How Imaging Predicts Stem Cell Therapy Outcomes
Stem cell therapy offers new possibilities for treating autoimmune diseases, but predicting success remains a challenge. Advanced imaging techniques like MRI and PET/CT now provide doctors with detailed, real-time insights into how transplanted cells behave, without needing surgical biopsies.
Here’s what you need to know:
- MRI tracks structural and functional changes, showing how cells integrate and repair tissues.
- PET/CT detects cellular activity and inflammation with extreme sensitivity, identifying early issues like immune rejection.
- Imaging can differentiate between live and dead cells, helping clinicians adjust treatments faster.
- Combining imaging with AI enhances predictions, personalizing therapy by analyzing subtle patterns and patient-specific data.
These tools not only improve monitoring but also help tailor stem cell therapies for better outcomes in autoimmune diseases. This progress is a cornerstone of regenerative medicine and its role in modern healthcare.
Primary Imaging Methods for Stem Cell Therapy
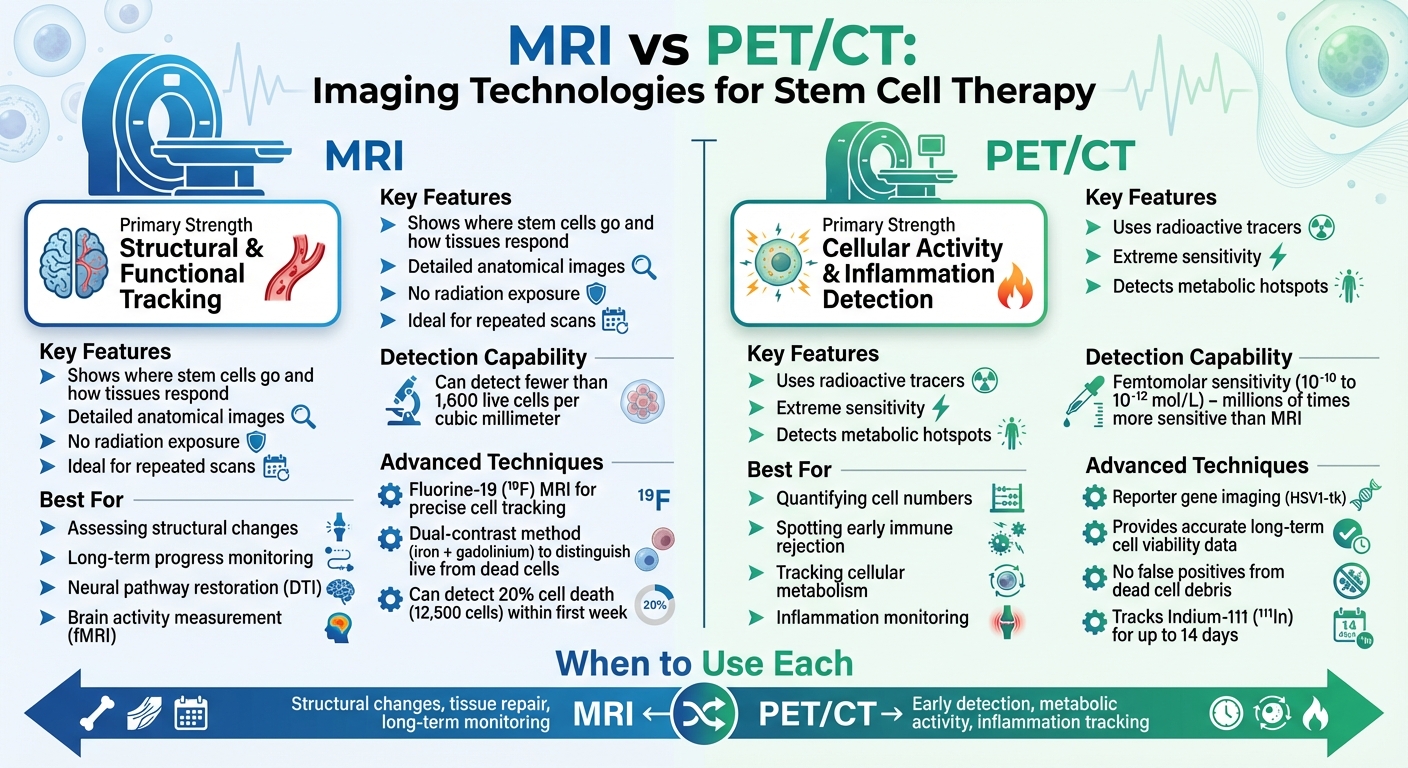
MRI vs PET/CT Imaging for Stem Cell Therapy Monitoring
When it comes to tracking stem cell therapies in clinical settings, two imaging technologies lead the way: MRI and PET/CT. Each method has its own strengths. MRI is exceptional for showing where stem cells go and how tissues respond, offering detailed anatomical images without exposing patients to radiation. This makes it a great option for repeated scans. On the other hand, PET/CT specializes in sensitivity, detecting cellular activity at concentrations millions of times lower than MRI. This capability makes PET/CT particularly effective for identifying metabolic changes and inflammation.
