How Scaffold-Based Culturing Expands Cord Blood Stem Cells
Scaffold-based culturing is transforming how we expand cord blood stem cells. The main challenge with cord blood is its low stem cell count, making it insufficient for adult transplants. Traditional 2D methods fail to provide the right environment for stem cell growth, often leading to rapid differentiation and poor expansion rates. Enter 3D scaffolds: these structures mimic the natural bone marrow environment, enabling stem cells to multiply while maintaining their undifferentiated state.
Key takeaways:
- 3D scaffolds outperform 2D cultures: While 2D methods achieve a 2.66-fold increase in CD34+ cells, 3D scaffolds can expand these cells up to 40-fold or more.
- Enhanced cell quality: Cells grown in 3D scaffolds show higher levels of homing markers like CXCR4, improving their ability to integrate into the bone marrow.
- Scalable production: Techniques like 3D bioprinting and perfusion bioreactors make it easier to scale scaffold-based methods for clinical use.
- Broader medical applications: Beyond blood-related conditions, these methods show promise for repairing heart tissue, spinal cord injuries, and more.
This approach addresses the limitations of traditional methods, offering a more effective way to prepare stem cells for cord blood transplants and regenerative medicine.
The Science Behind Scaffold-Based Culturing
What are Stem Cells and Scaffolds?
Scaffolds are three-dimensional structures designed to imitate the natural environment of stem cells, known as the stem cell niche. These structures can be made from synthetic polymers like PCL, PLGA, and PDMS or natural biomaterials such as fibrin, collagen, and alginate-gelatin hydrogels.
One standout feature of scaffolds is their porous design. These interconnected pores allow for the efficient flow of nutrients, oxygen, and waste, creating an ideal setting for cell growth. Unlike flat culture plates, scaffolds offer a larger surface area for cells to attach and interact, closely replicating the intricate geometry of bone marrow tissue. This unique design plays a critical role in how scaffolds support stem cell expansion.
How Scaffolds Work in Stem Cell Expansion
Scaffolds replicate the extracellular matrix (ECM) found in bone marrow, which naturally supports stem cells. The stiffness and elasticity of these structures send signals that help stem cells remain in their undifferentiated state.
"3D scaffolds provide structural support and approve cell-to-cell contact, thus fulfilling the important requirements of the niche."
– Biomaterials Journal
Additionally, scaffolds activate molecular pathways essential for maintaining stem cells. For example, 3D PDMS scaffolds stimulate pathways like SREBP, HIF1α, and FOXO, which are vital for preserving the primitive state of stem cells. Cells grown in these 3D environments also show higher levels of homing markers such as CXCR4, VLA-4, and VLA-5. These markers play a crucial role in guiding transplanted cells back to the bone marrow. This level of functionality is a stark improvement over traditional 2D methods.
Comparison with 2D Culturing Methods
In contrast to 3D scaffolds, 2D cultures restrict cell interactions and nutrient flow, often leading to rapid differentiation.
Research highlights the performance gap between these methods. For instance, while 2D systems achieved only a 2.66-fold increase in CD34+ stem cells, PCL scaffolds coated with fibronectin achieved a 40-fold expansion. Even more striking, 3D polyurethane scaffolds delivered a 54-fold expansion of cord blood cells without requiring cytokines - conditions under which 2D cultures failed within just four days. These results emphasize how the spatial organization, mechanical properties, and cell interactions in a 3D environment are essential for maintaining stem cell potential during expansion.
