Advances in Stem Cell Disc Regeneration
Chronic back pain affects millions, with degenerative disc disease (DDD) being a major cause. Traditional treatments like spinal fusion often fail to restore natural disc function and can lead to further complications. Stem cell therapy, particularly using mesenchymal stem cells (MSCs), offers a promising alternative. Here's what you need to know:
- How It Works: MSCs repair damaged discs by producing essential proteins, reducing inflammation, and promoting tissue healing. They can be delivered via minimally invasive injections.
- Key Results: Trials show over 60% pain relief, increased disc volume, and long-term improvements in function.
- New Approaches: Exosome-based treatments, biomaterial scaffolds, and gene-edited cells enhance effectiveness in harsh disc environments.
- Safety: Studies report minimal side effects and sustained benefits over years.
- Future Potential: Stem cell banking ensures access to high-quality cells for future therapies.
Stem cell therapy is reshaping how we treat DDD, offering hope for pain relief and disc regeneration without invasive surgery.
How Stem Cell Therapy Works for Disc Regeneration
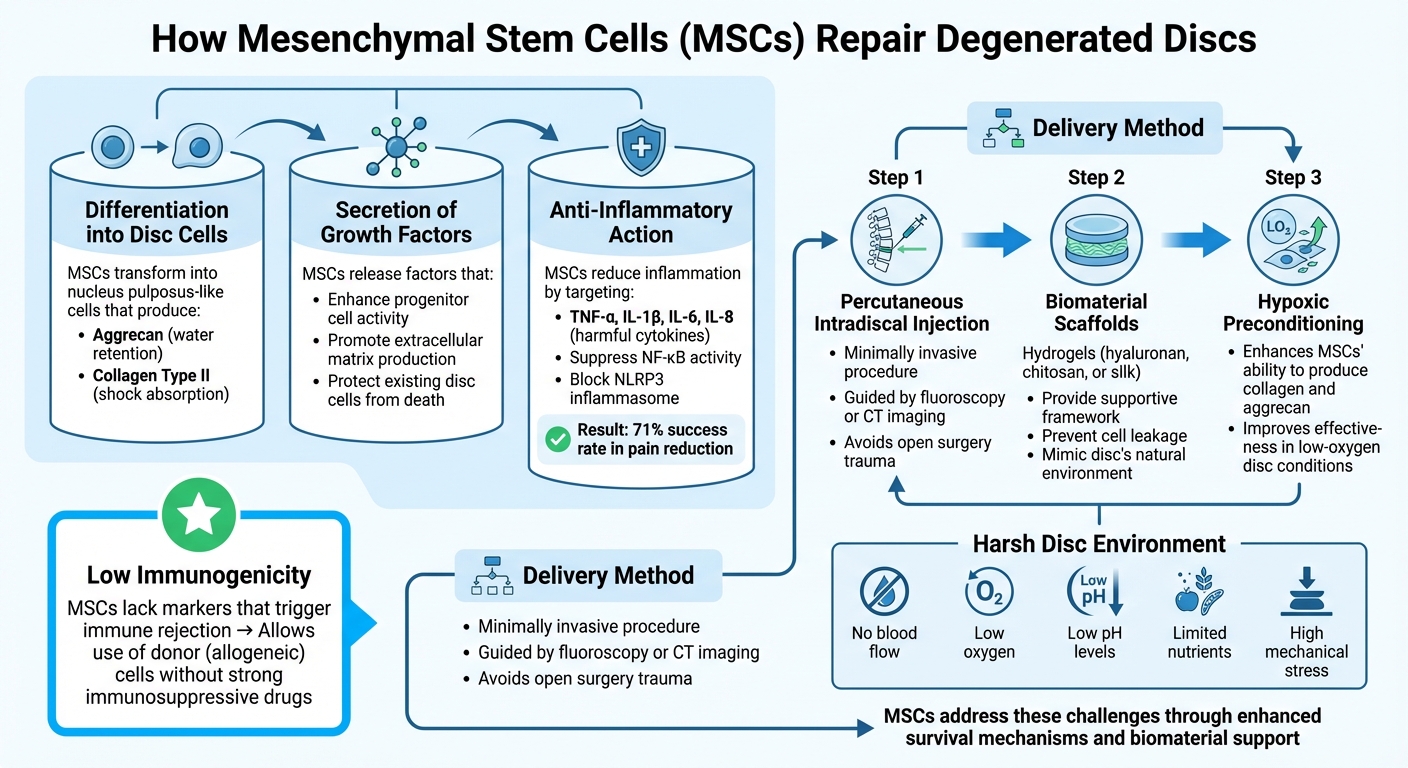
Stem cell therapy tackles the root causes of disc degeneration by leveraging mesenchymal stem cells (MSCs). These cells are known for their ability to repair damaged tissue through several mechanisms. They actively reshape the disc's environment to encourage healing. Let’s break down how MSCs contribute to disc repair and how they're delivered for maximum effectiveness.
