Emerging Stem Cell Therapies for Age-Related Diseases
Aging increases the risk of diseases like osteoarthritis, Alzheimer's, and heart failure, with 66% of global daily deaths tied to age-related causes. Stem cell therapies offer a promising approach by repairing damaged tissues, reducing inflammation, and addressing the root causes of aging. Preserving young stem cells at birth, such as through banking cord blood, cord tissue, and placental tissue, ensures access to cells with peak regenerative potential for future treatments.
Key insights include:
- Stem Cell Exhaustion: Aging reduces stem cells' ability to repair tissues effectively.
- Therapeutic Potential: Stem cells help treat joint damage, neurodegenerative diseases, and heart conditions by regenerating tissues and improving immune responses.
- Exosome-Based Therapies: These cell-free treatments are safer alternatives, delivering regenerative benefits without risks linked to cell transplantation.
- Preservation at Birth: Banking newborn stem cells ensures access to younger, undamaged cells for future therapies.
Stem cell therapies are advancing rapidly, with clinical trials exploring their potential to combat age-related conditions and improve quality of life.
Limitations of Using Your Own Stem Cells as You Age
How Aging Affects Stem Cell Function
As we get older, our stem cells lose some of their regenerative abilities. One major factor is telomere shortening - each time a cell divides, these protective caps on chromosomes get a little shorter, eventually stopping the cells from replicating. Add to that mitochondrial dysfunction, which reduces energy production, and the increased presence of reactive oxygen species that damage cellular components, and it’s clear why aging takes a toll on stem cells.
A 2019 study by Lewis-McDougall revealed some striking findings: about 50% of cardiac progenitor cells from patients over 70 showed signs of senescence (like p16INK4A and SA-β-gal markers). These cells struggled to repair damaged hearts, unlike cells from younger donors aged 34–62.
"As organisms age, somatic stem cells progressively lose their ability to sustain tissue homeostasis and support regeneration." - Cell Stem Cell Review
Aging also brings accumulated DNA damage and epigenetic changes. Years of exposure to environmental stressors, radiation, and the natural wear and tear of cellular processes lead to somatic mutations. On top of that, the stem cell niche - the microenvironment that supports stem cells - begins to break down. This deterioration disrupts crucial signals that guide stem cells to repair tissues, making regeneration less effective. These challenges illustrate why preserving stem cells at birth can be so important for future therapies, as younger cells are far better equipped for regenerative medicine.
Benefits of Young, Preserved Stem Cells
Stem cells preserved at birth retain their full regenerative potential, unlike those affected by aging. These young cells have long telomeres, efficient mitochondria, and stable genomes because they haven’t endured years of division and stress. They excel at self-renewal and can differentiate into a wide range of specialized cells needed for tissue repair. Unlike aged stem cells, which often face limitations like differentiation bias, young cells remain highly adaptable and respond effectively to repair signals.
Cord blood and cord tissue stem cells are particularly valuable for preservation. They are collected at birth in a non-invasive way and are free from the cumulative damage that typically impacts adult stem cells. This makes them a powerful resource for regenerative therapies down the road.
Stem Cell Treatments for Age-Related Conditions
Treating Osteoarthritis and Joint Disorders
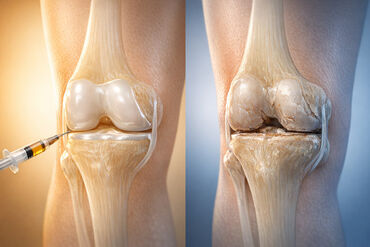
Mesenchymal stem cells (MSCs) are bringing new hope to people with knee osteoarthritis, a condition affecting over 360 million individuals worldwide. These cells work by transforming into cartilage cells, releasing growth factors that assist in repair, and calming inflammation in the joints.
One crucial factor in the success of these treatments is the source of the MSCs. Umbilical cord-derived MSCs (UC-MSCs) are gaining popularity due to their ability to multiply efficiently and deliver better clinical outcomes. Studies have shown that repeated injections of UC-MSCs into the joint provide longer-lasting pain relief compared to single injections or traditional treatments like hyaluronic acid.
