Umbilical Cord Blood in Alzheimer's Treatment
Umbilical cord blood-derived stem cells are showing promise in Alzheimer's research, offering a potential new way to address the disease. Unlike current drugs that only manage symptoms, these stem cells work by releasing proteins and growth factors that may help repair brain damage. Early studies suggest they can reduce inflammation, support neuron growth, and improve memory in animal models. Clinical trials in humans have demonstrated safety, with some evidence of benefits like reduced amyloid plaques and improved brain function. As Alzheimer's continues to affect millions and current treatments fall short, cord blood stem cells could represent a step forward in tackling this complex condition. Is cord blood banking worth it for your family? Banking at birth may offer future access to these emerging therapies.
Alzheimer's Disease and Current Treatment Limitations
What Is Alzheimer's Disease?
Alzheimer's is a disease that progressively damages the brain, leading to memory loss and cognitive decline. This happens because of two key processes in the brain: beta-amyloid plaques (protein fragments that build up between nerve cells) and neurofibrillary tangles (twisted fibers of tau protein inside the cells). These changes often begin in the hippocampus and entorhinal cortex - regions vital for memory - before spreading to other parts of the brain.
As neurons lose their connections and die, the brain physically shrinks. What starts as mild forgetfulness can progress into severe issues with language, reasoning, and even basic social interactions. Over time, individuals may lose the ability to perform essential tasks like walking or swallowing. While most people live four to eight years after diagnosis, some can survive up to 20 years. Interestingly, the disease can develop silently, with biological changes occurring 10 to 20 years before symptoms become noticeable. These complexities make treating and reversing Alzheimer's an enormous challenge.
Why Current Treatments Fall Short
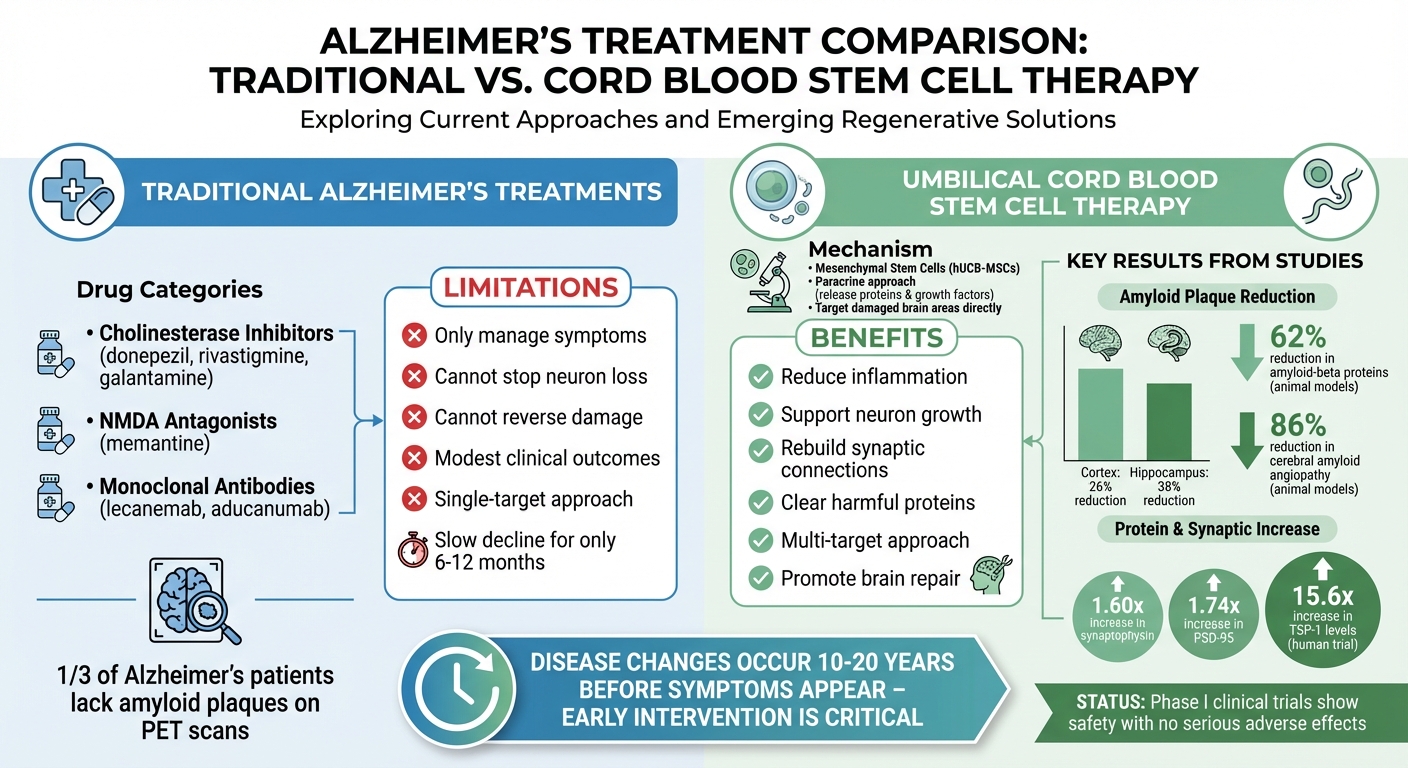
The drugs currently approved, such as cholinesterase inhibitors (donepezil, rivastigmine, galantamine) and NMDA antagonists (memantine), aim to manage symptoms but cannot stop or reverse the loss of neurons. Even newer treatments like monoclonal antibodies (lecanemab, aducanumab), which clear amyloid plaques from the brain, show only modest effects in clinical outcomes. Strikingly, about one-third of Alzheimer's patients lack amyloid plaques on PET scans, highlighting that the disease is more complex than initially thought. Many amyloid-focused treatments have failed to improve cognitive function in trials.
