Clinical Trials for Stem Cell Disc Repair
Clinical Trials for Stem Cell Disc Repair
Stem cell therapy is emerging as a potential solution for degenerative disc disease, a leading cause of chronic back pain that affects 30 million Americans and costs the U.S. economy $625 billion annually. Unlike traditional treatments that focus on symptom management, stem cell therapy aims to repair damaged spinal discs.
Key Takeaways:
- How it works: Stem cells regenerate disc tissue by producing essential proteins, reducing inflammation, and improving hydration.
- Clinical results: A trial with 60 patients showed a 62.8% reduction in pain and increased disc volume by 249 mm³ in one year.
- Challenges: Harsh disc conditions (low oxygen, acidity, and mechanical stress) limit cell survival and effectiveness.
- Future developments: Research includes hydrogel scaffolds, gene-editing, and exosome-based therapies to improve outcomes.
- Safety: Trials report low rates of adverse events, with allogeneic (donor-derived) stem cells offering convenient, off-the-shelf options.
While promising, stem cell therapy still faces hurdles, such as inconsistent pain relief and limited long-term success. Ongoing research focuses on refining techniques to improve patient outcomes and expand treatment access.
How Stem Cell Therapy Works for Disc Repair
How Stem Cells Regenerate Tissue
Mesenchymal stem cells (MSCs) play a key role in repairing damaged discs through three main actions: they transform into nucleus pulposus-like cells that produce type II collagen and proteoglycans, essential for disc structure; they release growth factors that encourage native cells to ramp up matrix production and enter repair mode; and they regulate immune responses, reducing inflammatory cytokines like IL-1β, IL-6, and TNF-α, which contribute to tissue breakdown and pain.
"The therapeutic effects of implanted stem cells are more influenced by the bioactive factors they secrete than by their direct cellular contributions." - Matic Munda and Tomaz Velnar, Department of Neurosurgery, University Medical Centre Ljubljana
Clinical trials have shown measurable improvements in disc volume and hydration, underscoring the potential of stem cell therapy. However, the harsh disc environment remains a major hurdle for these therapies to succeed fully.
Nucleus pulposus cells, though they make up only 1% of the total disc tissue, are responsible for maintaining the extracellular matrix. By replacing or stimulating these cells, stem cells help restore the disc's ability to retain water, maintain height, and absorb mechanical stress. Yet, the challenging conditions within the disc limit even these promising regenerative efforts.
Why Disc Repair Is Difficult
The intervertebral disc presents a uniquely harsh environment that makes regeneration challenging. As the largest avascular structure in the human body, it lacks a direct blood supply, creating an environment with minimal oxygen, high acidity, limited nutrients, and constant mechanical stress.
| Biological Obstacle | Impact on Regeneration |
|---|---|
| Avascularity | Limits nutrient delivery and waste removal |
| Hypoxia/Low pH | Creates an acidic environment that leads to cell death |
| Mechanical Loading | Continuous pressure affects cell survival |
| Endplate Calcification | Reduces nutrient diffusion from vertebral bodies |
Research highlights the difficulty of cell survival in such conditions. For example, a canine study found that implanted MSCs survived for just three weeks in the disc environment. To tackle this, scientists are investigating preconditioning techniques, such as exposing stem cells to hypoxic conditions before transplantation to improve their resilience. Other strategies include using hydrogel scaffolds or supportive matrices to shield the cells and enhance their retention in the disc space.
Disc degeneration further complicates repair efforts. As the disc deteriorates, it shifts from producing type II collagen (associated with healthy tissue) to type I collagen (linked to scar tissue). Proteoglycans, which help retain water, are lost, and nociceptive nerves and blood vessels grow into the disc, causing chronic pain. Stem cell therapy aims to counter these changes by restoring the disc's biochemical balance and internal pressure. These challenges help explain the mixed outcomes observed in clinical trials.
