Future of MS Treatment with Stem Cells
Stem cell therapy is emerging as a promising option for treating multiple sclerosis (MS), a chronic autoimmune disease affecting the central nervous system. Unlike current treatments that mainly manage symptoms, stem cell approaches aim to repair damage and reset the immune system. Here's a quick summary:
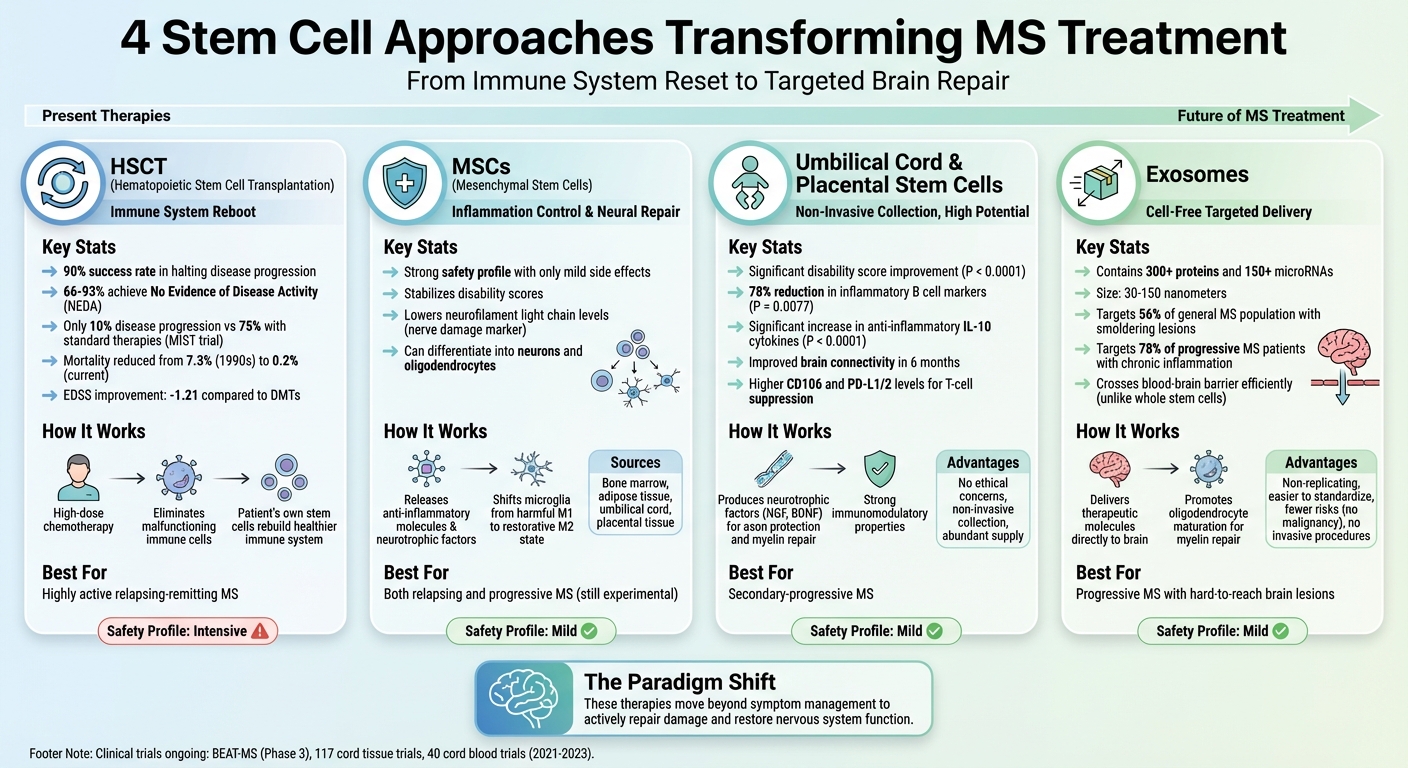
- Hematopoietic Stem Cell Transplantation (HSCT): Reboots the immune system, showing up to 90% success in halting disease progression in clinical trials.
- Mesenchymal Stem Cells (MSCs): Focuses on reducing inflammation and promoting neural repair, with strong safety profiles but still experimental.
- Umbilical Cord & Placental Stem Cells: Non-invasive collection methods with high immunomodulatory potential, showing early success in reducing disability scores and inflammation.
- Exosomes: A cell-free option delivering therapeutic molecules directly to the brain, offering better targeting of MS lesions.