Another major limitation is timing - treatments are often introduced after significant neuronal damage has already occurred, leaving little room for repair. Alzheimer's involves a variety of factors, including genetic, inflammatory, metabolic, and oxidative processes, making it difficult for single-target drugs to address the full scope of the disease. This has spurred growing interest in regenerative Alzheimer's disease and stem cell medicine approaches that aim to restore lost neural function.
Current medications that target tau protein tangles and Aβ plaques are not very effective because they only slow the symptoms of AD and do not repair damaged cells.
sbb-itb-df90ce4
How Umbilical Cord Blood Stem Cells Work
Properties of Cord Blood Stem Cells
Umbilical cord blood is rich in mesenchymal stem cells (hUCB-MSCs), which function differently compared to traditional medications. Rather than transforming into brain cells, these stem cells release proteins and growth factors that promote repair - a process known as the "paracrine" approach. Interestingly, the cells don’t need to survive long-term; their therapeutic molecules do the work before the cells naturally disappear.
What makes these cells stand out is their ability to migrate directly to damaged areas of the brain. They specifically target regions affected by amyloid-beta deposits and inflammation, releasing key molecules like soluble intracellular adhesion molecule-1 (sICAM-1) to stimulate microglia activity and Thrombospondin-1 (TSP-1) to help rebuild synaptic connections. These targeted actions highlight their potential in treating conditions like Alzheimer's.
How These Cells May Help Treat Alzheimer's
Thanks to their unique mechanisms, hUCB-MSCs bring a comprehensive approach to Alzheimer's treatment. early clinical research has begun to uncover the benefits of cord blood and its therapeutic potential. For instance, a Phase I trial conducted at Samsung Medical Center involved nine patients with mild-to-moderate Alzheimer's. Each patient received three injections of hUCB-MSCs (NEUROSTEM®) directly into their cerebrospinal fluid using an Ommaya reservoir. The results confirmed the treatment's safety, with one patient experiencing a 15.6-fold increase in TSP-1 levels, showcasing the cells' ability to deliver protective factors.