Step-by-Step Process of Scaffold-Based Stem Cell Expansion
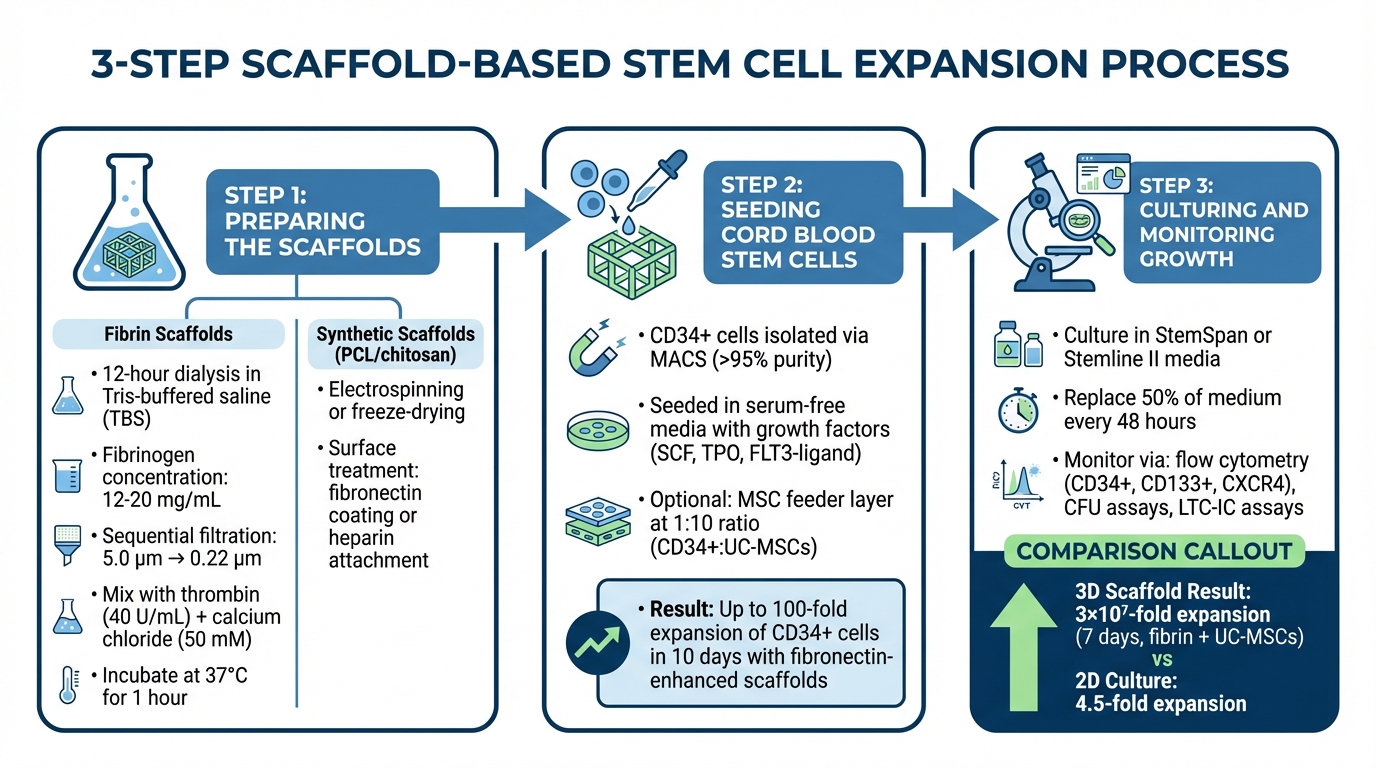
3-Step Scaffold-Based Stem Cell Expansion Process
Preparing the Scaffolds
The process begins with the fabrication and sterilization of scaffolds to ensure a safe environment for cell growth. For fibrin scaffolds, overnight dialysis (at least 12 hours) in Tris-buffered saline is necessary to remove inhibitory citrates. The fibrinogen solution, typically at concentrations of 12–20 mg/mL, undergoes sequential filtration: first through a 5.0 μm syringe filter to remove large particles, followed by a 0.22 μm filter for sterilization.
After sterilization, the fibrinogen is mixed with thrombin (around 40 U/mL) and calcium chloride (50 mM), then incubated at 37°C (98.6°F) for one hour to allow polymerization. Synthetic scaffolds, such as those made from PCL (polycaprolactone) or chitosan, require different techniques. PCL scaffolds are created using precision microfabrication or electrospinning, while chitosan scaffolds are typically produced through freeze-drying. These scaffolds are often treated further to improve cell attachment - fibronectin coatings or covalent attachment of heparin using carbodiimide chemistry are common enhancements.
