Mesenchymal Stem Cells for Joint Repair: Early vs Late Stages
Stem cell therapy, specifically mesenchymal stem cells (MSCs), which are a type of newborn stem cell, is being studied as a treatment for osteoarthritis (OA). Here's what you need to know:
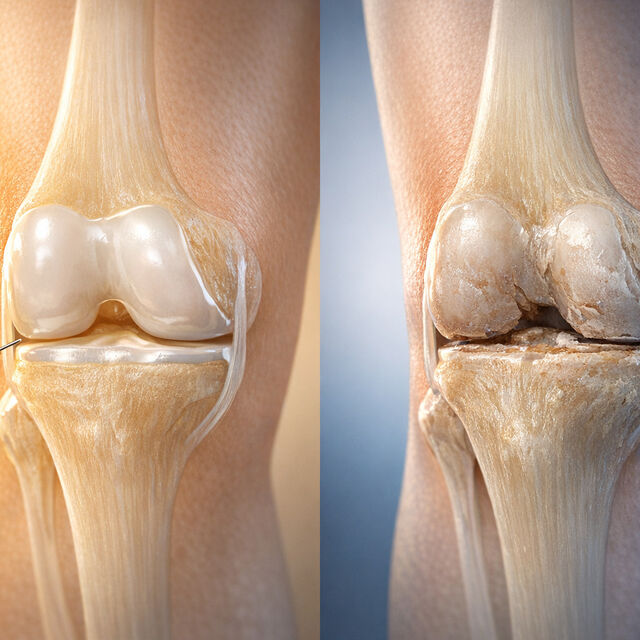
- Early-stage OA (mild damage): MSCs may reduce inflammation, slow disease progression, and even help repair cartilage. Results show pain relief lasting up to 5 years and structural improvements in the joint.
- Late-stage OA (severe damage): MSCs focus on reducing pain and delaying surgery. While some cartilage thickening can occur, reversing "bone-on-bone" damage isn’t realistic.
Key Takeaways:
- MSCs work better in early stages of OA when cartilage damage is minimal.
- For late-stage OA, MSCs help manage symptoms but offer limited repair potential.
- Safety is strong, with mild side effects like swelling or pain at the injection site.
- Costs range from $3,000 to $10,000, and insurance rarely covers it.
Timing is everything. Early intervention with MSCs might preserve joint health, while advanced cases focus more on pain relief and delaying surgery.
1. MSCs for Early-Stage Osteoarthritis
Treatment Protocols
For managing early-stage osteoarthritis, delivering MSCs (mesenchymal stem cells) with precision is crucial. The standard method involves a single injection directly into the knee joint, guided by ultrasound. These treatments often rely on advanced tissue cryostorage services to ensure cell viability. Typically, doses range from 10 to 100 million cells in 3–5 mL. Studies suggest that doses of 40 million to 50 million cells are particularly effective, though even lower doses, such as 25 million cells, have been shown to significantly reduce pain.
MSCs can be derived from various sources, including the patient's own bone marrow or adipose tissue (autologous) or from umbilical cord tissue (allogeneic). The yield of MSCs varies widely: adipose tissue provides about 10% MSCs, while bone marrow contains only 0.001% to 0.01% MSCs among its nucleated cells. Some protocols combine MSCs with additional agents like Hyaluronic Acid or Platelet-Rich Plasma to enhance outcomes. After the injection, patients are generally advised to avoid intense physical activity for 48 hours, with some protocols recommending limited weight-bearing for anywhere from 3 days to 4 weeks.
These carefully designed protocols have been shown to improve joint structure and function measurably.
Clinical Outcomes
A systematic review of 12 high-quality studies involving 539 patients with Kellgren-Lawrence grade I–III osteoarthritis revealed promising results. In grade II cases, a single MSC injection improved MOCART scores from 52.9 to 62.1 over 12 months. Additionally, bone marrow lesions decreased in size, shrinking from 123.5 mm² to 90.3 mm². Another study of 467 patients demonstrated long-term success, with 99.8% avoiding treatment failure at 5 years, 94.5% at 7 years, and 74.5% at 9 years.
"Single intraarticular injection of MSCs is a safe, reliable, and effective treatment option for Kellgren-Lawrence grade I - III knee osteoarthritis." - Journal of Experimental Orthopaedics
When compared to other treatments, MSC therapy stands out. For instance, MSC-based injections provide pain relief lasting 61.5 months, compared to 30.4 months with Hyaluronic Acid. Similarly, MSCs and Platelet-Rich Plasma offer comparable improvements over 24 months for early-stage osteoarthritis, but MSCs often show added structural benefits.
