the biggest challenges in cord blood transplantation is the limited number of cells in a single unit, especially for adult patients. To address this, researchers have been developing methods to multiply stem cells before transplantation, significantly improving patient outcomes.
In April 2023, the FDA approved Omisirge (Omidubicel), created by Gamida Cell Ltd. This is the first expanded cord blood product to hit the market. Clinical studies revealed that Omisirge expanded cell counts by 486-fold in just 21 days using nicotinamide. Patients who received Omisirge experienced a median recovery time of 12 days for neutrophil counts, compared to 22 days for traditional cord blood transplants.
Between 2007 and 2010, researchers at M.D. Anderson Cancer Center conducted a study involving 31 adults with blood cancers. Using mesenchymal stromal cell coculture to mimic the natural bone marrow environment, they expanded CD34⁺ cells by a median factor of 30.1. This resulted in an 88% cumulative incidence of neutrophil engraftment by day 26, compared to 53% in historical controls. As of mid-2024, there are 36 registered clinical trials exploring expanded cord blood cells, signaling a shift toward broader commercial use.
Other methods show promise as well. For instance, StemRegenin-1 (SR-1) can increase CD34⁺ cells by up to 17,100-fold over five weeks. Another approach, using Tetraethylenepentamine (TEPA) for copper chelation, achieves a median 90-fold increase. These advancements are particularly significant for adult patients, where a single unaltered cord blood unit often lacks sufficient cells for effective engraftment in individuals with higher body weights.
In parallel, gene editing technologies are adding new layers of potential to cord blood therapies.
Gene Editing and Cord Blood
Gene editing tools, like CRISPR, are enabling scientists to modify cord blood stem cells to correct genetic defects, paving the way for precision treatments.
Recent studies have demonstrated the potential for in vivo gene delivery in newborns using lentiviral vectors. Researchers have developed phagocytosis-shielded vectors that display CD47 - a "don't-eat-me" signal - on their surface. This innovation protects the vectors from immune system clearance, resulting in a fourfold increase in gene marking compared to standard vectors. For example, in a Fanconi anemia model, this gene transfer gave corrected cells a selective advantage, helping to prevent bone marrow failure.
"In vivo gene transfer provided a selective advantage to corrected HSPCs in Fanconi anaemia, leading to near-complete haematopoietic reconstitution and prevention of bone marrow failure." – Nature
Gene editing is also being used to create off-the-shelf CAR T-cell therapies from cord blood. By removing the T-cell receptor (TCR), researchers can develop allogeneic products that don't require strict HLA matching. This approach is being explored for conditions like ADA-SCID and autosomal recessive osteopetrosis, where fixing specific genetic defects can restore normal function.
Mesenchymal Stem Cell Research
Another area of focus is mesenchymal stem cell (MSC) research, which aims to address tissue repair and immune system regulation. While hematopoietic stem cells rebuild blood and immune systems, MSCs from cord tissue are showing promise for treating inflammation and tissue damage. These cells work through paracrine effects, secreting cytokines and growth factors that help the body repair itself.
In a University of Tokyo trial, seven patients with severe acute GVHD were treated with allogeneic UC-MSCs, achieving a 71.4% overall response rate and an 85.7% 100-day survival rate. Notably, no severe adverse events were reported.
One of the advantages of UC-MSCs is that they don’t require HLA matching, making them suitable for off-the-shelf therapies. Even when exposed to inflammatory signals, these cells remain negative for HLA-DR expression, allowing them to evade detection by activated T cells. As of May 2023, there are 95 clinical trials registered on the NIH website investigating UC-MSCs for conditions ranging from COVID-19-related ARDS to cerebral palsy and osteoarthritis.
Research is also exploring MSC-derived extracellular vesicles (EVs) or exosomes, which may provide similar therapeutic benefits with fewer safety concerns and simpler manufacturing processes. A pilot phase I trial in neonates with hypoxic-ischemic encephalopathy confirmed the safety of allogeneic UC-MSCs, further supporting their potential.
