History of Cord Blood Banking
Cord blood banking has transformed from medical waste to a life-saving resource. This process stores stem cells from umbilical cord blood, used in treating over 80 diseases like leukemia, sickle cell anemia, and metabolic disorders. Stem cells from cord blood also support regenerative medicine, with ongoing research into conditions like Parkinson’s and diabetes.
Key Highlights:
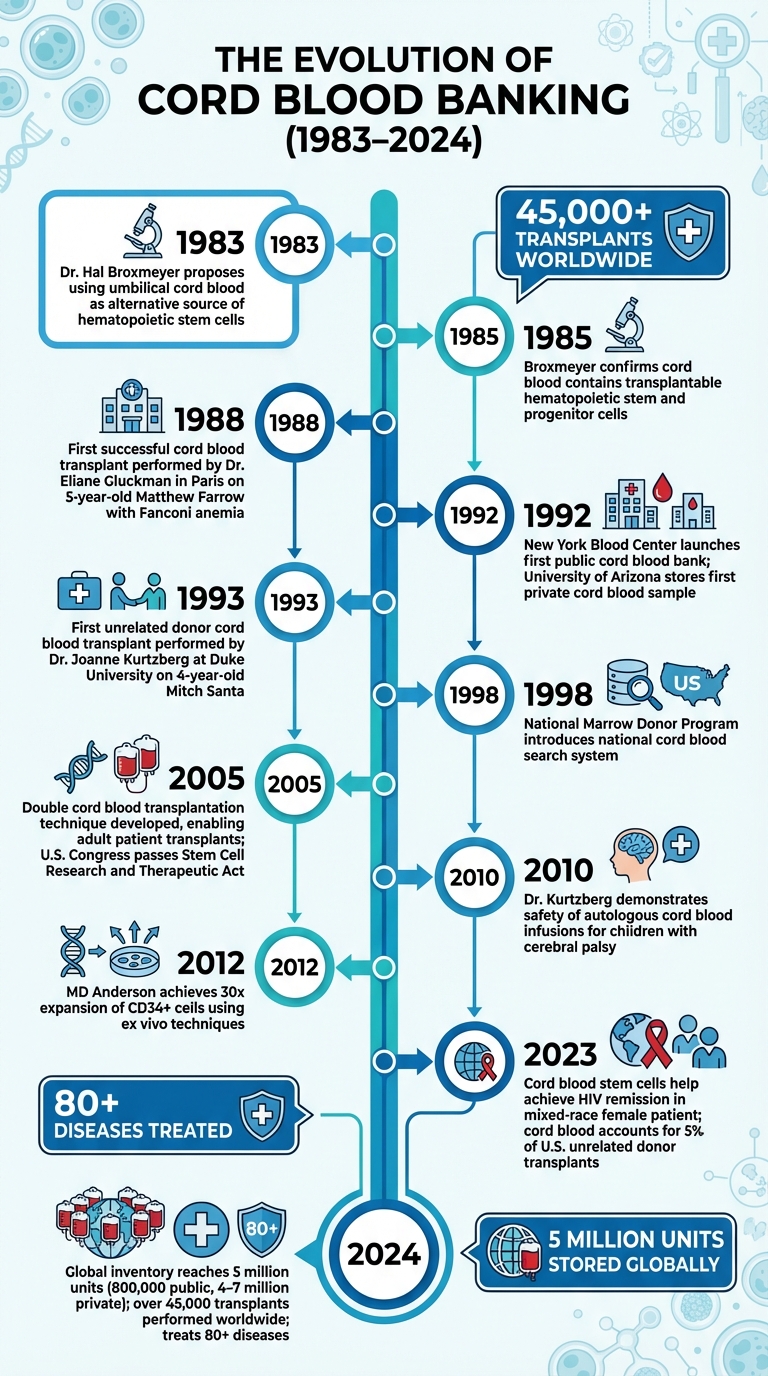
- 1980s Research: Dr. Hal Broxmeyer discovered hematopoietic stem cells (HSCs) in cord blood, leading to the first successful transplant in 1988 for Fanconi anemia.