Clinical trials reveal improved outcomes for MS patients, including reduced disability scores, fewer relapses, and slowed disease progression. Stem cell banking, especially at birth, is becoming a practical step for families to prepare for future treatments. These therapies represent a shift toward repairing and restoring the nervous system rather than just managing symptoms.
Stem Cell Therapy Types for Multiple Sclerosis Treatment Comparison
Current Stem Cell Approaches in MS Treatment
Two primary stem cell strategies are currently being explored for treating multiple sclerosis (MS): one aims to reset the immune system, while the other focuses on reducing inflammation and encouraging neural repair. These methods tackle the disease from different angles, offering hope for improved outcomes. Let’s dive into the details of each approach.
Hematopoietic Stem Cell Transplantation (HSCT)
HSCT is designed to "reboot" the immune system. The process starts with high-dose chemotherapy to eliminate the patient’s malfunctioning immune cells. Afterward, the patient’s own stem cells - collected in advance - are reinfused to help rebuild a healthier, more tolerant immune system.
"The rationale for the use of AHSCT in MS... is that this treatment eradicates disease-associated adaptive and innate immune components, followed by restoration of immune tolerance through robust immune reconstitution." - Consensus Statement from ECTRIMS and EBMT
The MIST trial, led by Dr. Richard Burt at Northwestern University, treated 110 patients with highly active relapsing-remitting MS. The results were striking: only 10% of HSCT patients experienced disease progression over five years, compared to 75% of those on standard disease-modifying therapies. Additionally, between 66% and 93% of HSCT patients achieved No Evidence of Disease Activity (NEDA), meaning no relapses, no new brain lesions on MRI, and no worsening of disability.
Safety has improved significantly over the years. In the late 1990s, transplant-related mortality was 7.3%, but advancements in patient selection and treatment protocols have reduced this figure to approximately 0.2%. Medium-intensity or non-myeloablative conditioning regimens, using drugs like cyclophosphamide and ATG, have further enhanced safety. However, HSCT remains an intensive procedure that requires hospitalization and temporarily weakens the immune system, increasing the risk of infections.
Mesenchymal Stem Cells (MSCs) and Their Potential
MSCs offer a less aggressive alternative, focusing on modulating the immune system and protecting neural tissue. These cells release anti-inflammatory molecules and neurotrophic factors that may help repair damage.
"MSC therapy may be a safe and tolerable treatment, but its clinical value is still under evaluation." - Fernando X. Cuascut and George J. Hutton
MSCs can be derived from various sources, including bone marrow, adipose tissue, umbilical cord, or placental tissue. They have the ability to differentiate into neurons and oligodendrocytes and can shift inflammatory microglia from a harmful M1 state to a more restorative M2 state. Early clinical trials have shown that MSC therapy can stabilize disability scores and lower neurofilament light chain levels, a marker of ongoing nerve damage.
The safety profile for MSC therapy has been promising, with most patients experiencing only mild side effects like local reactions or temporary fever. Researchers are also exploring intrathecal administration, which involves delivering the cells directly into the cerebrospinal fluid for potentially better targeting of the central nervous system. While MSCs hold potential for both relapsing and progressive forms of MS, this approach remains experimental, with questions about optimal dosing and delivery methods still unanswered.
Stem Cell Sources for MS Treatment
Umbilical Cord and Placental Tissue-Derived Stem Cells
Stem cells derived from umbilical cord and placental tissue - referred to as perinatal stem cells - are gaining attention as a potential therapy for MS. Unlike bone marrow extraction, which involves invasive procedures, these cells are collected from postnatal tissues that are typically discarded. This process avoids the ethical concerns often associated with embryonic stem cells.
"PLMSCs can be easily and non-invasively isolated from discarded pregnancy tissues, are abundant, have no ethical issues, and can differentiate into various neural cell lineages." - Scientific Reports
A key advantage of newborn stem cells is their strong immunomodulatory properties. They produce higher levels of CD106 and PD-L1/2 (programmed death ligand proteins), which help suppress harmful T-cell activity. A phase 1 trial conducted at Tehran University of Medical Sciences involved five secondary-progressive MS patients who received a single intravenous dose of 3 × 10⁶ PLMSCs per kilogram of body weight. Over six months, patients showed significant improvements in disability scores (P < 0.0001) and brain connectivity. Immunological analysis highlighted a marked reduction in inflammatory B cell markers (P = 0.0077) and a significant increase in anti-inflammatory IL-10 cytokines (P < 0.0001).