Future Possibilities and Obstacles in Cord Blood Medicine
Future Medical Applications
Recent advancements in cord blood research are opening doors to new possibilities, especially in neurological and cardiovascular repair. While traditionally focused on blood disorders, researchers are now exploring its regenerative potential in other areas. Neurological conditions like stroke, Alzheimer's disease, Parkinson's disease, and spinal cord injuries are particularly promising. The mechanism? Cord blood cells support tissue repair by enhancing neuroprotection, reducing inflammation, and encouraging the growth of new blood vessels.
One standout example is a 2017 Duke University trial. It revealed that children with cerebral palsy who received ≥2.5 x 10⁷ cells/kg showed significant improvements in motor function and brain connectivity compared to those given lower doses or a placebo. Similarly, a 2013 Phase 1 trial at Duke studied babies with moderate-to-severe hypoxic-ischemic encephalopathy. Those treated with their own cord blood had a 74% chance of normal development at one year, compared to just 41% in those who only received cooling therapy.
Cardiovascular treatments are also gaining momentum. Researchers are investigating ways to use cord blood stem cells for heart failure, myocardial infarction, and hypoplastic left heart syndrome. The goal is to regenerate damaged heart tissue. Beyond this, future research may apply cord blood to autoimmune diseases, kidney conditions, corneal repair, and even chronic wound healing.
Technical and Ethical Challenges
Despite its potential, cord blood medicine faces significant hurdles - both technical and ethical. A major technical issue is the limited cell yield from a single cord. One cord typically provides 50–200 mL of blood, which contains only about 10% of the hematopoietic cells needed for an adult transplant. To meet the requirements, adult patients often need two cord blood units, which can cost up to $80,000, not including hospital expenses.
"CB is limited by the small volume and total amount of hematopoietic cells. Therefore, ex vivo expansion may be required prior to adoptive cellular transfer." – Jiasheng Wang and Leland Metheny, Department of Hematology/Oncology, University Hospitals Seidman Cancer Center
Another challenge is cryopreservation. While cord blood can be stored for years, the long-term effects of storage beyond a decade on cell viability and functionality remain uncertain. Additionally, the cryoprotectant DMSO, used to preserve cells, can be toxic if not properly removed after thawing.
Ethical concerns further complicate the field. Private cord blood banking raises questions about fairness and access. High costs often make these services accessible only to wealthier families, creating a perception that regenerative therapies are exclusive to the affluent. The global cord blood banking market is projected to surpass $20 billion by 2026, yet unresolved issues around ownership rights persist - such as who controls the cord blood when the child becomes an adult or when parents pursue unproven treatments.
"A private stem cell preservation company may be perceived as unfairly catering to the wealthy and not to other members of the public who cannot afford such services, thereby reinforcing a perception that regenerative therapies will only be available to the affluent." – npj Regenerative Medicine
Public cord blood banks face their own struggles. Nearly 90% of these institutions report financial difficulties, and only about 10% of the stored units are ever used for transplants. These low utilization rates contribute to high operational costs, making it challenging to sustain public banks. Moving forward, the field must address these technical and ethical challenges while ensuring scientific progress is balanced with accessibility and financial sustainability.
sbb-itb-df90ce4
Americord Registry and Cord Blood Banking Services

Americord Registry transforms cutting-edge research in cord blood therapy into practical solutions for families, offering a range of services designed to preserve critical newborn biological resources.
Americord's Service Offerings
Americord Registry specializes in preserving umbilical cord blood, cord tissue, placental tissue, and exosomes, each with distinct regenerative potential:
- Cord blood is rich in hematopoietic stem cells (HSCs), which are already used in over 80 FDA-approved treatments for blood and immune disorders.
- Cord tissue and placental tissue contain mesenchymal stem cells (MSCs), currently being studied in more than 300 clinical trials for conditions like spinal cord injuries and Parkinson's disease.
- Exosomes are at the forefront of regenerative medicine, with ongoing research exploring their role in cell communication and tissue repair.
Americord employs CryoMaxx™ Processing, a manual technique based on the Rubenstein method, which yields higher stem cell counts compared to automated systems. This method also preserves essential growth factors and cytokines that automated processes might compromise. For storage, the company uses a 5-compartment storage bag, maintaining samples at -196°C. This system allows for precise use of stem cells for specific treatments while keeping extra cells available for future needs.