Mesenchymal Stem Cells (MSCs) in Disc Repair
MSCs repair discs through three main processes:
- Differentiation into nucleus pulposus-like cells: These cells produce essential proteins like aggrecan and collagen type II. These proteins play a critical role in retaining water and absorbing shock, which are vital for disc function.
- Secretion of growth factors: MSCs release growth factors that enhance the activity of progenitor cells and promote extracellular matrix production. At the same time, they help protect existing disc cells from dying.
- Anti-inflammatory action: MSCs reduce inflammation by targeting harmful cytokines such as TNF-α, IL-1β, IL-6, and IL-8. These molecules are responsible for breaking down the disc matrix and triggering pain. By suppressing NF-κB activity and blocking the NLRP3 inflammasome, MSCs interrupt the inflammatory cycle that fuels degeneration. Clinical studies have shown that this immunomodulatory effect contributes to a 71% success rate in pain reduction when using autologous bone marrow MSCs.
Another advantage of MSCs is their low immunogenicity. They lack the markers that typically trigger immune rejection, allowing for the use of donor (allogeneic) MSCs, such as those from cord blood, without the need for strong immunosuppressive drugs. This makes it possible to develop ready-to-use treatments that don’t require harvesting cells from the patient.
These combined effects explain the promising pain relief and structural improvements reported in clinical trials.
Delivery Methods for Stem Cell Therapy
Stem cells are introduced into the disc through a minimally invasive procedure called percutaneous intradiscal injection, guided by fluoroscopy or CT imaging. This approach avoids the trauma of open surgery while ensuring precise placement.
However, the disc's harsh environment poses challenges for cell survival. Degenerated discs lack blood flow and are marked by low oxygen, low pH levels, limited nutrients, and high mechanical stress. To address this, researchers use biomaterial scaffolds - often hydrogels made from materials like hyaluronan, chitosan, or silk. These scaffolds provide a supportive framework, prevent cell leakage at the injection site, and mimic the disc’s natural environment to improve cell survival and function.
Additionally, hypoxic preconditioning enhances MSCs’ ability to produce collagen and aggrecan, making them more effective in the disc's low-oxygen conditions. This further boosts their regenerative potential, even in challenging environments.
Recent Clinical Trials and Outcomes
Recent clinical trials have shed light on the potential of mesenchymal stem cell (MSC) therapies for both pain relief and disc regeneration. These studies provide a closer look at how MSCs are impacting symptoms and structural healing over time.
Pain Reduction and Functional Improvements
In February 2022, a Phase I/II FDA-approved trial conducted by DiscGenics, Inc. tested rebonuputemcel (IDCT), an allogeneic disc progenitor cell therapy, on 60 patients with single-level lumbar disc degeneration. Led by Dr. Kevin T. Foley and Dr. Matthew F. Gornet across 13 clinical sites, the trial revealed promising results. Patients receiving the high-dose treatment (9,000,000 cells/mL) reported a 62.8% reduction in back pain at 52 weeks - well above the threshold for clinical significance.