Animal studies have provided even more compelling evidence. At New York University Langone Medical Center, researchers injected human umbilical cord stem cells into APP/PS1 transgenic mice. The results? A 26% reduction in amyloid plaques in the cortex and a 38% reduction in the hippocampus, along with improved memory performance. Another study using 5XFAD mice showed that hUCB-MSC transplantation led to a 1.60-fold increase in synaptophysin and a 1.74-fold increase in PSD-95, both of which are critical for healthy synaptic connections. These changes occurred in just four weeks, underscoring the cells' ability to restore neural connections.
"hUCB-MSCs can attenuate Aβ42-induced synaptic dysfunction by regulating TSP-1 release, thus providing a potential alternative therapeutic option for early-stage AD." - Dong Hyun Kim, Researcher, Medipost Co., Ltd.
Clinical Trials and Research Findings
Recent clinical studies have built on preclinical evidence, offering a clearer picture of the potential for cord blood stem cells in Alzheimer's treatment.
Results from Animal Studies
At NYU School of Medicine, researchers led by Allal Boutajangout conducted a pivotal study using APP/PS1 mice (aged 9–10 months). They injected 500,000 human umbilical cord stem cells into the carotid artery after opening the blood-brain barrier with mannitol. This innovative approach improved cell delivery to damaged brain areas. The treated mice showed better performance on Novel Object Recognition tests, indicating enhanced short-term memory.
Meanwhile, researchers at Samsung Medical Center and MEDIPOST explored the mechanisms behind these improvements in 5XFAD mice. They demonstrated that injections into the cisterna magna triggered hippocampal neurogenesis, driven by GDF-15 secretion. The treatment also increased synaptic density markers. Interestingly, only 5.7% of the injected human MSCs remained in the brain after one week, suggesting the benefits stem from secreted factors rather than the cells' long-term presence.
"Human umbilical cord mesenchymal stem cells appear to reduce AD pathology in a transgenic mouse model as documented by a reduction of the amyloid plaque burden compared to controls." - Allal Boutajangout, NYU Langone Medical Center
These animal studies have laid the groundwork for advancing to human clinical trials. These stem cell treatments represent a significant shift in neurodegenerative research.
Current Clinical Trials in Humans
Samsung Medical Center in Seoul, South Korea, has spearheaded two Phase I clinical trials to evaluate NEUROSTEM, a hUCB-MSC drug product.
The first trial (NCT01297218), published in 2015, involved stereotactic injections into the bilateral hippocampi and right precuneus. Nine patients received either a low dose (3.0 × 10⁶ cells) or a high dose (6.0 × 10⁶ cells). Over a 24-month period, the treatment was deemed safe, with no reports of fever or serious side effects.
The second trial (NCT02054208), conducted between March 2014 and June 2017, used intracerebroventricular (ICV) delivery via an Ommaya reservoir. Nine patients with mild-to-moderate Alzheimer's received three injections spaced four weeks apart. The procedure was also safe, with all participants experiencing only temporary fevers that resolved within 24–36 hours. Cerebrospinal fluid (CSF) analysis showed a temporary rise in white blood cell counts one day after injection, which normalized within four weeks. A transient reduction in CSF levels of Aβ42, total tau, and phosphorylated tau was also observed, returning to baseline within four weeks.
| Trial Route | Identifier | Participants | Key Safety Findings |
|---|---|---|---|
| Stereotactic Injection | NCT01297218 | 9 Patients | No fevers; no structural abnormalities over 24 months |
| ICV (Ommaya Reservoir) | NCT02054208 | 9 Patients | Temporary fevers resolved within 36 hours |
| ICV Extended Follow-up | NCT03172117 | Ongoing | Evaluating long-term safety up to 36 months |
Cord Blood Banking for Future Treatment Options
Benefits of Banking Cord Blood
Banking cord blood at birth could open doors to future regenerative therapies, including those for Alzheimer's. Since cord blood can only be collected during childbirth, planning ahead to preserve its mesenchymal stem cells is crucial.