Clinical Trial Results and Patient Outcomes
Stem Cell Therapy Clinical Trial Results for Disc Repair
Major Clinical Trials
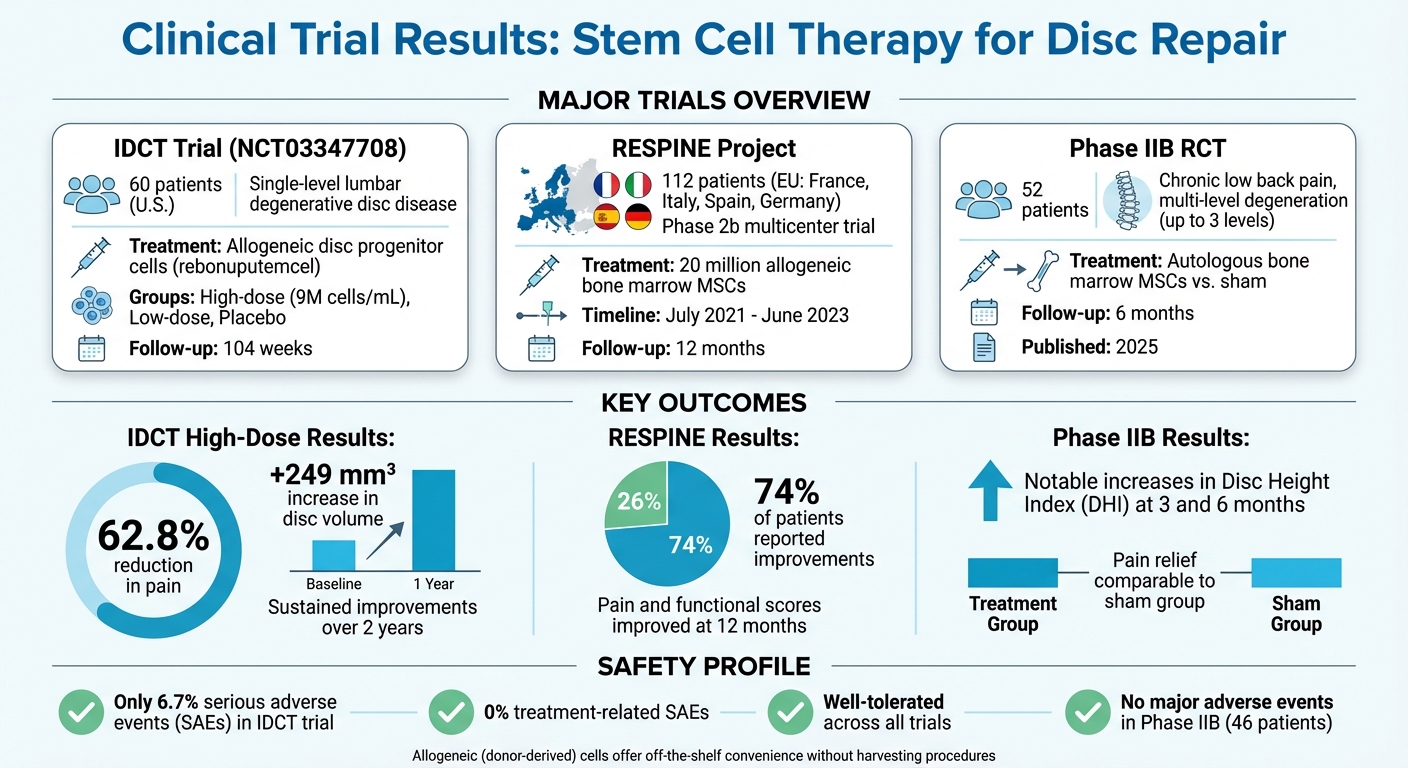
From 2018 to 2024, several large-scale studies have explored stem cell therapy for disc repair, each offering unique insights. One notable trial, the IDCT trial (NCT03347708), involved 60 U.S. patients with single-level lumbar degenerative disc disease. Participants were treated with allogeneic disc progenitor cells, known as rebonuputemcel, through high-dose (9,000,000 cells/mL), low-dose, or placebo injections. This Phase I/II study, led by top researchers, monitored outcomes for up to 104 weeks.
The RESPINE project, a Phase 2b multicenter trial backed by the European Union, treated 112 patients in France, Italy, Spain, and Germany between July 2021 and June 2023. Each patient received an injection of 20 million allogeneic bone marrow-derived mesenchymal stem cells. Although the trial didn’t meet its primary endpoint, it provided critical data on safety and efficacy at the 12-month mark.
Another Phase IIB randomized controlled trial focused on autologous bone marrow-derived MSCs in 52 patients with chronic low back pain and moderate-to-advanced multi-level disc degeneration (up to three levels). Published in 2025, the study compared stem cell injections with a sham procedure, tracking both structural and clinical improvements over six months.