"High-dose allogeneic disc progenitor cells produced statistically significant, clinically meaningful improvements in back pain and disc volume at 1 year following a single intradiscal injection and were safe and well tolerated." - Matthew F. Gornet, MD, The Orthopedic Center of St. Louis
The RESPINE trial, conducted across France, Italy, Spain, and Germany, involved 112 patients and indicated that 74% of participants experienced improvements in both pain and functional scores at 12 months after receiving 20 million allogeneic bone marrow MSCs. While the trial didn’t meet its primary endpoint, most patients reported meaningful relief.
Long-term outcomes also stand out. Between 2015 and 2016, Regenexx Cayman treated 13 patients with lab-expanded bone marrow MSCs combined with platelet lysate or platelet-rich plasma. By the 6-year mark, these patients showed a 24.14-point improvement in Functional Rating Index scores. Even more striking, seven patients followed for 10 years reported an average 78.1% improvement in their Single Assessment Numeric Evaluation (SANE) scores - marking the longest follow-up data recorded for this therapy.
These clinical results are complemented by evidence of structural disc regeneration.
Structural Disc Repair Evidence
Pain relief is one thing, but can MSCs actually repair damaged discs? The IDCT trial provided clear evidence. Patients in the high-dose group showed a mean disc volume increase of 249.0 mm³ at 52 weeks, which further grew to 402.1 mm³ by 104 weeks. This wasn’t just inflammation subsiding - the discs were regenerating.
A Phase IIB trial involving 52 patients treated with autologous bone marrow MSCs also demonstrated structural benefits, with significant increases in the Disc Height Index compared to baseline. In another study, 85% of patients treated with bone marrow-derived MSCs experienced a 23% average reduction in disc bulge size.
However, structural improvements don’t always translate to immediate clinical benefits. Some trials have shown that while imaging reveals better disc height and composition, patients may not experience noticeable improvements compared to those receiving placebo treatments within the first six months. This suggests that structural healing may precede functional recovery, requiring time for the benefits to fully manifest.
Safety and Long-Term Results
Safety and long-term outcomes are critical in evaluating these therapies. In the 60-patient IDCT trial, serious adverse events occurred in 6.7% of participants, but all were in the control groups receiving vehicle or placebo. Importantly, no treatment-related serious events were reported, and the therapy was well-tolerated, with no signs of immune rejection even when donor cells were used.
"This study presents the longest known follow-up data for intradiscal MSC therapy for discogenic LBP, demonstrating sustained improvements in pain and function." - Nicholas Hooper, Emory University School of Medicine
An analysis of a clinical registry involving 127 patients showed a low suspected discitis rate of 3.94%, which was linked to repetitive intradiscal injections. This highlights the need for strict sterile techniques and careful patient selection. Additionally, 10-year follow-up data from Emory University reported no long-term adverse events, suggesting that once the cells integrate successfully, they pose minimal ongoing risks.
The durability of benefits appears to depend on dosage. High-dose treatments showed sustained improvements over two years, while lower doses were less consistent. This underscores the importance of delivering an adequate number of viable cells to overcome the challenging environment within degenerative discs. These findings highlight the potential of MSC therapies to address both the symptoms and structural challenges of degenerative disc disease effectively.
New Developments in Stem Cell Disc Regeneration
The field of stem cell therapies for disc regeneration is advancing rapidly, moving beyond traditional methods like mesenchymal stem cell (MSC) injections. Researchers are now exploring cutting-edge approaches such as exosome-based therapies, treatments combining stem cells with biomaterials, and gene-edited allogeneic cells to tackle disc degeneration more effectively.
Exosome-Based Therapies
Exosomes, tiny vesicles ranging from 30 to 150 nanometers, are emerging as a promising alternative to live stem cell therapies. These cell-free agents thrive in low-oxygen, acidic environments and deliver proteins, lipids, and genetic material that can stimulate healing, all while avoiding risks like immune rejection or abnormal cell growth.
