Umbilical Cord Stem Cells in Cardiovascular Treatments
Umbilical cord stem cells are transforming heart disease treatment by focusing on repair rather than symptom management. These cells, collected safely after childbirth, can regenerate heart muscle, form blood vessels, and reduce inflammation. They are fast-dividing, immune-friendly, and deliver healing factors to damaged tissue. Studies show improved heart function, reduced scar tissue, and better recovery in both animal models and human trials. Collection at birth through stem cell banking ensures immediate access to these therapies, offering hope for millions living with heart conditions.
Key Properties of Umbilical Cord Stem Cells for Heart Repair
Umbilical cord stem cells offer unique abilities that support heart tissue repair through various mechanisms, making them a promising tool for cardiovascular therapy.
Differentiation and Tissue Regeneration
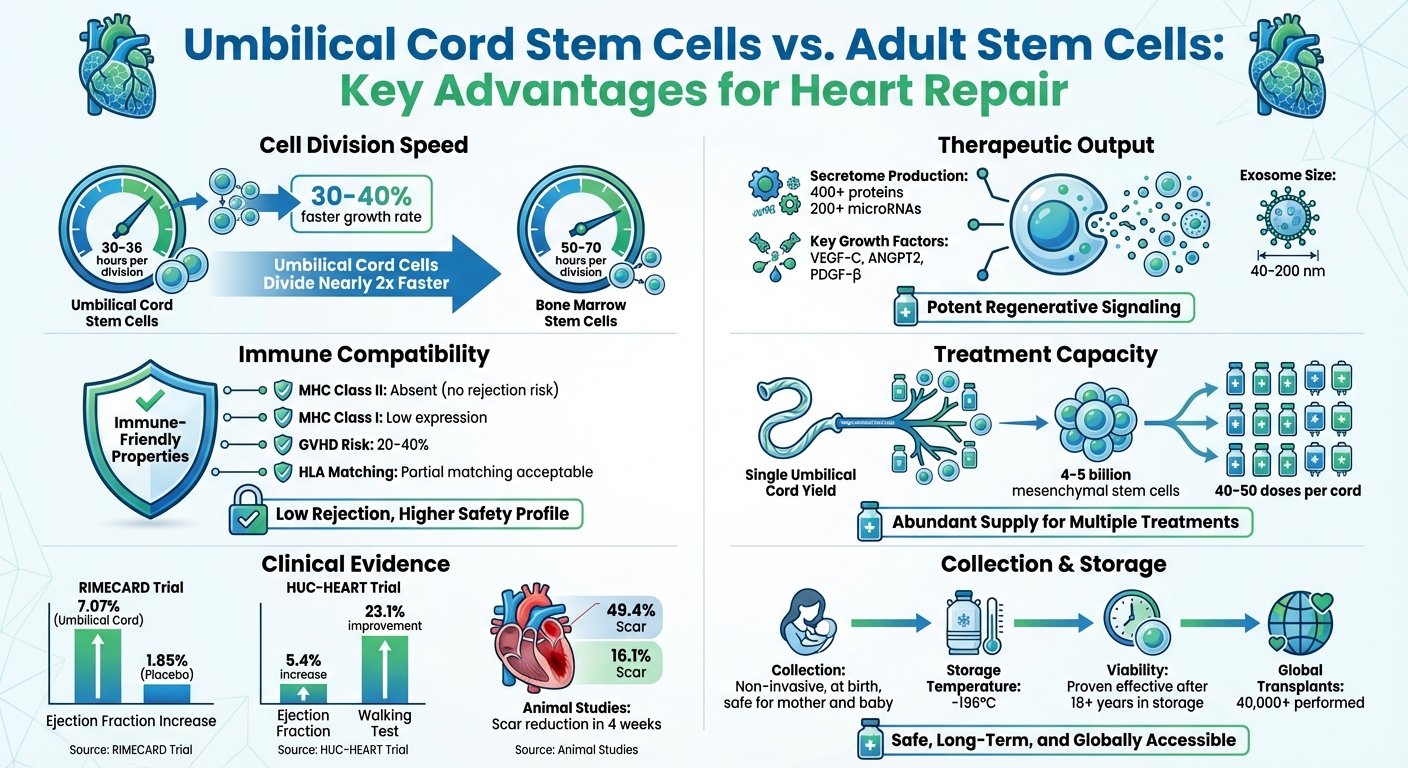
Umbilical cord mesenchymal stem cells have the potential to transform into heart muscle–like cells, but their main contribution lies in their ability to secrete healing factors rather than directly replacing damaged tissue. These cells divide at an impressive rate - approximately every 30–36 hours - nearly twice as fast as bone marrow–derived stem cells, which take 50–70 hours. Recent research highlights their exosomes, which carry circASXL1, as a means to stimulate adult cardiomyocytes to re-enter the cell cycle and generate new heart muscle. Additionally, these stem cells aid in vascular repair through precise paracrine signaling.