These stem cells also release neurotrophic factors like nerve growth factor (NGF) and brain-derived neurotrophic factor (BDNF), which are crucial for protecting axons and potentially repairing damaged myelin sheaths. The treatment's safety profile has been encouraging, with only mild side effects, such as headaches, reported in a small number of patients.
Advancements in stem cell therapy are also paving the way for complementary approaches, such as exosome-based treatments.
Exosomes and Their Role in MS Treatment
Exosomes provide a cell-free option for delivering therapeutic molecules. These tiny vesicles, ranging from 30 to 150 nanometers, are naturally released by stem cells and carry a rich payload of over 300 proteins and 150 microRNAs that can influence the behavior of recipient cells.
One of their standout features is their ability to cross the blood-brain barrier far more efficiently than whole stem cells, which often get trapped in the lungs after intravenous injection. This makes exosomes particularly effective at targeting "smoldering" lesions - chronic inflammatory areas found in 56% of the general MS population and up to 78% of progressive MS patients - that are otherwise difficult to treat.
"Exosomes from MSCs may represent a potential cell-free treatment modality to prevent disease progression and promote remyelination in MS." - Torbjørn Kråkenes, Researcher in Neurobiology, Haukeland University Hospital
Exosomes play a dual role in MS treatment. They help shift microglia (a type of immune cell in the brain) from a pro-inflammatory state to an anti-inflammatory one, while also promoting the maturation of oligodendrocyte progenitor cells, which are essential for repairing myelin. Certain microRNAs within exosomes, like miR-23b-3p, have been shown to reduce inflammatory cell death in microglia, providing further protection for neural tissue. Since exosomes are non-replicating and easier to standardize than living cells, they come with fewer risks, such as malignancy or complications from invasive procedures.
Key Findings from Clinical Trials
Clinical Trial Results Comparison
Recent clinical trials have shed light on the safety, delivery methods, and effectiveness of various therapies for multiple sclerosis (MS). The table below highlights key outcomes and safety findings from these studies:
| Trial / Therapy Type | Delivery Route | Target MS Type | Key Clinical Outcome | Primary Safety Finding |
|---|---|---|---|---|
| MSC-NP (Phase II) | Intrathecal | Progressive (SPMS/PPMS) | Improved walking (EDSS 6.0-6.5) & 76% bladder function improvement | Transient headaches (34%) |
| AHSCT (Meta-analysis) | Intravenous | Relapsing (RRMS) | EDSS reduction of -0.57 & lesion volume decrease of 7.05 mm³ | Hematological disorders (RR = 2.33) |
| hfNPCs (STEMS) | Intrathecal | Progressive | Reduced grey matter atrophy at higher doses | No severe adverse reactions over 2 years |
The Tisch MS Research Center of New York conducted a Phase II trial (NCT03355365) from August 2018 to June 2020, enrolling 54 patients with progressive MS. Led by Dr. Saud Sadiq, the study involved six intrathecal injections of autologous MSC-NPs. While the primary endpoint wasn't met for all participants, those with EDSS scores between 6.0 and 6.5 showed notable improvements in the 6-minute walk test (p=0.036). Among this high-disability subgroup, only 10% experienced a decline in walking ability, compared to 54% of patients receiving a saline placebo.
These findings, combined with other trial data, offer a clearer picture of the potential benefits and risks of these therapies.
Patient Outcomes and Safety Data
A meta-analysis of nine randomized controlled trials, involving 422 patients, demonstrated that stem cell transplantation led to an average improvement of -0.57 on the EDSS scale within two months. Imaging studies also showed an average reduction in brain lesion volume by 7.05 mm³, pointing to decreased disease activity.
In a separate study by Northwestern University, AHSCT was found to improve EDSS scores by -1.21 compared to disease-modifying therapies (DMTs), with a statistically significant p-value of 0.002, indicating measurable disability reversal.
"SCT can improve the disability of MS patients and reduce their brain lesion volume. The transplantation was generally safe and tolerated, with no mortality or significant serious AEs." - Scientific Reports
Safety profiles varied across treatments. Intrathecal MSC-NP injections caused mild, temporary headaches in 34% of patients. AHSCT, while effective, posed higher risks due to the aggressive immune conditioning required, including hematological disorders. However, recent trials reported no transplant-related deaths, and infusion site reactions were manageable. The San Raffaele Scientific Institute's STEMS trial (NCT03269071) followed 12 patients with progressive MS from May 2017 to May 2019. Higher doses of human fetal neural precursor cells were linked to reduced grey matter atrophy (p=0.02) over a two-year period, with no severe adverse reactions reported.
