Umbilical Cord Stem Cells for Knee Osteoarthritis
Umbilical Cord Stem Cells for Knee Osteoarthritis
Can umbilical cord stem cells (UC-MSCs) help with knee osteoarthritis? Yes, they show promise in reducing pain, improving mobility, and potentially regenerating cartilage. Unlike traditional treatments like NSAIDs or hyaluronic acid injections, which only manage symptoms, UC-MSCs may address the root cause by repairing cartilage and reducing inflammation.
Key Points:
- Why it matters: Knee osteoarthritis affects 27 million Americans and costs over $185 billion annually. With an aging population, effective treatments are urgently needed.
- What UC-MSCs are: These stem cells, derived from umbilical cord tissue (Wharton's jelly), are collected safely after childbirth. They have high regeneration potential and a low risk of immune rejection.
- How they work: UC-MSCs reduce inflammation, promote cartilage repair, and release growth factors that protect joint tissues.
- Clinical results: Studies show reduced pain (VAS scores dropping from 50.7 to 17.7) and improved joint function (WOMAC scores improving from 39.3 to 11.0). Side effects are mild and temporary, such as minor swelling.
- Advantages: Collection is painless, and these cells have higher proliferation rates compared to bone marrow or adipose-derived stem cells.
UC-MSCs offer a forward-looking option for managing knee osteoarthritis, especially when combined with other therapies like hyaluronic acid or surgical techniques. With a strong safety profile and promising outcomes, they could reshape how we treat joint degeneration.
What Are Umbilical Cord Stem Cells?
Umbilical Cord MSCs vs Other Stem Cell Sources for Knee Osteoarthritis Treatment
Umbilical cord stem cells are undifferentiated cells collected from the umbilical cord and placenta after a live birth. The process is completely painless and safe for both mother and baby. These cells are particularly effective in addressing cartilage issues, such as those found in osteoarthritis.
There are two main types to know about. Hematopoietic and Mesenchymal Stem Cells play different roles; HSCs, found in cord blood, generate blood and immune cells, while MSCs, derived from the cord tissue's Wharton's jelly, can develop into cartilage, bone, muscle, and nerve cells. For treating knee osteoarthritis, MSCs from Wharton's jelly are the ones making waves.
Umbilical cord tissue contains 10 times more stem cells than the same amount of adult bone marrow. Between 2019 and 2023, 87% of perinatal MSC trials used cord tissue, and over 4,000 cord blood transplants are performed annually. Since the first successful transplant in 1988, more than 40,000 cord blood transplants have taken place worldwide. These numbers highlight their potential for regenerative treatments.
Biological Characteristics of Umbilical Cord Stem Cells
Umbilical cord MSCs offer a multi-faceted regenerative approach that goes beyond what traditional treatments can achieve.
These cells can renew themselves, transform into various cell types like bone and cartilage, and release signaling molecules that help repair cartilage and reduce inflammation. They produce cytokines, chemokines, and growth factors that encourage cartilage regeneration and prevent cell death in chondrocytes.
UC-MSCs also reduce inflammatory markers like IL-6, IL-8, and TNF-α, while lowering enzymes that break down cartilage (MMP-3, MMP-13). At the same time, they boost anabolic markers like COL2A1 and TGF-β1. One 12-week study showed that UC-MSC secretions increased TGF-β1 levels by an average of 10,221.60 pg/mL.
"Human umbilical cord mesenchymal stem cells (hUC-MSCs) are a promising source... particularly for the treatment of osteoarthritis (OA), a degenerative joint disease characterized by inflammation and cartilage damage." - Journal of Translational Medicine
These MSCs can also shift macrophages in the joint to a state that reduces inflammation, creating a more balanced environment for healing. They're considered "immunologically privileged", meaning they are less likely to trigger immune rejection. Clinical trials involving 385 knees found no severe adverse effects from hUC-MSC injections.