Paracrine Signaling and Blood Vessel Formation
When it comes to cardiovascular healing, umbilical cord stem cells secrete a powerful mix of more than 400 proteins and 200 microRNAs that encourage tissue repair. This secretome includes key growth factors like VEGF-C, ANGPT2, and PDGF-β, which are vital for angiogenesis and vascular remodeling. The therapeutic payload is delivered via exosomes - tiny particles (40–200 nm in size) - that carry microRNAs such as miR-133a-3p, which promotes blood vessel growth, and miR-29b, which reduces scar tissue. In low-oxygen environments, such as those found in damaged heart tissue, these cells increase the production of Hypoxia-Inducible Factor 1-alpha (HIF-1α), which triggers additional VEGF release to enhance survival and revascularization efforts.
Anti-Inflammatory and Immune Regulation Properties
Beyond their regenerative capabilities, umbilical cord stem cells play an important role in managing inflammation. One of their standout features is their ability to evade the immune system, as they lack MHC Class II molecules and express only low levels of MHC Class I, making them suitable for allogeneic transplants without the risk of rejection. These cells also reduce inflammation by shifting macrophages from a pro-inflammatory M1 state to a reparative M2 state. When injected directly into the heart, they activate the CCL5/CCR5 signaling pathway, which attracts CD4+FoxP3+ regulatory T cells (Tregs) to the injured area, further controlling inflammation. This process lowers levels of pro-inflammatory cytokines like TNF-α, IL-1β, and IL-6 while increasing IL-10, which helps limit scarring and improve heart function. This is especially critical for patients with cardiogenic shock after a heart attack, a condition with mortality rates exceeding 40%.
Research Evidence for Cardiovascular Applications
Animal Studies Showing Heart Repair
Research on animals highlights the potential of umbilical cord stem cells in repairing heart damage. In November 2012, scientists at the Institut Català de Ciències Cardiovasculars in Barcelona, led by S. Roura, conducted a study using human umbilical cord blood mesenchymal stem cells to treat mice with acute myocardial infarction. These cells were applied through a fibrin patch. After just four weeks, the treated mice showed a remarkable reduction in infarct scar sizes, from 49.4% to 16.1%, and an increase in blood vessel density from 4.9 mm² to 8.1 mm².
A more recent 2023 study published in Nature explored combination therapy, using immunodeficient rats. Researchers paired first-trimester umbilical cord perivascular cells with endothelial colony-forming cells. This approach boosted the ejection fraction to 139% of the control group's value within four weeks and significantly reduced cardiomyocyte apoptosis. The implanted cells not only survived for four weeks but also formed network structures that integrated into the host's circulatory system.
"The combination of FTM HUCPVCs and ECFCs synergistically reduced fibrosis and cardiomyocyte apoptosis, while promoting favorable cardiac remodeling and contractility." - Nature
These animal studies laid the foundation for subsequent human clinical trials.
Clinical Trials in Human Patients
Human trials have also shown encouraging results. The RIMECARD trial, conducted in Santiago, Chile, enrolled 30 patients with stable heart failure and reduced ejection fraction between December 2012 and June 2014. Each patient received an intravenous infusion of 1×10^6 umbilical cord mesenchymal stem cells per kilogram of body weight. After 12 months, treated patients experienced a 7.07% increase in left ventricular ejection fraction, compared to just 1.85% in the placebo group. They also reported improvements in functional status and quality of life, with no adverse events or immune rejection.
Meanwhile, the HUC-HEART trial in Ankara, Turkey, took a more direct approach. Conducted between May 2015 and December 2018, this study involved 54 male patients with chronic ischemic cardiomyopathy. Researchers injected 23×10^6 cells directly into the heart muscle during coronary artery bypass surgery. At the 12-month mark, patients showed a 5.4% rise in ejection fraction, a 19.7% increase in stroke volume, and a 7.7% reduction in necrotic myocardium. Additionally, their performance on the 6-minute walking test improved by 23.1%.
"Intravenous infusion of UC-MSC was safe in this group of patients with stable heart failure and reduced ejection fraction under optimal medical treatment. Improvements in left ventricular function, functional status, and quality of life were observed." - RIMECARD Trial Investigators
Delivery Methods for Umbilical Cord Stem Cell Therapies
Direct Injection into Heart Tissue
Direct intramyocardial injection delivers stem cells straight into damaged heart tissue, offering the highest cell retention rates among available methods. This makes it particularly effective for repairing specific areas of the heart.