A noteworthy development in this field is the growing interest in cell-free therapies, particularly those using MSC-derived exosomes. These tiny vesicles, measuring between 30 and 200 nanometers, carry the regenerative characteristics of their parent cells. They offer a safer alternative to whole-cell transplants by minimizing risks while still delivering powerful anti-inflammatory and healing signals. Researchers are even engineering these exosomes to carry specific therapeutic molecules, such as miR-140, which helps protect cartilage by targeting enzymes that degrade it.
| MSC Source | Key Advantages | Primary Mechanism in Osteoarthritis |
|---|---|---|
| Bone Marrow (BM-MSCs) | Well-researched, strong potential for bone repair | Repairs cartilage and reduces pain via the PGE2 pathway |
| Adipose (AD-MSCs) | Easy to collect, high cell production | Reduces joint inflammation and promotes healing |
| Umbilical Cord (UC-MSCs) | High yield, low immune rejection risk | Reduces cartilage breakdown and encourages cartilage growth |
Ongoing Phase III trials are now focusing not just on pain relief but also on rebuilding damaged tissue. These advancements highlight the importance of preserving young stem cells at birth, as their "youthfulness" directly impacts their effectiveness in treatments. The progress in joint therapy is also laying the groundwork for similar breakthroughs in treating neurological conditions.
Treating Neurodegenerative Diseases
Stem cell therapies are making strides in addressing conditions like Alzheimer’s and Parkinson’s disease. Between 2019 and 2024, over half of Parkinson’s clinical trials and about one-third of Alzheimer’s trials were initiated. By late 2025, a total of 76 trials had been conducted - 27 for Alzheimer’s and 48 for Parkinson’s.
These therapies aim to replace lost neurons, such as the dopamine-producing cells affected in Parkinson’s, while also reducing chronic brain inflammation and breaking down harmful protein buildups like amyloid-beta plaques.
For instance, a 2023 Phase I trial of Lomecel-B, which uses bone marrow-derived MSCs, showed promising results in patients with mild Alzheimer’s. The treatment improved vascular function and reduced inflammation markers (IL-4 and IL-6) compared to a placebo. Similarly, Medipost’s NEUROSTEM, derived from umbilical cord blood MSCs, was tested in a 2021 Phase 1/2a trial on nine patients aged 50–85. The treatment was not only safe and well-tolerated but also showed minimal side effects, such as temporary fever or headache.
"Stem cell-based therapies have emerged as a promising approach for treating various neurological disorders by harnessing the regenerative potential of stem cells to restore damaged neural tissue and circuitry." - European Journal of Medical Research
The shift toward MSC-derived exosomes is especially exciting. These cell-free treatments avoid complications like immune rejection or tumor risks while retaining the healing properties of stem cells. In animal studies, infusions of young bone marrow MSCs extended lifespans, demonstrating their potential. Advances in cardiovascular therapy further underscore how stem cells could transform treatments for age-related conditions.
Treating Cardiovascular Diseases
Cardiovascular disease remains the leading cause of death worldwide, claiming 17.9 million lives in 2019 - accounting for 32% of all deaths. While conventional medications manage symptoms, they don’t reverse the damage caused by events like heart attacks, which can destroy up to 1 billion heart cells. Compounding the problem, the body’s ability to replace heart cells declines sharply with age, dropping from 1% at age 25 to just 0.45% by age 75.
Stem cell therapies aim to do more than manage symptoms - they strive to regenerate lost heart tissue. MSCs play a key role through paracrine signaling, where they release exosomes and extracellular vesicles that reduce inflammation, encourage new blood vessel growth (angiogenesis), and protect existing cells from dying. They can even transfer healthy mitochondria to damaged heart cells, helping restore energy production.
In a Phase I trial (NCT02065245) completed around 2017, Longeveron treated 15 elderly patients with frailty using intravenous Lomecel-B. After six months, patients who received 100 million cells showed a 76.6-meter improvement in their six-minute walk test - a meaningful boost in physical function. Another trial, the BioVAT-HF Trial (NCT04396899), used epicardial patches made from stem cell-derived heart cells. Patients receiving the highest dose (800 million cells) experienced functional improvements, including a reduction in heart failure severity from stage III to stage II.
Meanwhile, Heartseed’s LAPiS Trial (NCT04945018) is testing heart muscle cell spheroids derived from stem cells in patients with severe heart failure. Early data suggests improvements in heart function and reduced levels of NT-proBNP, a marker of heart stress.