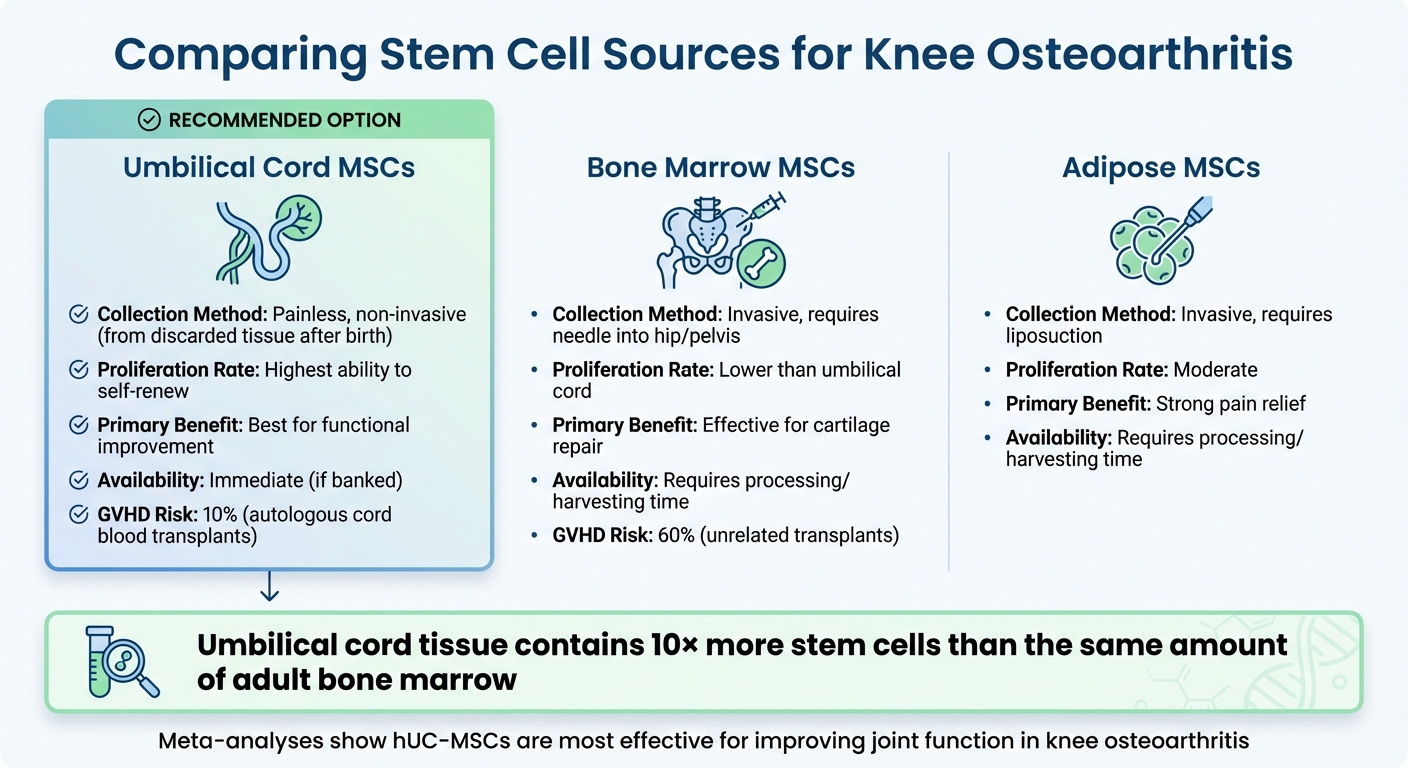
Umbilical Cord MSCs Compared to Other Stem Cell Sources
UC-MSCs not only excel in their regenerative properties but also offer practical advantages over other types of stem cells.
Their collection is painless and uses tissue that would otherwise be discarded, unlike bone marrow MSCs, which require a painful extraction from the hip or pelvis, or adipose MSCs, which need liposuction. Additionally, Graft-Versus-Host Disease (GVHD) occurs in about 60% of unrelated bone marrow transplants but only 10% of autologous cord blood transplants.
| Feature | Umbilical Cord MSCs | Bone Marrow MSCs | Adipose MSCs |
|---|---|---|---|
| Collection | Painless, non-invasive (from discarded tissue) | Invasive, requires needle into hip/pelvis | Invasive, requires liposuction |
| Proliferation | Highest ability to self-renew | Lower than umbilical cord | Moderate |
| Primary Benefit | Better functional improvement | Effective for cartilage repair | Strong pain relief |
| Availability | Immediate (if banked) | Requires processing/harvesting time | Requires processing/harvesting time |
Meta-analyses suggest that while adipose MSCs may provide better pain relief, hUC-MSCs are often the best for improving joint function, thanks to their anti-inflammatory and cartilage-repairing effects. A February 2024 study compared 20 knees treated with hUCB-MSCs to 19 knees treated with bone marrow aspirate concentrate (BMAC). Despite the hUCB-MSC group being older and having larger lesions, the 12-month follow-up showed no significant differences in pain or function scores between the two groups.
