AI in Autoimmune Therapy: Measuring Success Rates
AI is transforming autoimmune disease treatments, especially stem cell therapies. By analyzing patient data, predicting outcomes, and improving therapy precision, AI is helping address challenges in treating conditions like lupus, multiple sclerosis, and Crohn's disease. Current stem cell treatment success rates vary by condition, with AI boosting effectiveness and safety. Advances include predictive models, digital monitoring, and personalized therapies based on molecular data. While challenges like data privacy and bias remain, AI's role in healthcare continues to grow, offering new possibilities for managing autoimmune diseases.
Key points:
- AI improves therapy success rates by predicting patient responses and enhancing safety.
- Stem cell therapy outcomes for autoimmune diseases range from 50% to 93% success, depending on the condition.
- AI tools like machine learning, digital twins, and federated learning are advancing personalized care.
- Challenges include data diversity, privacy concerns, and long-term monitoring.
The future of AI in autoimmune therapy includes earlier diagnosis, better treatment predictions, and more precise, patient-specific care strategies.
Current Success Rates of Stem Cell Therapies for Autoimmune Diseases
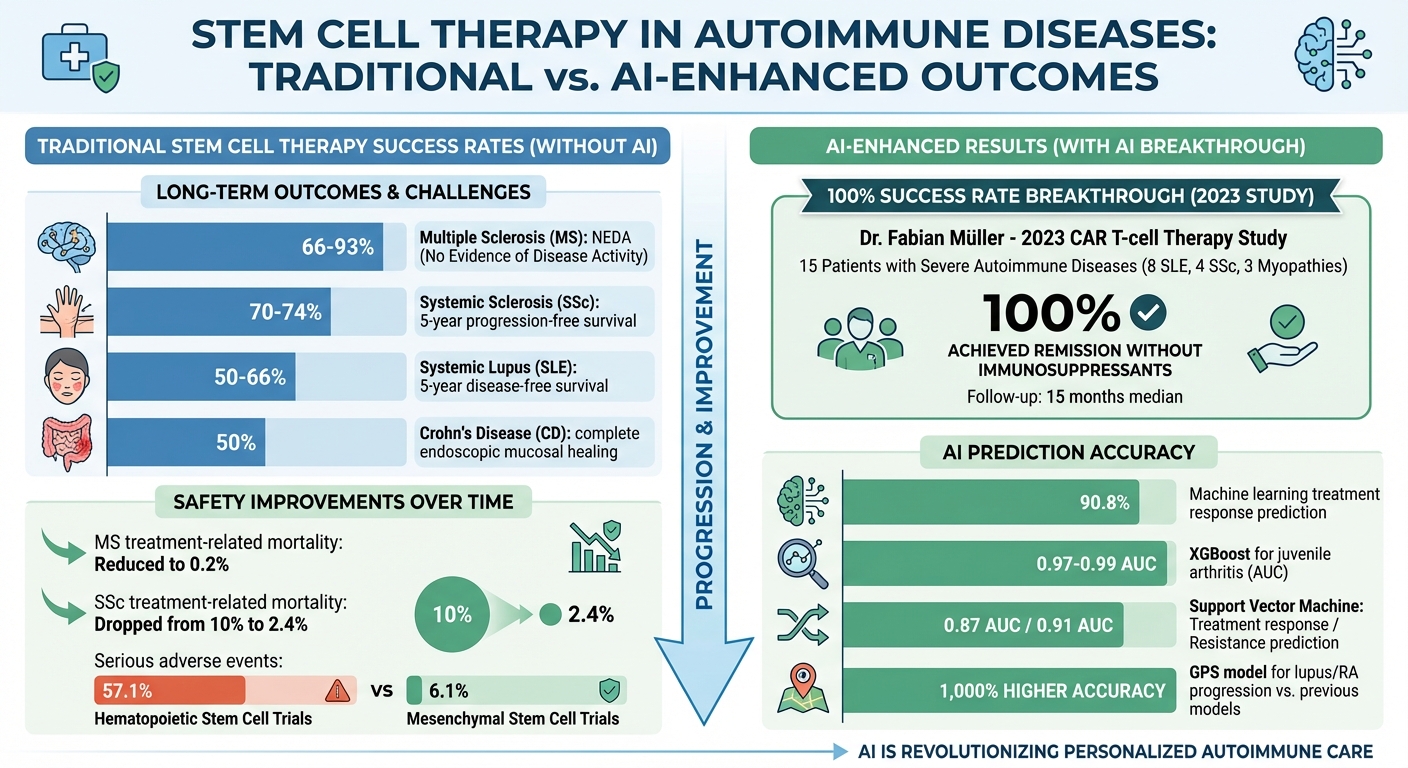
Stem Cell Therapy Success Rates for Autoimmune Diseases: Traditional vs AI-Enhanced Outcomes
Stem Cell Therapy Outcomes Without AI
The effectiveness of stem cell therapies for autoimmune diseases varies significantly, depending on the condition and how success is measured. For Multiple Sclerosis (MS), autologous hematopoietic stem cell transplantation (AHSCT) shows "No Evidence of Disease Activity" (NEDA) rates ranging from 66% to 93%. These outcomes are assessed using tools like the Expanded Disability Status Scale (EDSS), the Timed 25-Foot Walk for motor function, and MRI scans to monitor brain lesion volume.