Future Directions in Stem Cell Therapy for MS
Current Trials and Next-Generation Therapies
The BEAT-MS trial (NCT04047628) is a six-year Phase 3 study comparing AHSCT (autologous hematopoietic stem cell transplantation) with natalizumab and ocrelizumab. Taking place across 19 sites in the U.S. and the UK, it involves 156 patients. Early trials of AHSCT have reported "No Evidence of Disease Activity" (NEDA) rates ranging from 70% to 90%, a marked improvement compared to the roughly 50% seen with other powerful MS therapies.
"The conditioning phase is the actual therapy for MS, as it provides potent immune suppression. The subsequent stem cell transplantation reboots the patient's immune system, but now in a milieu that no longer promotes MS activity." - Jeffrey Cohen, MD, Director of the Experimental Therapeutics Program, Cleveland Clinic
Advancements in AHSCT protocols have significantly reduced associated risks. Researchers are also exploring exosome therapy, which focuses on reducing inflammation and encouraging repair. Another exciting avenue involves combination therapies that pair stem cells with compounds like metformin or (R)-α-lipoic acid to boost the regenerative capabilities of the body's own oligodendrocyte progenitor cells.
These developments are pushing the boundaries of what’s possible, laying the groundwork for treatments tailored to each patient’s unique biology.
Personalized Stem Cell Treatments for MS
New insights from clinical trials are driving the rise of personalized therapies. One key innovation involves Induced Pluripotent Stem Cells (iPSCs), where researchers reprogram a patient’s skin or urine cells into iPSCs. These cells are then used for autologous treatments, bypassing ethical concerns and minimizing the risk of immune rejection. This approach eliminates the need for donor matching while reducing potential complications.
Another major step forward is the use of combined therapies. For example, MSC-derived Neural Progenitors (MSC-NPs) target both the immune system and neurodegeneration, offering the potential for direct neural repair and remyelination. Additionally, researchers are investigating antigen-specific tolerogenic dendritic cells, which aim to induce precise immune tolerance rather than broad suppression. Early Phase I trials (NCT02283671, NCT02903537, and NCT02618902) have already demonstrated the safety and feasibility of this targeted approach.
It’s important to note that early intervention is crucial. Stem cell therapies are most effective in younger, ambulatory patients with highly active disease and a recent onset.
Newborn Stem Cell Banking and MS Treatment
Americord Registry's Services for Regenerative Medicine
As advancements in MS treatment continue, storing high-quality stem cells at birth has become an important step for families looking to prepare for future therapies. Many of the MS treatments mentioned in this article rely heavily on access to premium stem cells, making newborn stem cell banking a practical choice for those planning ahead.
Americord Registry provides several plans - ranging from Essential to Ultimate - that preserve various types of stem cells, including hematopoietic, mesenchymal, placental, and exosome-derived cells. These cells support therapies like HSCT and other regenerative treatments for conditions like MS. The cost of processing ranges from $1,500 to $3,000, with annual storage fees between $175 and $250. Americord also offers a $110,000 quality guarantee, the highest in the industry, which compensates families if stored cord blood fails to engraft during a transplant.
"With the confidence of the technologies and processes we are providing to families who want to store stem cells from their newborn, as well as the breakthroughs we hope to bring to market in the coming years, we are excited to offer the highest quality guarantee in the industry." - Martin Smithmyer, CEO, Americord
Americord’s advanced 5-compartment storage system allows for multiple therapeutic uses, making it a valuable option for chronic conditions like MS. Between 2021 and 2023, researchers initiated 117 clinical trials involving cord tissue and 40 trials focused on cord blood. Additionally, nearly 41% of advanced cellular therapy trials conducted from 2020 to 2022 utilized perinatal stem cells. These figures highlight the growing interest in the same tissues preserved through newborn stem cell banking. By securing these cells, families are not only safeguarding a vital resource but also preparing for the development of personalized MS treatments.
Planning Ahead for Your Family's Health
Considering that MS often develops in early to mid-adulthood, newborn stem cell banking serves as a form of biological insurance. It preserves stem cells at their youngest and most effective state, potentially making them available decades later if needed.
Cord blood provides a perfect match for the child and has a 25% chance of matching siblings, significantly lowering the risk of rejection. The odds of an individual requiring a stem cell transplant by age 70 are approximately 1 in 217, while about 1 in 3 people could benefit from regenerative medicine or stem cell therapy during their lifetime.