Pain Relief and Function Improvements
The IDCT trial's high-dose group showed statistically significant pain relief and structural regeneration sustained over two years, surpassing the threshold for meaningful back pain improvement. In comparison, the low-dose and placebo groups experienced less notable changes.
"High-dose allogeneic disc progenitor cells produced statistically significant, clinically meaningful improvements in back pain and disc volume at 1 year following a single intradiscal injection and were safe and well tolerated." - PubMed Abstract, Study NCT03347708
In the RESPINE trial, 74% of patients reported improvements in pain and functional scores at the 12-month follow-up, even though the trial didn’t achieve its primary endpoint. This highlights the variability in patient responses, influenced by factors like disease progression and selection criteria.
The Phase IIB trial of autologous bone marrow MSCs revealed encouraging structural changes. Patients receiving stem cell therapy showed notable increases in Disc Height Index (DHI) at three and six months compared to the sham group, signaling potential regeneration. However, pain relief, measured by VAS pain and Oswestry Disability Index (ODI) scores, was comparable between the treatment and sham groups during the six-month follow-up. These results underscore the challenge of linking structural regeneration to consistent pain relief.
Safety Profile and Patient Selection
Safety remains a key focus in stem cell therapy trials. Across multiple studies, treatments have shown a strong safety profile. For example, the IDCT trial reported that only 6.7% of participants experienced serious adverse events (SAEs), none of which were related to the treatment. Similarly, the Phase IIB trial confirmed that autologous bone marrow-derived MSC injections were well-tolerated, with no major adverse events reported among 46 patients.
Patient selection also plays a crucial role. Most trials include individuals with symptomatic single- or limited multi-level lumbar disc degeneration who haven’t responded to at least six months of conservative care. Patients with severe spinal stenosis, spondylolisthesis, or other structural instabilities requiring surgery are typically excluded. For instance, the IDCT trial focused on patients averaging 37.9 years of age with single-level disease, while the Phase IIB study included those with up to three levels of degeneration to assess outcomes in more advanced cases.
The growing preference for allogeneic (donor-derived) cells over autologous (patient-derived) cells is reshaping the field. Treatments like IDCT offer "off-the-shelf" solutions, eliminating the need for bone marrow or adipose tissue harvesting, which can involve additional surgical risks. This approach also ensures standardized processing and immediate availability, making therapies more accessible and consistent across different patients.
Future Developments in Stem Cell Therapy
New Delivery Methods and Combined Treatments
Researchers are working on improving how stem cells are delivered to ensure they thrive in the harsh environment of damaged discs. One promising approach involves injectable hydrogels and biological scaffolds. These materials - made from substances like alginate, hyaluronic acid, and atelocollagen - help protect stem cells by creating a supportive structure. Once injected, they solidify, keeping the cells in place and shielding them from the hostile conditions inside the disc.
Some teams are taking this a step further by developing complete intervertebral disc replacements. These replacements are designed to mimic both the mechanical and biological functions of a healthy disc, combining components like the annulus fibrosus and nucleus pulposus for optimal integration.
Another exciting avenue involves combining stem cells with substances like growth factors (GDF-5, GDF-6, TGF-β), platelet-rich plasma (PRP), or hyaluronic acid. These combinations can improve stem cell survival and encourage the production of essential components like type II collagen and aggrecan, which are crucial for healthy disc tissue. Researchers are also exploring genetic engineering to enhance stem cells. By modifying them to produce specific growth factors or withstand challenging conditions, scientists have seen promising results in animal studies, including reduced cell death and improved tissue regeneration.
Exosomes and Gene-Editing Approaches
Beyond traditional stem cell therapies, exosome-based treatments are emerging as an innovative, cell-free solution. Exosomes are tiny particles secreted by stem cells, packed with bioactive molecules that encourage native cells to repair tissue. Unlike whole-cell transplants, exosomes carry fewer risks, such as immune rejection or uncontrolled cell growth, making them a safer alternative.
Meanwhile, gene-editing tools like CRISPR are revolutionizing how stem cells are used. Scientists can tweak these cells to overexpress helpful genes, like Sox-9, which supports tissue growth, or suppress harmful inflammatory signals that speed up disc degeneration. This precision allows stem cells to not only survive longer but also actively contribute to tissue repair by delivering the right therapeutic signals at the right time.
Stem Cell Banking and Regenerative Medicine
With the progress seen in clinical trials, storing high-quality regenerative cells at birth has become a practical way to prepare for future therapies.