Once the scaffolds are ready and sterilized, they are seeded with cord blood stem cells in the next step.
Seeding Cord Blood Stem Cells
CD34⁺ cells are isolated using Magnetic-Activated Cell Sorting (MACS), ensuring a purity level above 95%. These cells are then seeded onto the scaffolds in serum-free media containing growth factors like Stem Cell Factor (SCF), Thrombopoietin (TPO), and FLT3-ligand. The natural expression of integrins on the cells facilitates proper adhesion to the scaffold surface.
Some advanced setups use mesenchymal stem cells (MSCs) as a feeder layer. These MSCs can either be seeded onto the scaffold first or encapsulated within it. Research suggests that maintaining a 1:10 ratio of CD34⁺ cells to UC-MSCs (umbilical cord-derived MSCs) is ideal for promoting expansion while avoiding competition for nutrients. Scaffolds enhanced with covalently attached fibronectin have shown impressive results, achieving up to a 100-fold expansion of CD34⁺ cells over 10 days - highlighting the importance of surface preparation.
Culturing and Monitoring Growth
After seeding, maintaining optimal culture conditions is critical for cell growth and preserving stemness. Cells are cultured in serum-free media, such as StemSpan or Stemline II, supplemented with cytokine cocktails. To sustain the environment, half of the medium is replaced with fresh, cytokine-enriched media every 48 hours.
Growth is monitored using electronic cell counters or manual hemacytometers with trypan blue exclusion to measure total cell counts and viability. Flow cytometry is used to track stemness markers like CD34⁺ and CD133⁺, along with homing receptors such as CXCR4. Functional quality is assessed through Colony-Forming Unit (CFU) assays in methylcellulose and Long-Term Culture-Initiating Cell (LTC-IC) assays.
For example, one study demonstrated that 3D fibrin scaffolds supported by UC-MSCs achieved a 3 x 10⁷-fold expansion of CD34⁺ cells within seven days. Similarly, microfluidic systems with fibronectin-coated scaffolds produced an 8.5-fold expansion of CD133⁺ cells over 12 days, significantly outperforming the 4.5-fold expansion observed in traditional 2D cultures.
Benefits of Scaffold-Based Culturing
Higher Expansion Rates and Cell Quality
3D fibrin scaffolds, when paired with umbilical cord-derived mesenchymal stem cells, have achieved a 3 × 10⁷-fold expansion of CD34+ cells in just seven days - more than enough for adult transplantation needs. Compared to traditional 2D methods, 3D scaffolds significantly outperform, with PCL scaffolds coated with fibronectin yielding a 58-fold total cell expansion and a 40-fold increase in CD34+ cells.
But it’s not just about producing more cells - the quality of these cells is equally critical. 3D scaffolds activate key signaling pathways like SREBP, HIF1α, and FOXO, which help maintain the pluripotency and undifferentiated state of long-term hematopoietic stem cells. Cells grown in these 3D environments show enhanced morphology, migration, and adhesion properties, along with higher CXCR4 expression, a marker crucial for stem cell "homing" to the bone marrow post-transplantation. Notably, materials like polyurethane coated with collagen type I achieved a 54-fold expansion without the need for external cytokines, highlighting the efficiency of these biomimetic conditions.
Medical Applications for Transplants
The superior cell yields from scaffold-based methods directly address one of the biggest challenges with cord blood: low initial cell counts. This progress paves the way for treating current and future stem cell applications, including chemoresistant high-risk malignancies, bone marrow failures, hemoglobinopathies, immune deficiencies, and autoimmune diseases. Beyond hematological treatments, these techniques hold potential for regenerating bone, cartilage, ligaments, and skeletal muscle.