- First Transplants: The 1988 sibling transplant and 1993 unrelated donor transplant proved cord blood's potential.
- Public and Private Banks: Public banks began in 1992, offering free donation options. Private banks emerged for family storage, growing into a global industry.
- Expanding Uses (2000–2020): Applications expanded to regenerative therapies, including cerebral palsy and Type 1 diabetes. Double cord transplants enabled adult treatments.
- Current Landscape (2020s): Over 45,000 transplants performed globally. Public banks hold ~800,000 units, while private banks store millions. Research focuses on cellular therapies and advanced storage methods.
Cord blood banking continues to evolve, offering hope for blood disorders and regenerative medicine advancements.
Early Foundations (1980s)
Early Research and Key Discoveries
The 1980s marked a pivotal moment in medical research when scientists began exploring the potential of discarded cord blood. In 1983, Dr. Hal Broxmeyer and his team proposed using umbilical cord blood as an alternative source of hematopoietic stem cells for transplantation. By 1985, Broxmeyer confirmed that cord blood contained transplantable hematopoietic stem and progenitor cells.
What stood out about this discovery was the presence of highly proliferative hematopoietic cell progenitors in cord blood - cells capable of regenerating blood and immune systems.
"Dr. Broxmeyer's research had confirmed that cord blood was enriched for highly proliferative hematopoietic cell progenitors - even more so than bone marrow - and hypothesized that cord blood could serve as a substitute donor for bone marrow transplantation." - Joanne Kurtzberg, M.D., President of the Cord Blood Association
In May 1989, Broxmeyer, along with Edward A. Boyse and Gordon W. Douglas, published a groundbreaking study in the Proceedings of the National Academy of Sciences. By analyzing over 100 human umbilical cord blood samples, they demonstrated that a single cord blood unit contained enough stem cells for hematopoietic reconstitution. They also showed that these cells remained viable for up to 72 hours during transport when stored at temperatures of 39°F or 77°F. This research laid the groundwork for the first human transplants and the creation of cord blood banks.
These discoveries opened the door to practical applications and advancements in storage techniques.
Moving Toward Clinical Use
As interest grew, researchers focused on refining preservation methods to meet clinical needs. They developed a cryopreservation process using DMSO, which allowed cord blood cells to be frozen long-term without losing their functionality. Studies later demonstrated successful recovery of these cells even after 24 years of cryopreservation.
To advance therapeutic applications, Broxmeyer, Ted Boyse, and Judy Bard founded the company Biocyte in the 1980s. They secured the first patent for freezing and banking cord blood and established a proof-of-principle bank in Broxmeyer's laboratory at Memorial Sloan Kettering Cancer Center. This facility assessed stem cell content and cryopreserved units for HLA-matched sibling transplants. These efforts validated the feasibility of cord blood banking and laid the foundation for the first transplants. By the end of the decade, the necessary infrastructure was in place to transition from research labs to life-saving clinical treatments.
First Successful Transplants (1988–1993)
The 1988 Fanconi Anemia Transplant
In October 1988, Dr. Eliane Gluckman at L'Hôpital Saint Louis in Paris performed the first successful cord blood transplant in the world. The patient, 5-year-old Matthew Farrow, was battling Fanconi anemia, a rare genetic condition that leads to bone marrow failure. The cord blood used for the transplant came from his newborn sister. Dr. Gordon Douglas collected it in North Carolina, Dr. Hal Broxmeyer cryopreserved it at Indiana University School of Medicine, and it was transported under highly controlled conditions.
Because patients with Fanconi anemia are highly sensitive to chemotherapy, Dr. Gluckman employed a reduced-intensity regimen using low-dose cyclophosphamide and total lymphoid irradiation. The donor cells engrafted in just 19 days, and Matthew's blood system was fully restored. Today, more than 30 years later, Matthew is healthy and living a normal life as a father.