Storing Stem Cells for Future Treatments
Stem cell banking at birth provides a reliable supply of mesenchymal stem cells (MSCs), which are central to the success of treatments like spinal disc repair. These cells, collected from umbilical cord blood, cord tissue, and placental tissue, are more effective and multiply faster than those obtained later in life from sources like bone marrow or fat tissue. By preserving these cells at their peak potency, families gain what researchers often refer to as bioinsurance - a safeguard for future regenerative needs.
Using umbilical cord-derived MSCs eliminates the need for invasive procedures later in life. These cells have been utilized in over 50,000 procedures over the past 15 years without rejection. For example, they can be injected directly into spinal discs to reduce inflammation and even reverse degeneration. Banking at birth ensures cells are stored in their optimal state, ready to support promising therapies as they continue to emerge from clinical trials.
Americord Registry's Advanced Processing
 Americord Registry employs CryoMaxx™ Processing, a state-of-the-art freezing method designed to prevent ice crystal damage and maintain the viability of stored cells. This process ensures the cells remain functional for clinical use, which is essential for successful regenerative therapies.
Americord Registry employs CryoMaxx™ Processing, a state-of-the-art freezing method designed to prevent ice crystal damage and maintain the viability of stored cells. This process ensures the cells remain functional for clinical use, which is essential for successful regenerative therapies.
Americord offers a variety of storage options, including cord blood, cord tissue, placental tissue, and even exosome preservation. Their 5-compartment storage vials allow for multiple uses from a single collection, providing flexibility for future treatments. All processing adheres to FDA standards and is accredited by the AABB, ensuring the highest level of safety and quality.
Conclusion
Stem cell therapy is showing promise as a game-changer for treating degenerative disc disease. Clinical trials reveal that high-dose stem cell treatments can significantly reduce pain and even regenerate damaged discs. However, these breakthroughs come with challenges.
The disc environment itself poses a major obstacle. Low oxygen levels, high acidity, and limited nutrients make it tough for transplanted cells to survive long-term. Some Phase IIB studies indicate that while structural improvements are evident, they don’t always lead to better pain relief compared to placebo treatments, especially in short-term follow-ups. Adding to the complexity, many studies use small sample sizes and inconsistent cell mixtures, making it hard to predict which therapies will deliver reliable results.
Timing also plays a crucial role. Early intervention is key because severe degeneration limits the potential for regeneration, making it harder to achieve optimal outcomes.
While clinical trials underscore the potential of stem cell therapy, refining these treatments for long-term success remains critical. Allogeneic, or "off-the-shelf", stem cell products offer a streamlined approach, removing the need for invasive cell harvesting and ensuring consistent quality. Regulatory advancements like FDA Fast Track designations are paving the way for progress, but innovation in delivery systems and biomaterials will be vital moving forward. Considering chronic low back pain costs the U.S. healthcare system over $100 billion annually, the push for better solutions couldn’t be more urgent.
FAQs
How do stem cells help repair spinal discs affected by degenerative disc disease?
Stem cells have an impressive ability to repair spinal discs by regenerating damaged tissue. They can develop into specific cell types, such as nucleus pulposus and annulus fibrosus cells, which are vital for rebuilding and maintaining the structure of intervertebral discs. Beyond this, stem cells also release bioactive compounds that help reduce inflammation, encourage the growth of native disc cells, and boost the production of extracellular matrix - an essential component for restoring the disc's integrity.
What challenges do stem cell therapies face in treating spinal disc degeneration?
Stem cell therapies for spinal disc degeneration hold promise, but they come with several hurdles that need careful attention. Among the top concerns is ensuring safety during implantation. Clinical trials are focused on minimizing risks while improving precision through advanced methods like image-guided injections. However, patient responses to these treatments can vary widely, and more research is needed to confirm whether the benefits last over the long term.
What recent advancements are improving stem cell therapies for spinal disc repair?
Recent progress in stem cell therapies for spinal disc repair is zeroing in on making treatments safer, more effective, and geared toward genuine regeneration. Clinical trials have demonstrated that using allogeneic disc progenitor cells can safely increase disc volume while also easing pain and reducing disability for individuals with lumbar disc degeneration. Some findings even hint at the possibility of slowing - or potentially reversing - the progression of this condition.
The views, statements, and pricing expressed are deemed reliable as of the published date. Articles may not reflect current pricing, offerings, or recent innovations.