Efficacy and Mechanisms
The effectiveness of MSC therapy is rooted in its biological mechanisms. Rather than permanently integrating into the joint, MSCs act through immunomodulation and paracrine signaling. They help shift macrophages from an inflammatory (M1) state to an anti-inflammatory (M2) state and release growth factors like TGF-β, which protect cartilage cells from dying. Additionally, MSCs inhibit enzymes like MMP-13 and ADAMTS5, which degrade cartilage, while promoting markers that support cartilage health, such as Sox9, aggrecan, and type II collagen.
Structural improvements have been widely documented. Out of 21 clinical studies, 18 reported cartilage protection or regeneration. In one 5-year MRI follow-up, a 5.9% reduction in full-thickness cartilage defect size was observed, along with significant decreases in serum levels of Hyaluronic Acid and COMP, both markers of cartilage breakdown.
Safety
The safety profile of MSC therapy is another key advantage. Most side effects, such as temporary swelling and pain at the injection site, resolve within 2 to 3 days. No serious complications, including tumor formation, have been reported in clinical trials. Allogeneic MSCs, particularly those derived from umbilical cord tissue, are especially appealing because they are readily available and have low immunogenicity. This eliminates the need for invasive harvesting procedures required for autologous sources, making the process less burdensome for patients.
2. MSCs for Late-Stage Osteoarthritis
Treatment Protocols
When osteoarthritis progresses to advanced stages (Kellgren-Lawrence Grade III-IV), treatment approaches require significant adjustments. For these cases, higher doses of mesenchymal stem cells (MSCs) are often necessary - protocols may call for up to 100 million cells per injection to achieve measurable effects. Unlike earlier stages, where a single injection might suffice, late-stage patients typically need annual booster injections as the benefits tend to plateau after 2–3 years.
For advanced osteoarthritis, adipose-derived MSCs are commonly used. These cells are easier to harvest in large quantities, as adipose tissue can yield approximately 2 million cells per gram, meeting the higher cell count demands. Structured exercise programs following the injections are also crucial - patients who engage in regular physical activity tend to experience better outcomes over time. However, these protocols are generally viewed as a way to delay total knee replacement surgery, rather than offering a cure, since reversing "bone-on-bone" damage remains elusive.
Clinical Outcomes
The outcomes for late-stage osteoarthritis are less promising compared to earlier stages. For example, in a phase II trial at Shafa Yahyaian Hospital (June 2019–September 2021), 40 patients with knee OA were treated with either 100 million allogeneic adipose-derived MSCs or a placebo. The MSC group experienced a 70% reduction in WOMAC scores (a measure of pain and disability) within six months. Additionally, inflammatory markers like IL-6 decreased after three months, and MRI scans showed increased cartilage thickness in specific knee areas after 12 months.
However, broader studies paint a more modest picture. A 2024 meta-analysis revealed that MSC therapy improved pain by just 0.73 cm on a 10 cm scale after 12 months - well below the 1.5 cm threshold considered clinically significant. Similarly, the MILES study found that MSC injections offered no significant advantage over corticosteroid injections after one year. Furthermore, about 13% of patients still required total knee replacement within two years of treatment.
"Biologic therapies, including stem cell and PRP injections, cannot currently be recommended for the treatment of advanced hip or knee arthritis." - American Association of Hip and Knee Surgeons (AAHKS)
Efficacy and Mechanisms
In advanced osteoarthritis, MSCs primarily work through immunomodulation and paracrine signaling, but their effectiveness is more limited compared to earlier stages. The damaged joint environment reduces MSC survival rates, and their ability to repair structural damage is minimal. Unlike early-stage treatments, where gradual improvements are common, advanced cases present significant challenges for MSC therapy, including harsher conditions that limit regenerative potential.
Adipose-derived MSCs may have an edge in these cases because they are better suited to survive in the low-oxygen conditions of severely damaged joints. The therapy works by suppressing cartilage-degrading enzymes such as MMP-13, TNF-alpha, and IL-1β, while promoting factors like Type II collagen and aggrecan, which support cartilage health. However, there is still no strong imaging evidence that MSCs can reverse the structural damage associated with "bone-on-bone" arthritis.
Safety
Despite variability in clinical outcomes, MSC therapy has a strong safety record for osteoarthritis treatment. The most common side effects include temporary joint swelling and localized pain, which typically subside within 2–7 days. In a large phase 3 trial, 24.1% of patients receiving umbilical cord-derived MSCs experienced joint swelling, compared to 7.4% in the corticosteroid group. Adipose-derived MSCs were associated with higher rates of post-procedure complications, such as contusions (38.6%) and hematomas (12.4%) at the harvest site.