This groundbreaking procedure demonstrated that cord blood contains enough viable stem cells for successful transplantation. It also proved that cord blood could be cryopreserved, transported, and used as a life-saving resource from what was previously considered medical waste. Following this success, researchers began exploring transplants using cord blood from unrelated donors.
First Unrelated Donor Transplant (1993)
Building on the success of sibling donor transplants, 1993 marked a turning point with the first cord blood transplant from an unrelated donor. Dr. Joanne Kurtzberg at Duke University performed the procedure on 4-year-old Mitch Santa, who had T-cell acute lymphoblastic leukemia resistant to treatment. The cord blood unit was obtained from a newly established public bank in New York.
What set this transplant apart was the HLA match - only 4 out of 6 loci matched. In bone marrow transplants, such a mismatch typically leads to severe graft-versus-host disease. However, the transplant succeeded, with Mitch achieving full donor chimerism, normal immune system function, and more than 15 years of remission without graft-versus-host disease.
"The success of these first transplants established cord blood as a viable alternative donor for patients unable to identify a matched related or unrelated donor for transplantation and paved the way for the entire field." - Joanne Kurtzberg, M.D., President of the Cord Blood Association
This achievement expanded cord blood therapy to the 70% of patients who do not have a matched sibling donor, highlighting the difficulty of finding a match in a public bank for certain populations. Within just two years, Duke University performed 25 consecutive unrelated donor transplants, proving the reliability of cord blood as an alternative to bone marrow. By 2013, 25 years after the first successful procedure, more than 30,000 cord blood transplants had been performed worldwide.
Building Banking Infrastructure (1990s–2000s)
During the 1990s and 2000s, the push to formalize cord blood banking gained momentum, fueled by early clinical achievements.
First Public Cord Blood Banks
In 1992, the New York Blood Center (NYBC), under Dr. Pablo Rubinstein and with funding from the NIH, launched the first public cord blood bank. This facility developed processes for collecting, processing, and storing cord blood for unrelated donor transplants. By the end of its first year, the bank had around 60,000 donations. A significant milestone came in 1993 when the NYBC supplied the cord blood used in a groundbreaking unrelated donor transplant at Duke University, proving that public cord blood banks could serve patients globally.
Inspired by NYBC's success, more public banks began to emerge across the U.S. In 1996, the FDA initiated the Cord Blood Transplantation Study (COBLT), which ran from 1997 to 2004. This program standardized essential practices like donor screening and cryopreservation. Duke University's Carolinas Cord Blood Bank became one of the largest public banks in the country and was among the first to receive FDA approval through a Biologic License Application. In 1998, the National Marrow Donor Program introduced a national system for cord blood searches, further advancing the field.
Rise of Private Cord Blood Banking
While public banks focused on donations for unrelated recipients, private banking arose to meet other needs. The University of Arizona in Tucson stored the first cord blood sample for family use in 1992, marking the start of private cord blood banking. This new industry offered families the chance to preserve their child’s stem cells for potential future treatments, often referred to as "biological insurance."
Private banking expanded rapidly due to limited public donation options, which led to the disposal of nearly 90% of cord blood as medical waste. Companies like Cryo-Cell International (founded in 1992), ViaCord (1993), and the Cord Blood Registry (CBR, 1995) entered the market. CBR eventually became the largest private cord blood bank worldwide, later acquired by AMAG Pharmaceuticals for $700 million in 2015, and by CooperSurgical for $1 billion in 2019. By 2013, private family banks held about 2.5 million units globally, compared to 650,000 in public banks. Families paid an initial fee of $1,500–$2,000, with annual storage costs ranging from $90 to $200.
Development of Regulatory Standards
As cord blood banking grew, the need for strict regulations became clear. In the mid-1990s, the FDA began overseeing cord blood under the Investigational New Drug framework, standardizing practices through the COBLT program. Organizations like the AABB and NetCord-FACT established international standards for quality management in cord blood banking, reflecting its global reach.