Americord operates an FDA-registered, AABB-accredited lab in New Jersey, featuring 24/7 monitoring and over 22,000 cryogenically stored units as of 2023. Pricing begins at $3,799 for cord blood banking, with monthly payment options starting at $145. Storage terms range from 20 years to a lifetime (78 years). The company also offers a $110,000 engraftment guarantee, the highest in the industry, to secure an alternative source if a transplant fails to engraft.
These advanced techniques reflect Americord's commitment to supporting regenerative medicine and ensuring families have access to emerging therapies.
Supporting Regenerative Medicine
Americord's services play a critical role in advancing regenerative medicine by providing biological resources for both current treatments and future research. Since 2008, the company has facilitated 12 successful medical releases, with over 10 families currently participating in clinical trials at institutions like Duke University and Riley Children's Hospital for conditions such as autism and cerebral palsy.
Research into co-transplantation - combining HSCs from cord blood with MSCs from cord or placental tissue - shows promise in improving engraftment rates and lowering the risk of Graft-versus-Host Disease. Americord's multi-compartment storage system supports this approach by keeping different cell types separate. Additionally, their placental tissue preservation method isolates MSCs genetically matched to the mother. These cells are being studied for their ability to expand cord blood HSCs ex vivo, potentially making single cord blood units suitable for adult treatments - a major step toward overcoming the challenge of limited cell yields.
Parents have just one chance to collect these stem cells - at birth. Americord aligns with ACOG's recommendation for delayed cord clamping (30–60 seconds), ensuring that cord blood collection remains possible without compromising the baby's health.
Conclusion
Cord blood has transitioned from being considered medical waste to becoming a key player in regenerative medicine. These stem cells have shown their effectiveness in treating blood disorders, immune system conditions, and even neurological injuries. In fact, about 80% of cord blood units released by some private banks now serve regenerative medicine purposes rather than traditional blood transplants.
As therapies advance, new technologies are reshaping the potential of regenerative treatments. Innovations like gene editing, CAR-T therapy, and ex vivo expansion are putting banked cord blood at the forefront of personalized medicine. Jennifer D. Newcomb from the Center of Excellence for Aging and Brain Repair highlights its importance:
"Cord blood banking offers expectant parents a biological insurance policy that can be used in the event of a child or family member's life-threatening illness and puts patients in a position of control over their own treatment options."
For families, storing cord blood is a once-in-a-lifetime opportunity to ensure future access to regenerative treatments. Studies confirm that these cells remain functional even after 24 years in storage. Banked cord blood provides an immediate, perfect genetic match, removing the uncertainty of finding compatible donors.
FAQs
What makes cord blood a better option than bone marrow for transplants?
Cord blood has some clear benefits compared to bone marrow when it comes to transplants. For starters, it’s immediately available after birth since it’s collected and stored ahead of time. This eliminates the often time-consuming process of finding a compatible donor. Plus, the collection process is quick and completely painless, posing zero risk to both the mother and the baby.
Another major benefit is that cord blood carries a lower chance of graft-versus-host disease - a common and serious complication in transplants - thanks to its unique immune characteristics. These advantages make cord blood a valuable option for treating various blood and immune disorders, while also opening doors for progress in regenerative medicine.
How is gene editing advancing the use of cord blood in regenerative medicine?
Gene editing, particularly through CRISPR technology, is transforming how cord blood is used in regenerative medicine. This method allows scientists to make precise changes to hematopoietic stem cells (HSCs), which opens the door to correcting genetic mutations responsible for inherited blood disorders. The result? Treatments that are not only more effective but also have the potential to cure these conditions.
By modifying cord blood cells outside the body before transplantation, researchers can boost safety, refine the accuracy of treatments, and even create therapies tailored to individual patients. This approach is showing promise for addressing conditions like sickle cell anemia and thalassemia. As advancements in these technologies continue, they’re expanding the horizons of cord blood therapies, bringing new hope to those affected by genetic and blood-related diseases.
The views, statements, and pricing expressed are deemed reliable as of the published date. Articles may not reflect current pricing, offerings, or recent innovations.