The overall risk of adverse events is higher with MSC therapy compared to placebo, with a risk ratio of 2.67 for any side effect and 1.58 for knee pain and swelling specifically. However, no serious complications - such as infections, allergic reactions, or tumor formation - have been reported in clinical trials. Costs remain a barrier, with treatments typically ranging from $3,000 to $10,000 out of pocket. Insurance coverage is rare, as these procedures are still considered experimental and lack FDA approval for joint regeneration.
Pros and Cons
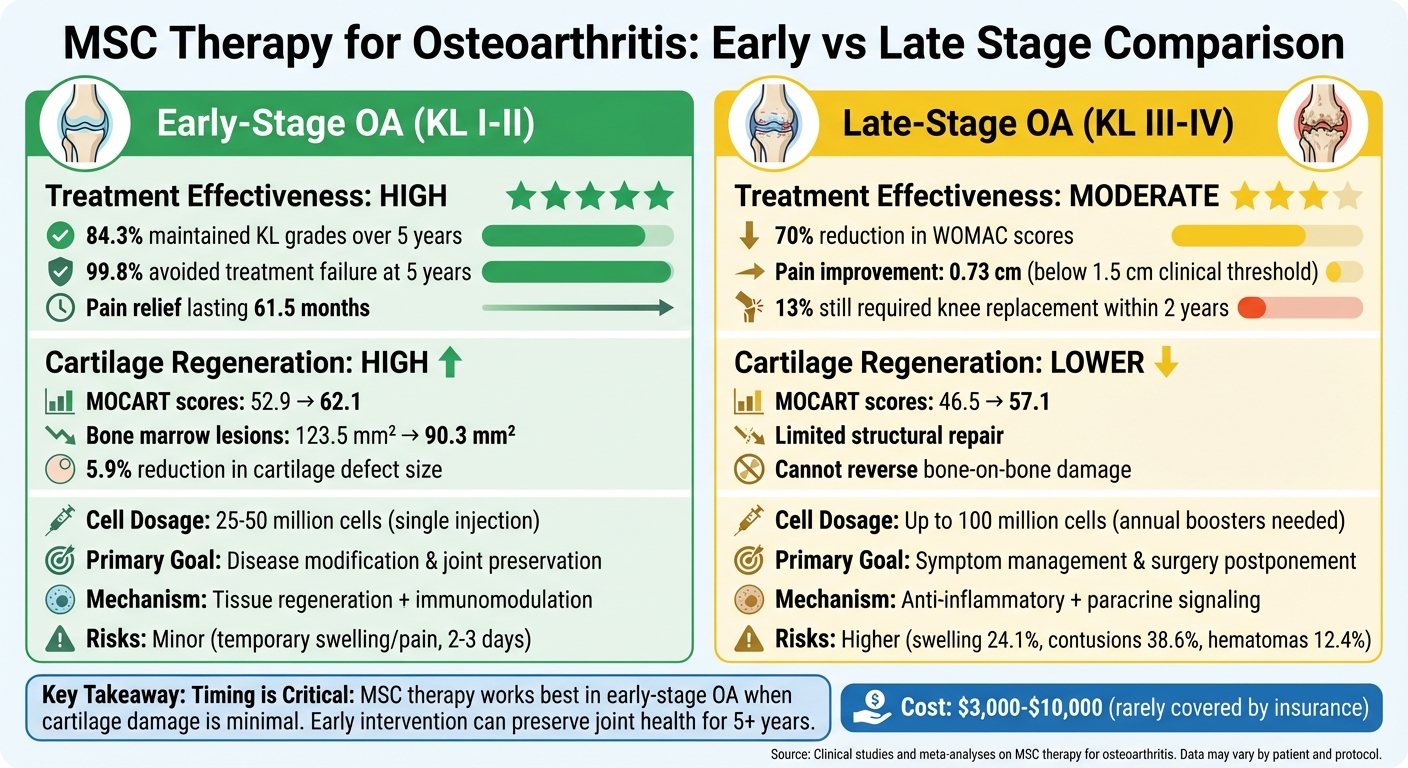
MSC Therapy Effectiveness: Early vs Late Stage Osteoarthritis Comparison
Mesenchymal stem cell (MSC) therapy offers both advantages and limitations, depending on the stage of osteoarthritis (OA). The timing of the treatment plays a crucial role in determining its effectiveness and the challenges involved.