In 2005, the U.S. Congress passed the Stem Cell Research and Therapeutic Act, requiring the creation of a national inventory of 150,000 high-quality cord blood samples. By 2011, the FDA mandated that all unrelated donor cord blood units used in transplants in the U.S. either be licensed or covered under an FDA-approved IND. Public banks faced stricter requirements than private ones, including obtaining Biologics License Applications for unrelated donor products. Nearly 90% of collected cord blood units didn’t meet the rigorous clinical standards for storage and transplantation, underscoring the high bar set during this period. These advancements laid the foundation for future developments in cord blood use and research.
Expanding Medical Uses and Technology (2000–2020)
From 2000 to 2020, cord blood banking transitioned from a specialized treatment option to a key player in regenerative medicine. This period saw a steady rise in global transplants, driven by expanded medical applications and advancements in technology.
Broadening Treatment Possibilities
During these years, the scope of cord blood applications widened significantly. While it remained a cornerstone for treating leukemias and blood disorders through hematopoietic stem cell transplants, researchers began investigating its potential for non-hematopoietic conditions. A pivotal moment came in 2010 when Dr. Joanne Kurtzberg from Duke University demonstrated that autologous cord blood infusions were safe for children with cerebral palsy and acquired brain injuries. This finding opened new avenues in regenerative medicine. Clinical trials soon explored its use for conditions like Type 1 diabetes, autism spectrum disorder, and even strokes in adults. Another remarkable discovery was its ability to treat epidermolysis bullosa, a rare skin disorder, using sibling cord blood transplants to help the body produce missing skin proteins.
One of the most notable advancements was the development of double cord blood transplantation in 2005. Previously, single units of cord blood didn't provide enough cells for adult patients. By combining two partially matched units, researchers like Juliet Barker and John Wagner made transplants viable for adults, significantly increasing the number of eligible patients. This technique also reduced the median search time for a cord blood match to as little as two weeks, compared to the three to four months often required for bone marrow matches - an invaluable improvement for patients needing urgent care.
These breakthroughs in clinical applications laid the groundwork for significant technological progress in cord blood banking.
Advances in Technology and Storage
The clinical successes of cord blood therapies spurred innovations in how cord blood was processed and stored. In 2012, MD Anderson achieved a major milestone by expanding CD34+ cells up to 30 times using ex vivo techniques with mesenchymal progenitor cells. This advancement cut the median time for neutrophil engraftment from 24 days to just 15 days, speeding up recovery for patients. Banks also shifted from storing whole, unprocessed cord blood to preserving volume-reduced nucleated cell fractions, which saved space while maintaining stem cell integrity.
Long-term studies confirmed that cord blood could be cryopreserved for 20 to 25 years without losing functionality, reinforcing confidence in storage methods. Researchers also introduced "homing enhancement" techniques to improve how quickly stem cells migrated to the bone marrow post-transplant. For example, clinical trials led by Dr. Sherif Farag at Indiana University showed that using agents like sitagliptin reduced neutrophil engraftment times to a median of 19 to 21 days.
Cord Blood Banking Today (2020s)
Cord blood banking has grown into a global industry worth $46.23 billion, blending traditional transplant medicine with the promise of regenerative therapies.
Global Inventory and Usage Statistics
Globally, around 5 million units of umbilical cord blood are stored. Private banks are estimated to hold between 4 million and 7 million units, while public registries account for approximately 800,000 units. In the United States, the donor registry included over 246,500 cord blood units by late 2024, with 122,500 of those from the National Cord Blood Inventory. Despite these numbers, only 3% of American parents opted to bank their child's cord blood by 2021. Internationally, participation rates vary widely: Singapore leads with 30% of births banking cord blood, while Greece and Portugal each report about 10%.
To date, more than 45,000 cord blood transplants have been performed worldwide. However, in the U.S., cord blood accounted for just 5% of unrelated donor transplants in 2023, compared to 85% for peripheral blood stem cells and bone marrow. These statistics highlight both the progress made and the opportunities for future advancements in the field.