In the case of Systemic Sclerosis (SSc), stem cell therapy results in 5-year progression-free survival rates of 70% to 74%, based on improvements in skin fibrosis scores and pulmonary function. For Systemic Lupus Erythematosus (SLE), success is defined by achieving seronegativity for antinuclear antibodies, with a 5-year disease-free survival rate of 50% to 66%. Meanwhile, for Crohn's Disease, the ASTIC trial found that 50% of patients achieved complete endoscopic mucosal healing after treatment.
| Autoimmune Condition | Success Metric | Reported Success Rate |
|---|---|---|
| Multiple Sclerosis (MS) | NEDA (No Evidence of Disease Activity) | 66%–93% |
| Systemic Sclerosis (SSc) | 5-Year Progression-Free Survival | 70%–74% |
| Systemic Lupus (SLE) | 5-Year Disease-Free Survival | 50%–66% |
| Crohn's Disease (CD) | Complete Endoscopic Mucosal Healing | 50% |
Safety has also improved over time. For example, treatment-related mortality (TRM) in MS has dropped to 0.2%, and for SSc, it has fallen from 10% to 2.4% according to the CAST trial. However, serious adverse events are still a concern, occurring in 57.1% of hematopoietic stem cell trials compared to 6.1% in mesenchymal stem cell trials. These results provide a baseline for evaluating how AI can further enhance outcomes.
Case Studies: Results After Adding AI
Recent advancements show how integrating AI into stem cell therapies can significantly boost success rates. In December 2023, a study led by Dr. Fabian Müller at the Bavarian Cancer Research Institute and Friedrich-Alexander University treated 15 patients with severe, refractory autoimmune diseases using CD19-targeting CAR T-cell therapy, a cutting-edge cellular engineering approach. The group included 8 SLE patients, 4 with SSc, and 3 with immune-mediated myopathies. Remarkably, all 15 patients (100%) improved to the point that they no longer needed immunosuppressants. After a median follow-up of 15 months, the first lupus patient treated remained in remission and was jogging five days a week - an incredible turnaround for someone who had been given only weeks to live.
"CD19 CAR T cells are well tolerated in B-cell driven autoimmune diseases, and the disease is entirely controlled without any additional disease-related therapeutics so far." - Fabian Müller, MD, Bavarian Cancer Research Institute
Another breakthrough came in 2023 from the University of British Columbia, where Dr. Nika Shakiba and her team used machine learning to monitor individual human pluripotent stem cells during large-scale production. This AI system detects "rogue" genetic variants that could compromise therapy batches by growing faster than healthy cells. By identifying these risks early, the system prevents costly losses and ensures safer, more reliable treatments.
These examples highlight how AI is transforming stem cell therapy by improving both safety and effectiveness, turning theoretical possibilities into real-world clinical successes.
AI Technologies Used in Autoimmune Therapy
Machine Learning for Predicting Treatment Results
Machine learning is transforming how clinicians predict patient responses to stem cell therapies. By analyzing complex clinical, genomic, and molecular data, these algorithms uncover patterns and biomarkers that human analysis might miss. This allows doctors to foresee complications like graft-versus-host disease, relapse risks, and treatment-related mortality - well before they occur.
One standout example is SCORPIO, an ensemble model developed in January 2025 by researchers at Memorial Sloan Kettering Cancer Center and Mount Sinai. This model analyzed routine blood tests from nearly 10,000 patients across 21 cancer types, significantly outperforming FDA-approved biomarkers. It offers a low-cost and effective tool for monitoring, making advanced care more accessible.