Early-stage osteoarthritis (Kellgren-Lawrence grades I-II) tends to respond well to MSC therapy. Research highlights that 84.3% of patients maintained their Kellgren-Lawrence grades over five years, indicating that the therapy can help stabilize the condition and preserve joint function. The primary focus at this stage is on modifying the disease and preserving the joint, with minimal risks such as minor swelling or localized pain at the injection site.
In contrast, late-stage osteoarthritis (grades III-IV) presents a different set of challenges. While MSC therapy can still provide pain relief and delay the need for total knee replacement, its regenerative capacity is significantly reduced. At this stage, the emphasis shifts from regenerating tissue to managing symptoms. The primary goal is to postpone surgery rather than reverse existing damage. Severe joint surface damage, such as "bipolar kissing lesions" where cartilage is lost on both joint surfaces, further complicates treatment and increases the likelihood of failure.
| Feature | Early-Stage OA (KL I-II) | Late-Stage OA (KL III-IV) |
|---|---|---|
| Treatment Effectiveness | High; noticeable improvement in function and pain scores | Moderate; focused on pain relief and delaying surgery |
| Cartilage Regeneration Potential | High; MOCART scores improved from 52.9 to 62.1 | Lower; MOCART scores improved from 46.5 to 57.1 |
| Primary Mechanism | Tissue regeneration and immunomodulation | Anti-inflammatory and paracrine signaling for symptom control |
| Clinical Goal | Disease modification and joint preservation | Symptom management and surgery postponement |
| Risks | Minor local swelling or pain | Greater procedural discomfort, especially in older patients using autologous cells |
For late-stage patients, the quality of MSCs is a critical factor. Older individuals, who often have advanced OA, may experience reduced capacity in their own (autologous) MSCs. In such cases, allogeneic (donor) MSCs, such as those derived from umbilical cord blood, can be a more practical and effective alternative. This highlights the importance of customizing treatment approaches based on the patient's condition and stage of osteoarthritis.
Conclusion
The timing of mesenchymal stem cell (MSC) therapy plays a critical role in joint repair. Clinical studies consistently reveal that early-stage treatments lead to more noticeable structural improvements compared to interventions in later stages. For many patients treated early, joint conditions remain stable for up to five years, highlighting MSC therapy's potential to slow disease progression when applied promptly.
Looking ahead, advancements in precision medicine aim to fine-tune treatment strategies. The next frontier for MSC therapy involves matching specific patient profiles with tailored cell therapies. Researchers are working to identify unique osteoarthritis (OA) "molecular endotypes", such as inflammatory or metabolic subtypes, and pairing them with MSCs that possess the right "immunomodulatory fitness" for each joint's environment. This approach could significantly improve regenerative outcomes by aligning therapies with individual biological needs.
In addition to optimizing cell-based treatments, innovative cell-free options like MSC-derived exosomes are gaining attention. These exosomes, tiny vesicles engineered to carry therapeutic molecules, may provide similar benefits without the complexities of cell engraftment. By modifying exosomes with targeting peptides, they could deliver treatments directly to damaged cartilage, offering potential benefits for both early and late-stage OA patients.
For those in advanced stages of OA, higher doses of MSCs - up to 100 million cells - primarily focus on symptom relief and delaying surgery. While the emphasis shifts from regeneration to managing pain and postponing surgical interventions, this remains an important goal for millions of individuals worldwide.
As protocols become more refined and standardized, MSC therapy is poised to address a critical gap in osteoarthritis treatment, especially for early-stage patients who currently have limited options beyond lifestyle changes and pain relief.
FAQs
Am I early-stage or late-stage osteoarthritis?
Determining where you are with osteoarthritis often hinges on how severe your symptoms are and the extent of joint damage. In the early stages, you might notice mild cartilage wear, slight narrowing of the joint space, and occasional discomfort or stiffness. On the other hand, late-stage osteoarthritis typically involves major cartilage loss, noticeable joint deformities, constant pain, and significant difficulty with movement. For a clear diagnosis, it's essential to consult a healthcare professional who can evaluate your symptoms and imaging results.
How long do MSC injections usually last?
MSC injections often offer relief that lasts between 6 months and 2 years. However, some individuals report benefits extending 3 to 7 years or more, depending on various factors like the severity of their condition and how their body responds to the treatment. The outcomes can differ significantly from person to person.
Can MSCs help if my knee is bone-on-bone?
Mesenchymal stem cells (MSCs) hold potential for easing symptoms and aiding cartilage regeneration in individuals with bone-on-bone knee osteoarthritis. That said, the research so far is limited, and additional studies are necessary to determine how effective they truly are.
The views, statements, and pricing expressed are deemed reliable as of the published date. Articles may not reflect current pricing, offerings, or recent innovations.