Current Research and Future Applications
The current focus of research reflects a shift toward "off-the-shelf" cellular therapies and regenerative medicine. For example, scientists are working on developing CAR T-cells and CAR NK-cells derived from cord blood, which offer a lower risk of rejection due to cord blood's unique immune properties. A significant breakthrough occurred in March 2023 when cord blood stem cells were instrumental in achieving HIV remission in a mixed-race female patient.
Research into ex vivo expansion techniques has also shown promise. Using small molecules like Nicotinamide and StemRegenin 1, scientists have reduced neutrophil engraftment times to just 10.5 to 15 days. Additionally, exosome-based therapies are being explored, with clinical trials underway for conditions such as autism spectrum disorder, Type 1 diabetes, and cerebral palsy.
Americord Registry's Contributions to Modern Banking
As the field evolves, companies like Americord Registry are redefining cord blood banking to meet the demands of future therapies. Americord goes beyond traditional storage by offering services that include cord blood, cord tissue, placental tissue, and exosome banking. This approach aligns with the trend of creating "family health ecosystems" to preserve multiple cell types for potential medical use.
The company’s CryoMaxx™ Processing technology and 5-compartment storage vials are designed to improve stem cell viability and provide greater flexibility for therapeutic applications. Martin Smithmyer, CEO of Americord Registry, shared:
"With the confidence of the technologies and processes we are providing to families who want to store stem cells from their newborn, as well as the breakthroughs we hope to bring to market in the coming years, we are excited to offer the highest quality guarantee in the industry."
Americord’s emphasis on transparency and personalized service reflects the growing consumer demand for clear, realistic information about storage options. Currently, the likelihood of using stored cord blood is estimated at 1 in 217 for family members by age 70.
Conclusion
Cord blood banking has come a long way, evolving from a groundbreaking discovery by Dr. Hal Broxmeyer in 1985 to a global industry shaping the future of personalized medicine. With over 45,000 transplants performed worldwide, it has already played a critical role in treating more than 80 serious conditions.
Key milestones, like the first successful transplant and the creation of public cord blood banks, laid the groundwork for cord blood to become a reliable alternative to bone marrow. This progress has provided hope to countless patients who previously lacked compatible donors.
Today, cord blood therapy continues to expand its horizons. Researchers are investigating its potential to address conditions like autism spectrum disorder, cerebral palsy, and Type 1 diabetes. Many experts believe cord blood cells could drive significant advancements in regenerative medicine in the years ahead.
With an infrastructure that includes approximately 800,000 public units and over 5 million private units, the field is well-positioned to meet both existing transplant demands and future therapeutic innovations. As research progresses, cord blood's role is expected to grow, transitioning from primarily treating blood-related disorders to becoming a key player in personalized regenerative medicine.
FAQs
How is cord blood different from bone marrow for transplants?
Cord blood and bone marrow are both sources of stem cells for transplants, but they differ in some important aspects. Cord blood is collected from a newborn's umbilical cord and placenta immediately after birth and is stored for potential future use. In contrast, bone marrow is extracted from inside bones, such as the hips or sternum, through a medical procedure. Cord blood has the advantage of being more forgiving when it comes to tissue mismatches, but it contains a smaller number of stem cells, which might make it less suitable for larger patients.
Why can cord blood work with a less-perfect HLA match?
Cord blood is unique because it can be used effectively even without a perfect HLA match. This is due to its stem cells being less likely to cause immune rejection. On top of that, cord blood has fewer immune cells, which lowers the chance of complications and makes partial-match transplants more viable.
What are cord blood’s most promising regenerative medicine uses today?
Cord blood holds exciting potential in regenerative medicine, primarily due to its hematopoietic stem cells (HSCs). These cells are already used in transplants to treat various blood and immune disorders. Emerging research points to even broader possibilities, including regenerative therapies for conditions like metabolic and neurologic diseases. Scientists are also working on techniques like stem cell expansion and improved engraftment, which could lead to better results and open up more treatment options for both children and adults.
The views, statements, and pricing expressed are deemed reliable as of the published date. Articles may not reflect current pricing, offerings, or recent innovations.