"The simplicity and affordability of this new approach could help ensure more equitable access to care while also reducing costs and helping ensure patients receive treatments most likely to benefit them individually." - Dr. Luc Morris, Surgeon and Research Lab Director, MSK
Machine learning models have also shown promise in other autoimmune conditions. For example, XGBoost algorithms achieved an area under the curve (AUC) of 0.97 to 0.99 when predicting treatment responses in juvenile idiopathic arthritis. Similarly, Support Vector Machine models reached an AUC of 0.87 for predicting treatment responses and 0.91 for forecasting resistance to glucocorticoids. These precision levels allow doctors to tailor treatments from the outset, paving the way for more personalized care.
AI-Based Customization of Stem Cell Therapies
AI is enabling a new level of personalized treatment by integrating genomic, transcriptomic, proteomic, and epigenomic data. This approach helps identify "endotypes", or molecularly defined patient subgroups, allowing therapies to be matched to specific immune mechanisms like interferon pathways or B-cell activity. Deep learning models further enhance this by analyzing microscopic images of live cell cultures, identifying the most potent mesenchymal stem cell subpopulations without invasive labeling. These insights ensure that each patient receives the most effective cells for their treatment.
In January 2025, researchers at Penn State College of Medicine introduced the Genetic Progression Score (GPS). By applying transfer learning to biobank data from Vanderbilt University and the "All of Us" initiative, this AI model predicted the progression of lupus and rheumatoid arthritis with up to 1,000% higher accuracy than earlier models. This capability allows for earlier intervention, potentially preventing irreversible organ damage.
AI is also advancing drug response predictions. For instance, models using DNA methylation profiles have achieved 85.9% accuracy in predicting responses to adalimumab and 88% for etanercept. These tools help doctors quickly move past ineffective treatments, focusing on therapies more likely to succeed for each individual patient. By aligning treatments with molecular profiles, AI-driven approaches are significantly improving success rates and driving progress in autoimmune therapy.
Continuous Monitoring and Post-Treatment Analysis
AI is playing a critical role in real-time patient monitoring, ensuring that potential issues are detected early and addressed promptly. Tools like digital phenotyping use data from smartphones and wearables to track mobility, heart rate, sleep, and fatigue. This continuous data offers a more complete picture of treatment outcomes than occasional clinic visits.
Digital twins - virtual patient models - are another groundbreaking tool. These simulations are trained on clinical data and lab results to replicate cellular interactions, allowing clinicians to test various treatment adjustments without risking patient safety. Additionally, natural language processing converts unstructured clinical notes into structured data, capturing side effects and patient feedback that might otherwise be overlooked.
Federated learning is also changing the game by enabling medical centers to train AI models on post-treatment data without sharing sensitive patient information. This ensures patient privacy while improving the accuracy of monitoring tools across diverse populations. By making advanced tracking more accessible, these technologies are helping improve care for patients nationwide.
Challenges in Using AI to Measure Success Rates
Accuracy and Reliability of AI Systems
AI systems face notable obstacles when it comes to evaluating the outcomes of stem cell therapies for autoimmune diseases. One major issue is the lack of large, high-quality datasets. For example, a review of 244 global clinical trials found that only 35.4% of Phase I trials were successfully completed, while 23% were terminated due to technological and financial hurdles.
Another challenge lies in the inconsistency of stem cell preparation methods. Differences in sourcing (like cord blood, bone marrow, or adipose tissue), expansion protocols, and processing techniques lead to varied data, making it difficult for AI systems to detect reliable patterns. On top of this, patient data is often scattered across specialists and electronic records, complicating the continuous monitoring required to track long-term therapy success. For instance, an artificial neural network showed promise with an AUC of 0.783 in predicting early responders among nearly 600 patients with ankylosing spondylitis. However, this result also underscores the need for further refinement to achieve consistent performance in real-world scenarios. These technical hurdles are compounded by ethical and privacy concerns, as explored below.
Data Privacy and Ethics
Developing AI systems while safeguarding patient data presents a significant challenge. Weak security measures can lead to data breaches, which limit the adoption of AI in healthcare. Furthermore, if AI training datasets lack demographic diversity, the models may perpetuate biases based on factors like race or gender.
Regulatory frameworks are also struggling to keep up with the rapid evolution of AI, particularly in high-stakes areas like immunosuppressive therapies.
"Flaws in these explanations, such as contradictions, omissions, or unsupported logic, can correlate with inaccurate or biased outputs and provide a flag for clinician oversight." - Arjun Mahajan, Harvard Medical School
One promising approach to address these issues is federated learning, which allows multiple healthcare systems to train AI models collaboratively without sharing raw patient data. This method could enhance both privacy and model robustness.
Expanding AI to More Conditions and Patients
AI also faces challenges in adapting to diverse patient groups. Many models, such as Polygenic Risk Scores, are often based on datasets from individuals of European ancestry, leading to inconsistent performance across different populations. This is a significant concern given that nearly one in ten people worldwide is affected by an autoimmune rheumatic disease. However, the rarity of specific conditions makes it difficult to create large, diverse datasets for AI training.