Families considering this option should aim to enroll during the second trimester, as the American College of Obstetricians and Gynecologists (ACOG) advises securing a collection kit at least six weeks before the due date. The collection process is quick - taking just 5–10 minutes - and is safe, even with delayed cord clamping (30–60 seconds). Including cord blood collection in your birth plan and informing hospital staff upon arrival ensures a smooth process.
Americord’s 78-year lifetime storage plan is designed to provide access to these cells throughout a child’s life, aligning with the emergence of next-generation MS treatments. Many families use Flexible Spending Accounts (FSAs) or Health Savings Accounts (HSAs), along with a Letter of Medical Necessity, to help manage the costs of stem cell banking.
Conclusion
Stem cell therapy is changing the way we approach treatment for multiple sclerosis (MS). Instead of just slowing the disease's progression, these therapies actively work to repair damaged tissue and recalibrate the immune system. With millions of people impacted by MS worldwide, the need for advanced treatments has never been more pressing.
Hematopoietic and Mesenchymal Stem Cells play distinct roles in treatment; HSCT is a standout option for replacing harmful T cells, while MSCs provide strong neuroprotective effects, particularly for progressive forms of MS. Emerging therapies, such as MSC-derived neural progenitors and exosomes, are also showing immense potential, supported by growing clinical research.
Recent studies highlight these regenerative benefits:
"Stem cell therapy represents a promising therapeutic strategy for the treatment of MS... including differentiation into various neural cells, secretion of trophic factors, and immunomodulation." - Frontiers in Neurology
For families, banking newborn stem cells offers a forward-looking option. Preserving cord blood, cord tissue, and placental tissue at birth creates a flexible resource that could be key for future regenerative treatments targeting MS.
FAQs
What’s the difference between HSCT and MSC therapy for treating multiple sclerosis?
The key distinction between hematopoietic stem cell transplantation (HSCT) and mesenchymal stem cell (MSC) therapy lies in their goals and methods. HSCT aims to "reset" the immune system. This is done by first suppressing the existing immune system and then rebuilding it using the patient’s own hematopoietic stem cells. This approach has been particularly effective in reducing inflammation and slowing the progression of relapsing-remitting multiple sclerosis (MS).
On the other hand, MSC therapy is being investigated for its ability to repair damaged tissues, regulate immune responses, and encourage nerve regeneration. While MSCs hold potential due to these regenerative qualities, research is still ongoing to establish their clinical effectiveness and long-term outcomes.
In short, HSCT focuses on halting disease activity through immune system reprogramming, whereas MSC therapy is being studied for its potential to heal and protect against further damage in MS.
How can umbilical cord and placental stem cells help in treating multiple sclerosis (MS)?
Stem cells from umbilical cord and placental tissues are showing exciting potential in treating multiple sclerosis (MS) due to their healing and immune-regulating abilities. These cells could play a key role in repairing nerve damage, calming inflammation, and encouraging nerve regeneration - all crucial for managing MS effectively.
Researchers are focusing on stem cells from cord blood, cord tissue, and placental tissue for their ability to repair the myelin sheath, the protective layer around nerve fibers, while also helping regulate the immune system to address the autoimmune component of MS. With ongoing advancements, storing these stem cells through services like Americord Registry might one day offer access to tailored therapies that could slow or even reverse the progression of the disease.
How do exosomes contribute to advancements in multiple sclerosis (MS) treatment?
Exosomes are tiny extracellular vesicles, ranging from 30 to 150 nanometers, that play a crucial role in advancing treatments for multiple sclerosis (MS). These microscopic carriers transport therapeutic molecules such as proteins, RNA (including microRNA), and other bioactive compounds directly to target cells in the central nervous system (CNS).
In the context of MS therapy, exosomes derived from mesenchymal stem cells (MSCs) have shown great promise. They can help regulate immune responses, reduce inflammation, and even promote the repair of damaged nerve cells by supporting remyelination. One of their standout features is their ability to cross the blood-brain barrier, a significant obstacle in delivering treatments to the CNS.
Because exosomes are non-toxic and capable of carrying therapeutic agents, they represent a cell-free approach to MS management. This opens up possibilities for better immune regulation and neural repair, offering a potential new avenue for addressing the challenges of MS.
The views, statements, and pricing expressed are deemed reliable as of the published date. Articles may not reflect current pricing, offerings, or recent innovations.