"Umbilical cord-derived mesenchymal stem cells (hUC-MSCs) might be the most effective for improving function among other MSCs in knee osteoarthritis injection treatment." - Karol Pałka, Department of Biochemistry, Medical University of Silesia
Since UC-MSCs come from tissue that would otherwise be discarded, their use avoids ethical concerns. These cells are also "younger" and less likely to carry damage from environmental exposure or replication errors compared to adult stem cells. Once processed, they can be cryopreserved in liquid nitrogen and remain viable for decades - some have been successfully transplanted after 18 years in storage.
Clinical Studies on Umbilical Cord MSCs for Knee Osteoarthritis
Trial Results and Patient Outcomes
Recent clinical trials have explored how human umbilical cord mesenchymal stem cells (hUC-MSCs) can help manage knee osteoarthritis. The results are promising: across seven studies, VAS scores dropped from 50.7 to 17.7, while WOMAC scores improved from 39.3 to 11.0, showing a significant reduction in pain and improved joint function.
The delivery method seems to play a key role in outcomes. For general pain relief and improved mobility, intra-articular injections are effective. However, for more severe cases involving full-thickness cartilage defects, surgical implantation has shown better results. A notable example is the Cartistem product by MEDIPOST, which uses a surgical technique involving small holes (around 4 mm in diameter) drilled into the subchondral bone. These are then filled with a mixture of MSCs and hyaluronic acid. At the 12-month mark, this method achieved an ICRS cartilage repair score of 9.41, outperforming traditional microdrilling, which scored 7.94.
In a Phase I study, patients receiving four weekly injections experienced significant improvements in both VAS and WOMAC scores. This suggests that repeated dosing may provide better long-term benefits, even though 35.7% of patients reported temporary joint pain and swelling. These side effects resolved on their own without requiring treatment.
Interestingly, a Phase 2/3 trial found that hUC-MSC injections were comparable to corticosteroids after 12 months. While this shows their potential, it also highlights the importance of combining treatments to achieve more durable cartilage regeneration. Cartilage repair outcomes, however, remain inconsistent. Some studies show measurable increases in cartilage thickness and improved MOCART scores - one trial reported a jump from 42.0 to 58.0 over 12 weeks - but others found no significant structural changes visible on MRI.
The table below provides a comparison of key studies, delivery methods, dosing strategies, and outcomes, showcasing the potential of hUC-MSCs in treating knee osteoarthritis.
Study Comparison Table
| Study/Product | Delivery Method | Dosing Strategy | Key Outcome | Follow-Up |
|---|---|---|---|---|
| Cartistem (MEDIPOST) | Surgical (drilling + implantation) | Single application during surgery | Superior cartilage repair (ICRS 9.41 vs 7.94 for microdrilling) | 12 months |
| Cellistem OA | Intra-articular injection | Baseline + 6-month dose | Repeated dosing led to better pain reduction | 23.4 months avg. |
| Phase I Study (Ao et al.) | Intra-articular injection | 4 weekly injections | VAS improved from 6.0 to 3.5; WOMAC improved from 26.0 to 8.5 | 12 weeks |
| Dilogo et al. | Intra-articular injection | 1 MSC dose + 2 HA doses (weekly) | Significant pain reduction in severe cases by 6 months | 6 months |
Safety and Patient Experiences
Safety Data and Side Effects
Umbilical cord MSCs have shown a strong safety profile across various clinical trials. Studies monitoring patients for up to 48 months have reported no serious adverse events, treatment-related deaths, or dose-limiting toxicities.
The most common side effects are minor and temporary. These include knee swelling (effusion) and mild pain at the injection site, both of which typically resolve on their own within a few days.
A US-based Phase 1/2a study, led by Brian J. Cole and Andreas H. Gomoll, followed 12 patients for 24 months after CARTISTEM implantation. The study recorded zero serious adverse events and no dose-limiting toxicities. While all patients experienced a temporary decrease in range of motion, and 92% reported joint pain shortly after surgery, these effects were categorized as mild to moderate (Grade 1-2).
"No serious adverse effects were observed in any of the studies. The most common adverse effect was knee effusion, which resolved spontaneously in all cases."