Cardiovascular and neurological applications are particularly promising. Combining 3D scaffolds with stem cells aids in repairing myocardial infarctions by promoting angiogenesis and improving cardiomyocyte survival. In spinal cord injuries and stroke, these methods encourage axon regeneration and protect remaining neurons. The increased CXCR4 expression in 3D-expanded cells leads to better multi-organ engraftment and multilineage differentiation in animal studies compared to cells cultured in 2D. These advancements not only improve transplant outcomes but also highlight the broader potential of scaffold-based methods in regenerative medicine.
Scalability for Future Applications
As clinical demand grows, scalable production methods are becoming a priority. Scaffold-based culturing is moving beyond research labs, thanks to automated fabrication technologies. Techniques like 3D bioprinting - using extrusion-based, droplet-based, or photocuring methods - allow for precise, computer-aided scaffold designs with consistent and reproducible results. Electrospinning, a nanofiber fabrication method, offers a cost-efficient approach while enabling precise control over fiber dimensions to mimic the extracellular matrix.
Integration with 3D perfusion bioreactors ensures continuous monitoring and consistent cell proliferation, which is critical for large-scale production. The flexibility of scaffold materials - ranging from synthetic polymers like PCL and PLGA to natural substances like collagen and fibrin - allows customization for different tissues and organs. Beyond transplantation, these systems are also being developed for drug discovery and studying bone marrow niche dynamics in vitro. As automation and precision manufacturing evolve, scaffold-based culturing is set to play a central role in advancing cord blood therapies and regenerative medicine.
Conclusion
Scaffold-based culturing tackles one of the biggest challenges in cord blood use: the limited number of initial cells. By replicating the natural 3D structure of bone marrow, these systems deliver far better expansion results than traditional methods. For example, one study reported an impressive 3×10⁷-fold increase in CD34⁺ cells within just seven days using fibrin scaffolds[1]. Even more importantly, these expanded cells maintain their quality, including improved homing abilities and an undifferentiated state - key factors for successful transplantation.
The potential applications of these advancements go well beyond treating blood-related conditions. Expanded cord blood cells are showing promise in addressing cancers, immune deficiencies, and other serious disorders. Clinical studies have highlighted this potential, with one reporting an 86% disease-free survival rate when using expanded cord blood units[2]. These results underline the growing role of these methods in improving patient outcomes.
For families considering if stem cell banking is worth it, these innovations add a new layer of value. A single cord blood collection, once limited in its therapeutic scope, could now be expanded to support treatments for adults or even multiple therapies over a lifetime. Companies like Americord Registry are prepared to support these advancements, offering families the ability to store cord blood that remains adaptable for future expansion technologies.
With breakthroughs like 3D bioprinting making scaffold-based culturing more accessible, the transition from research to real-world application is accelerating. Combining reliable storage solutions with cutting-edge expansion techniques is turning cord blood into a powerful resource for regenerative medicine, paving the way for transformative treatments in the years ahead.
FAQs
Are 3D-expanded cord blood stem cells safe for transplants?
Research indicates that 3D-expanded cord blood stem cells are safe for transplantation. Studies have shown they grow effectively in scaffold-based systems without any reported safety concerns. These developments point to their promise in regenerative medicine therapies.
How do scaffolds help stem cells stay undifferentiated?
Scaffolds play a key role in keeping stem cells undifferentiated by imitating their natural surroundings. These structures provide both physical and biochemical signals that influence how cells behave. By creating a 3D framework similar to the cells' in vivo environment, scaffolds support critical interactions between cells and their surrounding matrix - interactions that are vital for maintaining stemness.
Key features like porosity, stiffness, and surface texture, combined with biochemical elements such as growth proteins, work together to encourage self-renewal. At the same time, they help prevent differentiation, ensuring the cells remain in their multipotent state. This preservation is crucial for their potential use in therapeutic applications.
Does cord blood banking matter if cells can be expanded later?
Cord blood banking remains important because it preserves original stem cells, which serve as a dependable resource for both current and potential medical treatments. Although advancements in cell expansion techniques are underway, these methods are not yet perfected, highlighting the continued value of storing original stem cells for therapeutic use.
The views, statements, and pricing expressed are deemed reliable as of the published date. Articles may not reflect current pricing, offerings, or recent innovations.