Umbilical cord blood is a rich source of mesenchymal stem cells, known for their stability and adaptability. Interestingly, only about 20% to 25% of patients in need of a stem cell transplant find a matching donor within their family. This highlights the versatility of cord blood for a wide range of treatments.
Research into cord blood isn’t limited to Alzheimer's. Studies are also exploring its potential in addressing conditions like cerebral palsy, hydrocephalus, and multiple sclerosis. Globally, over 4,000 cord blood transplants are performed annually, and properly stored cord blood can remain viable for decades. This longevity makes it a promising resource for future medical breakthroughs.
Americord Registry Services
Americord Registry offers a comprehensive approach to newborn stem cell banking. Accredited by the AABB, the company uses CryoMaxx™ Processing to optimize stem cell recovery and maintain their viability. Their advanced 5-compartment storage vials allow for multiple treatments from one collection - a valuable feature if future therapies require repeated doses.
Parents can choose from a variety of options, including cord blood, cord tissue, and placental tissue banking, as well as exosomes. These sources are particularly rich in mesenchymal stem cells, which are being studied for their anti-inflammatory and regenerative properties in Alzheimer's research. Americord prioritizes transparency in pricing and provides personalized support to ensure families understand their options well before delivery.
Conclusion
The potential of umbilical cord blood stem cells in addressing Alzheimer's is impressive. Studies show these cells can reduce amyloid-beta proteins by 62% and cerebral amyloid angiopathy by 86% in animal models - far surpassing the effects of current FDA-approved drugs, which only slow decline for 6–12 months[1].
Clinical trials at Samsung Medical Center further highlight their promise. Delivering these cells directly into the brain has proven both safe and practical, with no serious adverse effects reported during 24- and 36-month follow-ups[2]. These stem cells seem to work by releasing protective factors that lower inflammation, clear harmful proteins, and aid the brain's natural repair mechanisms.
Given these advancements, storing cord blood at birth offers families a forward-thinking approach to future regenerative treatments. As research progresses, cord blood stem cells stand out as a safe and ethical option for tackling one of medicine's toughest challenges. For expectant families, considering cord blood banking through comparing cord blood banks like Americord Registry could be a meaningful step toward accessing emerging therapies in the years to come.
FAQs
How soon could cord blood stem cells be available for Alzheimer’s patients?
Cord blood stem cells could soon offer hope to Alzheimer’s patients, possibly within the next few years. Researchers, including those at the University of Miami, are conducting clinical trials to evaluate their safety and how well they work. Some of these trials have already wrapped up, while others are still in progress. These efforts are crucial in shaping when this treatment might become more widely accessible.
Who might qualify for cord blood stem cell trials for Alzheimer’s?
Patients diagnosed with Alzheimer’s dementia, especially those in the early to moderate stages, might be eligible for cord blood stem cell trials. Eligibility typically hinges on several factors, including age, general health, the absence of serious additional health conditions, and the capacity to undergo specific procedures like intracerebroventricular injections. Potential participants undergo thorough screening to ensure they meet the necessary requirements. It’s crucial to consult with healthcare professionals to assess individual suitability for these trials.
Does banking cord blood now actually help my family later?
Banking umbilical cord blood today offers a way to potentially support your family’s health in the future, especially as advancements in stem cell therapies continue to grow. Current studies indicate that stem cells from cord blood could play a role in slowing or even reversing certain effects of Alzheimer’s disease. By preserving cord blood with Americord Registry, you secure access to these cells, which might one day be used in regenerative treatments for conditions like Alzheimer’s and other neurodegenerative disorders.
The views, statements, and pricing expressed are deemed reliable as of the published date. Articles may not reflect current pricing, offerings, or recent innovations.