– Mohd Ishak-Samrin et al., Department of Orthopaedic and Traumatology, Universiti Kebangsaan Malaysia
Long-term safety evaluations have also been reassuring. Over three years of post-treatment monitoring, there has been no evidence of tumor formation, joint deformity, or immune rejection. Serum tumor markers remained stable, further supporting the therapy's safety.
Patient-Reported Outcomes
The strong safety record is matched by positive patient-reported outcomes, emphasizing the functional benefits of umbilical cord MSC treatment. For example, in a study conducted at Southwest Hospital, quality of life scores (measured by the SF-12) improved from 39.0 at baseline to 46.0 within 12 weeks.
These improvements extend to real-world benefits as well. At Gangnam JS Hospital in Seoul, researchers tracked 128 patients treated with CARTISTEM for full-thickness cartilage lesions between January 2014 and December 2015. Over an average follow-up of 36.1 months:
- VAS pain scores dropped from 7.0 to 2.0.
- WOMAC scores improved from 39.3 to 13.9.
- IKDC scores increased from 32.5 to 61.2.
Importantly, the study reported no infections, allergic reactions, or postoperative complications during the minimum two-year observation period.
"Patients undergoing treatment of knee OA with hUC-MSCs might be expected to experience improvements in clinical outcomes."
– Jaydeep Dhillon, BS, Rocky Vista University College of Osteopathic Medicine
With only minor, short-term side effects and substantial functional improvements, newborn stem cell therapy has become an appealing option for those seeking relief from knee osteoarthritis symptoms.
Future Research and Combination Therapies
Combining Umbilical Cord MSCs with Other Treatments
Umbilical cord MSCs show improved results when paired with other therapies. These strategies aim to tackle the challenges of knee osteoarthritis by enhancing joint stability and promoting regeneration. One of the most common additions is hyaluronic acid (HA), which acts as both a lubricant and a scaffold, helping the cells stay within the joint.
From January 2014 to December 2015, Seok Jung Kim at Gangnam JS Hospital in Seoul treated 128 patients with CARTISTEM® combined with hyaluronic acid. The procedure involved creating 4-millimeter-diameter holes in the subchondral bone at 2-millimeter intervals to implant the mixture. After at least two years of follow-up, cartilage repair scores (MOCART) improved significantly, rising from an average of 30.58 to 55.44.
At Cipto Mangunkusumo Hospital, Ismail Hadisoebroto Dilogo treated 29 patients (57 knees) with a combination therapy: an initial injection of 10 million hUC-MSCs in secretome mixed with 2 mL of hyaluronic acid, followed by two weekly 2 mL HA doses. By the six-month mark, patients experienced reduced VAS scores, along with better IKDC and WOMAC scores.
For patients with severe joint misalignment, combining MSC therapy with High Tibial Osteotomy (HTO) has shown potential. This surgical technique shifts the axial load away from damaged cartilage, reducing mechanical stress and supporting the regenerative treatment.
"If there is an alignment deformity during allogenic hUCB-MSC implantation, we recommend conducting high tibial osteotomy to shift the axial load effectively."
– Seok Jung Kim, Gangnam JS Hospital
Researchers are also investigating alternative delivery methods. A study published in May 2025 highlighted that subchondral injections improved trabecular separation and number more effectively than intra-articular injections, suggesting a benefit in targeting the bone directly. Another promising approach involves repeated dosing schedules. A Phase I study from June 2023 revealed that four weekly intra-articular injections of 15 million UC-MSCs significantly reduced mean VAS scores (from 6.0 to 3.5) and WOMAC scores (from 26.0 to 8.5) over three months.
These combination approaches naturally pave the way for broader regenerative applications, including personalized biobanking services, which ensures access to high-quality therapeutic cells for future treatments.
Stem Cell Banking for Regenerative Medicine
Securing high-quality stem cells early in life can ensure better treatment options in the future. Cord tissue banking offers a personalized resource for regenerative therapies, preserving cells at their peak potential, regardless of the donor's age, lesion size, or BMI.
The Gangnam JS Hospital study highlighted a key benefit of cord-derived cells: their effectiveness is unaffected by the patient's age, lesion size, or BMI. This means cells banked at birth can remain therapeutically viable for decades.
Americord Registry provides families with cord blood and tissue banking services, offering CryoMaxx™ Processing to maximize stem cell yield. These services start at $3,799, with payment plans as low as $145 per month, making them accessible for families planning for future regenerative needs.