There are two main techniques: transepicardial injection (performed during open-chest surgery) and catheter-based transendocardial injection. Transepicardial injection allows surgeons to place cells directly on the heart's outer surface. On the other hand, the catheter-based method uses imaging tools to guide cells through the bloodstream to the heart, making it less invasive but requiring advanced equipment and expertise.
In a June 2022 study at Tongji University, Shanghai, researchers injected 300,000 umbilical cord mesenchymal stem cells into mice hearts immediately after heart attack surgery. The cells survived for at least 28 days and significantly improved heart function, as shown by better left ventricular ejection fraction and fractional shortening.
However, the heart's constant pumping action can displace the injected cells before they fully integrate, reducing the therapy's effectiveness. Even with precise placement, this remains a challenge.
Intravenous Infusion
Intravenous infusion is the simplest and least invasive option for stem cell delivery. Doctors administer the cells through a peripheral vein, allowing them to circulate in the bloodstream and respond to natural injury signals that guide them to damaged heart tissue.
This method works best during the early stages of a heart attack when the body produces strong chemical signals to attract stem cells to the injury site. After about a week, these signals fade, making IV infusion less effective for chronic conditions.
One major drawback is poor cell retention. Many cells get trapped in the lungs, liver, or spleen before reaching the heart. In some studies, no stem cells were detected in the heart post-infusion. Still, functional improvements have been observed, such as in the RIMECARD study.
To tackle this issue, the University of Louisville launched the CATO trial in August 2024. This Phase II study is testing whether multiple IV infusions are more effective than a single dose for 60 participants with ischemic cardiomyopathy. It’s the first U.S. trial to explore repeated dosing as a way to improve retention and long-term outcomes.
New Advances in Stem Cell Delivery
Researchers are constantly refining delivery techniques to boost cell retention and improve outcomes in cardiovascular repair. Several promising methods are emerging to address existing challenges.
Injectable hydrogels and cardiac patches provide a supportive framework to hold therapeutic cells in place. These biomaterials act as scaffolds, allowing cells to stay put and release gradually, even with the heart’s continuous motion.
Magnetic targeting is another innovative approach. By labeling stem cells with iron microspheres or nanoparticles, external magnets positioned over the heart can guide and retain cells within the damaged tissue. In a preclinical rat study led by Cheng et al., magnetic targeting reduced cell migration to other organs and improved heart function over three weeks.
One of the most groundbreaking developments is Stem Cell-derived Exosome Nebulization Therapy (SCENT). This method allows patients to inhale therapeutic exosomes through an oxygen mask. In a February 2026 study with Yorkshire pigs, this non-invasive approach led to an 11.66% improvement in ejection fraction and a 5.72% increase in fractional shortening within 28 days. The exosomes bypassed filtering organs like the liver and reached the heart through the pulmonary vein.
"Inhalable cell-free therapeutics... stand to revolutionize regenerative medicine practice by providing a noninvasive and convenient delivery route." - American Heart Association (AHA)
Another exciting advancement involves engineering "stickier" cells. By coating them with E-selectin-conjugated nanocarriers or modifying their surfaces to express GPS-like receptors, scientists have improved the cells' ability to adhere to damaged blood vessel linings. This ensures better homing and retention in heart tissue, enhancing therapeutic results.
These innovations, along with the support of organizations like Americord Registry, are shaping the future of regenerative medicine, ensuring that therapies are more effective and accessible.
The Role of Stem Cell Banking in Future Cardiovascular Therapies
Understanding Newborn Stem Cell Banking
Newborn stem cell banking involves storing cord blood, cord tissue, and placental tissue. These materials are collected at birth, preserving stem cells in their most robust state. At this stage, the cells have longer telomeres and higher proliferation potential compared to adult bone marrow stem cells.
The collection process is completely safe for both the mother and baby. Once the umbilical cord is cut, medical staff collect the cord blood and tissue. These are then processed and cryopreserved at an ultra-low temperature of -196°C, ensuring their long-term viability.
Cord blood is a source of FDA-approved hematopoietic stem cells (HSCs) used to treat over 80 conditions. Meanwhile, cord tissue provides mesenchymal stem cells (MSCs), which are key for cardiovascular repair. These cells can also produce therapeutic exosomes - tiny, stable nanovesicles known for their protective effects on the heart.
One of the major advantages of banking these cells is their immediate availability. In emergencies, such as heart attacks, these stored cells can be accessed without the delays associated with harvesting and processing bone marrow cells. Remarkably, cord blood units have been successfully used for transplants even after 18 years in storage.