The trend toward cell-free therapies, such as exosomes, is gaining traction because they lower the risks tied to cell transplantation. Innovations like engineered heart patches also offer better structural support and higher cell retention. These advances highlight the potential of preserving stem cells early in life to ensure access to cutting-edge treatments for cardiovascular and other age-related conditions.
New Methods to Improve Stem Cell Therapy Results
Restoring Youthful Properties to Aging Stem Cells
Scientists are making strides in rejuvenating aging stem cells, aiming to restore their regenerative capabilities. One method gaining attention is partial reprogramming using Yamanaka factors - four proteins (OCT4, SOX2, KLF4, and c-MYC) that can reverse a cell's biological age. When applied briefly (2–4 days), these factors reduce the epigenetic age of cells without altering their identity or triggering tumor formation. For instance, in human fibroblasts, a 4-day treatment reduced DNA methylation age by 3.4 years, while endothelial cells showed a 4.94-year reduction.
Another approach focuses on Cdc42, a protein that becomes overactive in aging stem cells. By using CASIN, a Cdc42 inhibitor, researchers have restored the function of aged hematopoietic stem cells in mice. These rejuvenated cells, when transplanted, improved both healthspan and lifespan by restoring cell polarity and responsiveness to repair signals.
"The replenishment of biologically youthful adult stem cells into aged organisms may offer compelling evidence that the exhaustion of adult stem cells is not only a hallmark of aging but also a pivotal cause of mammalian aging." - An Yu, Researcher, Yunnan Key Laboratory of Cell Therapy
Chemical treatments are also showing potential. Compounds like Repsox, CHIR99021, and Y27632 help aged cells regain proliferative capacity and differentiation potential by pushing them into an intermediate plastic state. Additionally, drugs like rapamycin enhance autophagy, enabling aged neural stem cells to respond better to growth factors. These reactivated cells can even transfer healthy mitochondria to damaged tissues, potentially aiding conditions like Alzheimer’s and heart failure.
Exosome-Based Therapies
Exosomes are changing the game in stem cell therapy delivery. These tiny vesicles (30–150 nanometers) act like miniature versions of their parent cells, carrying proteins, lipids, and nucleic acids to damaged tissues. Unlike whole-cell transplants, exosomes avoid risks like immune rejection and tumor formation.
"MSC-Exos have shown enormous therapeutic potential as a safer and more efficient cell-free therapy in various aspects of clinical treatment." - Stem Cell Research & Therapy
Their size allows them to cross barriers like the blood-brain barrier and navigate through the microvascular system without being easily targeted by immune cells. A 2023 study demonstrated this in rabbits with intervertebral disc degeneration. Researchers injected 15 micrograms of MSC-derived exosomes into damaged discs, which reduced inflammation and supplemented mitochondrial proteins, leading to visible improvements in degenerative changes.
Exosomes can also be engineered to carry specific therapeutic cargo, such as miRNAs or drugs, and can be lyophilized for convenient storage, remaining effective for about four weeks at room temperature. For long-term storage, -80°C is ideal. A Phase 1 clinical trial (NCT06202547) is underway to test bone marrow-derived MSC exosomes in patients with premature ovarian failure, aiming to improve hormone levels and follicle counts.
Preserving Stem Cells for Long-Term Storage
With advancements in therapy, preserving youthful stem cells is crucial for future treatments. Cryopreservation ensures that stem cells retain their regenerative capacity. Cells collected from young donors - especially at birth - are far more potent than those from older individuals.
"Preserving the integrity and function of stem cells is crucial for effectively mitigating the pathological manifestations of aging and the onset of age-related diseases." - Zenghui Xu, Shanghai Cell Therapy Research Institute
Professional banking services use precise protocols to maximize cell recovery. This involves enzymatic digestion, filtration, and centrifugation, followed by controlled freezing to prevent ice crystal damage. Different tissues offer unique benefits: umbilical cord tissue provides an abundant, non-invasive source of mesenchymal stem cells, while adipose tissue offers a higher yield of stem cells than bone marrow but declines in quality after age 60.