"hUCB-MSCs have advantages of non-invasive cell collection, high capacity for expansion, and low immunogenicity for therapeutic applications as an off-the-shelf allogeneic product."
– Nature Scientific Reports, 2024
With knee osteoarthritis affecting approximately 251 million people worldwide - and the number of older adults with OA expected to triple by 2050 - cord tissue banking provides a forward-looking solution. The collection process is non-invasive, using tissue that would otherwise be discarded, and the cells can be cryopreserved without losing their regenerative potential.
Conclusion
Umbilical cord–derived mesenchymal stem cells (UC-MSCs) are transforming the treatment of knee osteoarthritis. Unlike conventional therapies that focus on symptom relief, UC-MSCs address the root cause by repairing cartilage, reducing inflammation, and protecting tissues from further damage. Clinical studies involving 385 patients have demonstrated measurable improvements in key metrics like WOMAC, IKDC, and VAS scores, with benefits lasting up to 48 months.
Reported side effects, such as mild joint swelling or temporary discomfort, were minimal and resolved on their own. The collection process is also non-invasive, eliminating the pain often associated with harvesting bone marrow or adipose tissue.
"WJ-MSCs have good freeze–thaw properties and can be frozen in liquid nitrogen... and provides a good theoretical basis for the establishment of a 'clinical resource bank' in the future." - Human Cell
The ability of these cells to withstand freeze–thaw cycles makes UC-MSC treatments scalable. This opens the door to creating "off-the-shelf" solutions from banked umbilical cord tissue. With just 1 million cells, expansion to 10 billion is possible within four weeks, ensuring a sufficient supply for clinical use. Considering the global burden of osteoarthritis - impacting over 595 million people - and the $185 billion annual cost in the U.S. alone, banking cord blood and tissue at birth offers families a proactive way to access regenerative therapies.
Americord Registry supports this vision by offering clinically validated banking services. Starting at $3,799, with payment plans as low as $145 per month, families gain access to CryoMaxx™ Processing, which optimizes stem cell yield. Banked cells retain their therapeutic potential regardless of factors like age, BMI, or lesion size, ensuring future treatment options remain available. This service provides a practical and forward-thinking approach to securing regenerative care for your family.
FAQs
What makes umbilical cord stem cells a promising option for treating knee osteoarthritis?
Umbilical cord-derived stem cells (UC-MSCs) are gaining attention as a potential treatment for knee osteoarthritis, offering the ability to repair cartilage and alleviate symptoms. Research highlights their safety and effectiveness, showing they can aid cartilage regeneration, enhance joint function, and reduce pain levels.
When compared to stem cells derived from adipose tissue (AD-MSCs), UC-MSCs are often noted for their regenerative properties and favorable safety profile. While both types show promise, UC-MSCs are particularly recognized for their potential to support long-term joint health. Researchers are actively investigating the full range of benefits these stem cells may offer for osteoarthritis management.
What are the possible side effects of using umbilical cord stem cells to treat knee osteoarthritis?
Using umbilical cord stem cells for knee osteoarthritis treatment is generally regarded as safe, but it’s important to be aware of possible side effects. These might include temporary swelling, pain or discomfort at the injection site, or mild inflammation. Although rare, there’s a small chance of complications such as infection or an immune response.
Clinical studies indicate that most patients tolerate this therapy well, with adverse reactions being uncommon. If you’re thinking about this option, make sure to consult a healthcare professional to discuss the potential risks and benefits specific to your health situation.
Can umbilical cord stem cells be used alongside other treatments to improve knee osteoarthritis outcomes?
Yes, umbilical cord stem cells can be used alongside other treatments to potentially boost their effectiveness in addressing knee osteoarthritis. For instance, researchers have investigated combining these stem cells with hyaluronate-based hydrogels, which may aid in joint repair and help alleviate pain. Additionally, some studies point to the benefits of pairing stem cell therapy with procedures like high tibial osteotomy, which might enhance recovery and deliver better long-term outcomes.
Although these combinations are promising, more research is essential to fine-tune these methods and determine the most effective strategies for treating knee osteoarthritis. It's important to consult with a healthcare professional to find the right approach for your individual situation.
The views, statements, and pricing expressed are deemed reliable as of the published date. Articles may not reflect current pricing, offerings, or recent innovations.