Another important feature of umbilical cord blood is its hypo-immunogenic nature. Due to its high concentration of naïve T lymphocytes, transplants can succeed even with partial HLA matching. This lowers the risk of Graft-Versus-Host Disease (GVHD) to just 20–40%. Globally, over 40,000 cord blood transplants have been performed, with survival rates of 60–70% for children and 55–65% for adults with malignant conditions. These characteristics make newborn stem cell banking a game-changer for cardiovascular therapies.
Americord Registry: Supporting Regenerative Medicine
Americord Registry plays a key role in advancing regenerative medicine, particularly in cardiovascular repair. The company offers specialized banking services for cord blood, cord tissue, placental tissue, and exosomes. Using its CryoMaxx™ method, Americord ensures the preservation of multipotent cells, growth factors, and cytokines - critical components for heart tissue repair.
Americord is both AABB-accredited and FDA-registered, and it employs round-the-clock, multi-layered monitoring to safeguard stored specimens. As of 2023, the registry has cryogenically stored over 22,000 units and has facilitated 12 successful releases for medical treatments since 2008.
For families dealing with congenital heart conditions diagnosed during pregnancy - such as Hypoplastic Left Heart Syndrome (HLHS) or Transposition of the Great Arteries (TGA) - Americord provides access to stem cells for clinical trials. These cells can be utilized during early procedures like the Norwood or Glenn surgeries.
Americord also offers lifetime storage plans, ensuring cells remain available for up to 78 years. This long-term access could prove invaluable for addressing cardiovascular issues that arise with age. Additionally, the company provides a $110,000 engraftment guarantee. If a collection yields less than the minimum threshold (40 mL of blood or 100 million total nucleated cells) and the family decides not to store it, no fees are charged for processing or storage.
"Banking my baby's cord blood is like an insurance. I'd feel guilty if I'd pass doing something so simple, when it comes to having an opportunity to cure a disease." - Catherine W., Mother
With over 300 clinical trials worldwide exploring the potential of newborn stem cells and perinatal tissues, and with the expectation that 1 in 3 people could benefit from regenerative medicine in their lifetime, stem cell banking is an investment in future cardiovascular health. This approach ensures rapid access to treatments and strengthens the foundation for regenerative therapies in the years to come.
Conclusion
Umbilical cord stem cells are reshaping the landscape of cardiovascular treatment. These cells, capable of repairing damaged heart tissue through paracrine signaling - secreting over 400 proteins and 200 miRNAs - bring hope to more than 6 million Americans living with heart failure. Unlike adult stem cells, they are collected non-invasively at birth, grow 30–40% faster, and come with a low risk of immune rejection.
Thanks to these advantages, off-the-shelf therapies are becoming a reality. Remarkably, a single umbilical cord can produce 4 to 5 billion mesenchymal stem cells, enough to create 40 to 50 treatment doses. As Dr. Roberto Bolli, Director of the UofL Institute of Molecular Cardiology, states:
"If these cells are shown to be effective, the implications would be enormous. They would have significant potential to improve quality of life for heart failure patients and result in a paradigm shift in the treatment of heart failure."
Banking these cells at birth is a proactive way to prepare for future cardiovascular challenges. With thousands of successful transplants already performed worldwide, early preservation ensures these cells are available for regenerative therapies when needed.
In addition to storage, advancements in delivery methods, exosome-based therapies, and combination treatments are amplifying the therapeutic potential of these cells. Given that adult cardiomyocytes regenerate at a rate of less than 1% per year, umbilical cord stem cells provide a renewable and effective option for heart tissue repair. By preserving these cells, families can secure access to cutting-edge cardiovascular treatments. Americord Registry offers innovative newborn stem cell banking services to help protect your child’s regenerative potential for the future.
FAQs
Are cord stem cell heart treatments available in the U.S. now?
Yes, cord stem cell heart treatments are available in the U.S., primarily through clinical trials that are currently in progress. One such trial, led by the University of Louisville, is investigating the use of intravenous stem cells derived from umbilical cords as a possible treatment for heart failure. These trials are a promising avenue in the quest to advance regenerative medicine for heart-related conditions.
Who can receive umbilical cord stem cells without rejection?
Umbilical cord stem cells can be transplanted into individuals with compatible immune systems without triggering rejection. This is thanks to their low immunogenicity and immunomodulatory properties, which help minimize the risk of adverse immune reactions.
Should I bank cord blood or cord tissue for heart repair?
Banking umbilical cord blood can play a significant role in heart repair. It contains stem cells that have the potential to regenerate damaged heart tissue and enhance heart function. Additionally, cord tissue provides regenerative properties that could further support cardiovascular treatments. Storing both cord blood and tissue increases the range of possible therapeutic uses, making it a worthwhile consideration.
The views, statements, and pricing expressed are deemed reliable as of the published date. Articles may not reflect current pricing, offerings, or recent innovations.