The fragmented nature of autoimmune care - spread across various specialists and healthcare systems - further complicates data integration. In the context of stem cell therapy, academic institutions fund about 49.2% of AI trials, while industry sponsors roughly 25%, with 83.6% of these trials still in early Phase I-II stages. Expanding AI's reach may require techniques like model compression (e.g., weight pruning and quantization), which enable AI tools to operate on mobile or edge devices, even in low-resource settings.
For rare diseases with limited patient numbers, generative AI offers a potential solution by creating synthetic patient profiles. These profiles can provide the statistical power needed for meaningful treatment comparisons.
"The convergence of registries, federated learning, and transparent reporting standards marks a pivotal step toward pragmatic, equitable, and continuously learning systems of care." - Tobias Alexander and Raffaella Greco
Conclusion: The Future of AI in Autoimmune Therapy
Key Takeaways
AI is changing the game in how we evaluate and improve stem cell therapies for autoimmune diseases. With machine learning models achieving a 90.8% accuracy in predicting treatment responses, and digital twins simulating cellular interactions to fine-tune strategies, the potential is immense. This technology brings precision medicine to life by combining genomics, imaging, and data from wearables to deliver care tailored to each patient. Advanced donor matching tools now analyze genetic markers and HLA compatibility to time transplants more effectively, while noninvasive imaging ensures stem cell quality. Research shows that AI-driven approaches can boost outcomes by 30–40% while slashing costs by up to half.
How Americord Registry Supports These Advances
Americord Registry plays a vital role in advancing AI-powered therapies. By storing cord blood, cord tissue, and placental tissue at birth, they preserve neonatal stem cells, valued for their regenerative potential and lower mutation rates. These stored cells provide the foundation for future treatments enhanced by AI. The company’s on-site cryopreservation safeguards critical genetic markers and HLA matching data, ensuring precise donor-patient matches. With stem cell therapies for autoimmune diseases showing an 80% success rate, banked cells offer families a crucial resource for tapping into AI-optimized solutions.
What's Next for AI and Regenerative Medicine
The future of AI in regenerative medicine looks promising. Digital phenotyping through smartphones and wearables could soon detect disease flares before physical symptoms arise. Meanwhile, fragmentomics - analyzing cell-free DNA - offers a minimally invasive way to monitor tissue inflammation in real time. Generative AI is also beginning to integrate complex multimodal data to diagnose rare autoimmune diseases that often stump human specialists. With 83.6% of global stem cell trials currently in early stages (Phase I–II), the next few years may bring a wave of late-stage validation studies. Additionally, the global gene therapy market, supported by AI-driven sequencing, is projected to hit $46.5 billion by 2030. Endotype discovery, which groups patients by specific molecular mechanisms, will further refine personalized regenerative treatments.
"The convergence of AI and regenerative medicine holds great transformative potential, promising effective treatments and innovative therapeutic strategies." - European Journal of Pharmacology
FAQs
How does AI predict if stem cell therapy will work for me?
AI is transforming stem cell therapy by analyzing massive datasets to uncover patterns in treatment outcomes. Machine learning dives deep into patient-specific details - like genetic, molecular, and clinical data - to predict how individuals might respond to therapy. By evaluating the quality of stem cells and forecasting treatment success, AI empowers clinicians to create tailored therapy plans. This is especially impactful for autoimmune diseases, where patient responses can differ greatly, helping improve strategies and boost overall success rates.
What counts as “success” in autoimmune stem cell therapy?
In autoimmune stem cell therapy, success is often defined by long-term remission of the disease and an improved quality of life. This means symptoms are reduced or stabilized, there’s no recurrence of the disease, and the immune system functions properly without attacking the body. It can also involve measurable changes, such as normalized immune cell profiles or results from imaging and functional tests tailored to specific conditions like multiple sclerosis.
How is my health data kept private when AI is used?
Protecting the privacy of health data is a top priority when using AI in stem cell therapies for autoimmune diseases. Key measures include encrypting data during transmission, anonymizing patient details, and strictly following U.S. regulations like HIPAA. To further secure sensitive information, AI systems are housed in access-controlled environments, backed by advanced cybersecurity measures and round-the-clock monitoring to detect and prevent breaches. These precautions are essential for maintaining compliance and building trust in the handling of personal health data.
The views, statements, and pricing expressed are deemed reliable as of the published date. Articles may not reflect current pricing, offerings, or recent innovations.