In 2025, researchers at the First Affiliated Hospital of Zhengzhou University developed a hydrogel system combining exosomes derived from nucleus pulposus cells with senolytic drugs like Quercetin and Dasatinib. Tested on rats and goats, this approach cleared damaged cells and restored mitochondrial function, leading to significant extracellular matrix repair. That same year, a phase 1 clinical trial (NCT04849429) in India explored the use of platelet-rich plasma enriched with exosomes to treat chronic low back pain by injecting it directly into degenerated discs.
"Stem cell‐derived exosomes (SC‐Exos), specifically, present a significant opportunity for the safe and effective treatment of IVDD because of their ability to maintain the therapeutic benefit of their origin cells without the risks associated with stem cell‐based therapies." - Zoe Krut et al., Cedars-Sinai Medical Center
Recent innovations include engineering exosomes with targeted therapeutic cargo, such as circular RNAs, to prevent cell death and encourage the production of extracellular matrix proteins. This strategy sidesteps the survival issues live stem cells face in the harsh, oxygen-poor disc environment.
To enhance these therapies, researchers are combining exosomes and stem cells with biomaterials for added support and functionality.
Combination Therapies
Pairing stem cells with biomaterials is proving to be more effective than standalone cell injections. The disc environment, characterized by low oxygen, inflammation, and acidity, often limits the survival of transplanted cells. Biomaterial scaffolds, such as hydrogels, mimic the disc’s natural structure, providing both physical support and a conducive environment for cell survival and tissue repair.
In late 2024, a team advanced this approach by using a conductive scaffold made from nanochitosan, polypyrrole, and alginate, loaded with Noggin - a growth factor. When tested on rats with spinal injuries, this method improved motor function over four weeks by releasing Noggin to inhibit BMP-4 signaling and encourage neural regeneration.
"Hydrogels mimic the disc's microenvironment, providing optimal growth for MSCs and EVs in IDD therapy." - Biomedicine & Pharmacotherapy Review
Researchers are also creating "smart" biomaterials that adapt to mechanical stress, prevent tissue swelling, and enhance the regenerative capabilities of transplanted cells.
Beyond biomaterials, gene editing offers another exciting avenue for improving stem cell therapies.
Gene Editing and Allogeneic Stem Cells
Gene editing tools like CRISPR-Cas9 are transforming allogeneic (donor-derived) stem cell therapies. By modifying stem cells before transplantation, scientists can improve their survival in low-oxygen conditions and reduce the risk of immune rejection.
Gene-edited cells can boost the production of essential extracellular matrix proteins, such as type II collagen and proteoglycans, while silencing genes that contribute to disc degeneration. This approach addresses the underlying causes of the condition, offering more than just symptom management.
"Technologies such as gene editing and immune modulation can mitigate this risk [of immune rejection]." - World Journal of Stem Cells
Allogeneic cells also offer practical advantages. Unlike autologous cells, which require personalized harvesting and preparation, donor-derived cells can be pre-prepared, gene-edited for optimal performance, and stored for immediate use. Umbilical cord-derived mesenchymal stem cells are particularly appealing, as they are less likely to trigger immune responses and can be collected noninvasively at birth - a service offered by providers like Americord Registry through advanced tissue cryostorage services for cord blood and tissue.
The Role of Stem Cell Banking in Regenerative Medicine
Preserving Stem Cells for Future Use
Stem cell banking has become a cornerstone for advancing therapies aimed at disc regeneration, offering a reliable source of high-quality stem cells.
Cord tissue is particularly important because it contains mesenchymal stem cells (MSCs) that can transform into the specialized cells required for disc repair. These tissues, when banked, can be expanded in labs to produce millions of cells - essential for clinical applications. This is a game-changer, especially considering that over 90% of individuals above 50 experience some degree of disc degeneration, with up to 50% of disc cells becoming necrotic in advanced cases.