Americord Registry provides extensive preservation options, including cord blood, cord tissue, placental tissue, and exosome banking. Their CryoMaxx™ Processing technology and multi-compartment storage vials ensure the long-term viability of stored cells. With plans like the Ultimate Family Plan and Maximum Family Plan - including newborn exosome banking - families can secure stem cells at their peak regenerative potential, offering a valuable resource for future therapies.
The Future of Stem Cell Therapies and Banking
Combining Stem Cells with Other Treatments
Stem cell therapies are evolving, and their future lies in combining them with other cutting-edge treatments. Researchers are pairing stem cells with growth factors like VEGF and FGF, platelet-rich plasma (PRP), and bioengineered scaffolds to enhance tissue repair and improve cell survival. These multimodal strategies tackle multiple challenges at once - stem cells bring their regenerative power, while growth factors and other components help them integrate and function effectively in damaged tissues.
There’s already promising clinical evidence for these approaches. For example, a 2019 Phase I/II study in China used umbilical cord-derived MSCs to treat rheumatoid arthritis. Patients experienced stable improvements lasting up to three years, including reduced joint deformities and significant drops in inflammation markers like CRP and ESR. These results highlight the importance of preserving stem cells, which is where services like those from Americord Registry come into play.
How Americord Registry Supports Future Therapies
The materials preserved by Americord Registry - cord blood, cord tissue, placental tissue, and newborn exosomes - are at the heart of today’s clinical research. By December 2024, 115 regulatory-approved clinical trials worldwide are testing 83 different stem cell products. Over 1,200 patients have already been treated, with more than 100 billion cells administered. Umbilical cord and placental tissues are key sources for mesenchymal stem cells (MSCs), which are being studied for their potential to treat neurodegenerative diseases, cardiovascular issues, and even age-related frailty.
"Stem cells harvested from young donors exhibit higher capacity to delay aging process." - Li He et al., Henan Cell Therapy Group
Preserved cells like these are essential for both current and emerging therapies. Americord’s plans, such as the Ultimate Family Plan and Maximum Family Plan, even include newborn exosome banking. These "pre-aging" cells are free from the telomere shortening and environmental damage that affect adult stem cells, making them a powerful tool for future treatments.
Planning Ahead for Your Family's Health
By combining advanced stem cell therapies with proactive preservation, families can create a safety net for their health. Banking newborn stem cells ensures access to cells at their peak regenerative ability. Research supports this idea - studies in rats have shown that transplanting young mesenchymal stem cells can extend lifespan by 23.4% to 31.3%. In one case, a 6-month-old rat treated with young MSCs lived 44 months, compared to the 36-month average of untreated animals.
The opportunity to preserve these cells is fleeting - they can only be collected at birth from tissues that would otherwise be discarded. Unlike adult stem cells, which require invasive harvesting and lose quality with age, newborn stem cells are collected non-invasively and retain their full regenerative potential. As precision medicine and cellular rejuvenation advance, families who bank these cells today are making a forward-thinking investment in personalized healthcare for future generations.
FAQs
What age-related diseases are stem cell therapies closest to treating?
Stem cell therapies hold immense potential for addressing a variety of age-related conditions, particularly those affecting the brain, heart, immune system, and degenerative processes. Scientists are zeroing in on diseases such as Alzheimer’s, atherosclerosis, and other disorders commonly tied to aging. Current research efforts are pushing boundaries to explore how these therapies can be further developed for regenerative medicine.
Are exosome therapies safer than transplanting stem cells?
Exosome therapies are gaining attention as a safer alternative to stem cell transplants. Thanks to their immunomodulatory and anti-inflammatory effects, they can help manage oxidative stress while avoiding many of the risks tied to cellular transplantation. This makes them a less invasive option, especially when addressing age-related conditions.
Why bank newborn stem cells instead of using my own later?
Newborn stem cells, like those found in cord blood or tissue, are incredibly versatile and hold greater regenerative power compared to adult stem cells. Over time, adult stem cells age and become less effective, while newborn stem cells remain youthful and free from the wear and tear of aging. They are also less likely to carry genetic mutations or damage, which makes them a safer option for potential medical uses. Plus, since these cells are a perfect genetic match for the individual, they significantly lower the risk of immune rejection and provide a reliable, long-term resource for future treatments.
The views, statements, and pricing expressed are deemed reliable as of the published date. Articles may not reflect current pricing, offerings, or recent innovations.