Timing plays a critical role here. Newborn stem cells sourced from cord blood, cord tissue, and placental tissue are far more potent than adult-derived cells like those from bone marrow. They not only multiply faster but also have better differentiation potential. Additionally, their low immunogenicity makes them suitable for both personal (autologous) and donor-based (allogeneic) treatments. These preserved cells are key to the success of emerging therapies, forming a solid foundation for future regenerative treatments.
Stem cell banking also supports cutting-edge innovations like exosome-based therapies and gene editing. Exosome treatments, for example, rely on preserved stem cells to create therapeutic vesicles, while gene editing requires viable cells as templates. With an estimated 1 in 3 Americans (roughly 128 million people) likely to benefit from regenerative medicine for conditions like cardiovascular, neurological, or orthopedic issues in their lifetime, preserving these cells is akin to securing a biological safety net. This not only ensures a steady supply of potent cells but also accelerates the development of groundbreaking therapies, such as those pioneered by Americord Registry.
Americord Registry's Approach to Stem Cell Preservation
Recognizing the critical need for high-quality stem cells in regenerative medicine, Americord Registry employs advanced techniques to ensure these cells are readily available for future treatments.
Americord Registry offers comprehensive newborn stem cell preservation services, aligning with the latest regenerative medicine advancements. Their CryoMaxx™ Processing technology and 5-compartment storage vials ensure long-term cell viability, preserving cord blood, cord tissue, placental tissue, and exosomes. Backed by AABB accreditation and transparent pricing, their approach supports the development of next-generation therapies, including combination treatments, exosome-based applications, and gene-edited cell technologies - key innovations in spinal disc repair and beyond.
Conclusion
Stem cell therapy has made a remarkable leap from experimental trials to clinically validated treatments, offering a groundbreaking approach to managing chronic back pain. For example, the DiscGenics trial demonstrated that a single injection reduced pain by 62.8% and increased disc volume by 402.1 mm³ over two years - clear evidence of structural regeneration. These advancements bring new hope to those affected by the widespread challenges of chronic back pain.
The field is also moving toward more accessible solutions, such as allogeneic "off-the-shelf" therapies, exosome-based treatments, and gene-editing technologies. Additionally, combining stem cells with biomaterial scaffolds and growth factors is showing encouraging results, potentially leading to therapies that are easier to standardize and distribute.
"The key factor making stem cell treatment particularly attractive involves their potential to repopulate the disc with viable cells and their ability to restore damaged tissue and enhance tissue regeneration by modulating the inflammatory response." - Khadija H. Soufi, Department of Neurological Surgery, UC Davis
That said, there’s still work to be done. Researchers are focusing on refining delivery methods and improving the survival of cells in the challenging environment of the spine. Access to high-quality stem cells remains critical for the success of these therapies, which is why preservation services are becoming increasingly important.
Banking young, potent stem cells - such as those preserved through Americord Registry - can be a forward-thinking way to ensure access to future regenerative treatments. With up to 50% of disc cells becoming necrotic in advanced degeneration, having a reserve of preserved cells could be key to maintaining spinal health as these therapies continue to evolve.
FAQs
Who is a good candidate for disc stem cell therapy?
A suitable candidate for disc stem cell therapy is typically someone dealing with lumbar disc degeneration. This condition often brings pain, limited mobility, and a noticeable decline in quality of life. The therapy offers a regenerative approach and might appeal to those looking for treatments grounded in clinical research.
How long do results from a single intradiscal injection last?
The impact of a single intradiscal injection can persist for several months and, in some cases, over a year. Some studies even indicate that the benefits might last beyond the one-year mark. However, results can differ from person to person, influenced by factors such as the specific procedure used and the patient’s overall condition.
What are the biggest risks, like infection or immune rejection?
The primary risks associated with stem cell therapy for disc regeneration are infection and immune rejection. However, recent studies indicate that using allogeneic disc progenitor cells can significantly increase disc volume and lead to better clinical results while keeping adverse effects to a minimum. These developments point toward a future with safer and more efficient treatment options.
The views, statements, and pricing expressed are deemed reliable as of the published date. Articles may not reflect current pricing, offerings, or recent innovations.