The choice between MRI and PET/CT depends largely on what clinicians need to measure. MRI is ideal for assessing structural changes and long-term progress, while PET/CT is better suited for quantifying cell numbers or spotting early signs of immune rejection. Let’s dive deeper into how each technology contributes to monitoring and predicting outcomes for different types of stem cells used in therapy.
MRI: Tracking Structural and Functional Changes
MRI plays a key role in stem cell therapy by capturing both structural and functional changes without the risks of radiation exposure. Standard T1 imaging reveals anatomical updates, such as shrinking lesions or tissue repair, while advanced techniques focus on cellular activity.
For example, Diffusion Tensor Imaging (DTI) maps nerve fiber connections in the brain and spinal cord, making it possible to see if transplanted cells are helping restore damaged neural pathways. In conditions like multiple sclerosis, DTI has shown improved connectivity even before symptoms improve. Functional MRI (fMRI) takes it a step further by measuring brain activity, confirming whether structural changes actually lead to functional recovery.
A specialized approach, Fluorine-19 (¹⁹F) MRI, allows precise cell tracking. Since the human body contains no natural fluorine, fluorine-labeled stem cells appear as bright hotspots against a dark background. A 2015 study at Johns Hopkins University demonstrated a dual-contrast method using iron particles (T2 contrast) and gadolinium (T1 contrast) to distinguish live cells from dead ones. When just 20% of the transplanted cells died (about 12,500 cells), gadolinium released by the dying cells created a visible T1 signal, alerting doctors to potential graft rejection within the first week.
This level of detail enables clinicians to adapt therapies quickly based on observed changes in tissue structure and function.
PET/CT: Tracking Cellular Activity and Inflammation
While MRI focuses on anatomical and functional details, PET/CT stands out for its ability to detect cellular activity with extraordinary sensitivity.
PET/CT uses radioactive tracers to monitor cellular metabolism and function. With femtomolar sensitivity, it can detect metabolic hotspots, making it invaluable for spotting inflammation or immune responses early.
A particularly useful technique in stem cell therapy is reporter gene imaging. Here, genes like HSV1-tk are inserted into stem cells before transplantation. These genes generate signals only when the cells are alive and functioning. Unlike direct labeling methods, which can produce false positives from dead cell debris, reporter genes provide accurate long-term data on cell viability.
Using Imaging to Predict Outcomes in Autoimmune Diseases
When treating autoimmune conditions with stem cell therapy, imaging plays a key role in revealing how the body responds internally. It doesn’t just show where the cells travel - it provides early clues about whether the treatment is likely to succeed or not.
This predictive ability comes from tracking specific markers. In diseases like multiple sclerosis and rheumatoid arthritis, imaging identifies inflammation, tissue damage, and immune responses. With this information, doctors can fine-tune treatment plans without waiting for visible symptoms to appear. It also helps distinguish areas that might recover from those that are beyond repair.
Penumbra vs Core Analysis: Identifying Recovery Zones
Imaging goes beyond surface-level observations by using advanced markers to separate recoverable tissue from irreversibly damaged areas. Specifically, imaging identifies the "core" (irreparable tissue) and the "penumbra" (damaged but potentially recoverable tissue). In neuroinflammatory conditions, 23Na MRI is used to evaluate ionic balance restoration in these recovery zones.
A study from July 2022 highlighted this approach in a rat stroke model. Researchers found that within just 24 hours of stem cell therapies and clinical trials, imaging could detect improvements in sodium balance and lower lactate levels. These changes correlated with functional recovery over the next 21 days. Similar trends are now being explored for autoimmune neuroinflammatory disorders.
MRI also tracks whether stem cells are migrating toward these recovery zones. When cells successfully target the penumbra instead of the irreversibly damaged core, it indicates a better chance of treatment success.
Measuring Inflammation and Immune System Changes
Imaging also offers a way to monitor inflammation, which is a key indicator of how the body is responding to treatment. For example, radionuclides like Indium-111 (¹¹¹In), used in SPECT imaging, can track inflammatory cell movement for up to 14 days. This helps determine whether the immune response is calming down or ramping up.
Combining AI with Imaging Data
Artificial intelligence is revolutionizing how medical professionals interpret imaging results. By detecting subtle changes that might go unnoticed in manual analysis, AI can flag whether stem cell therapy is effective. Let’s explore how AI is advancing pattern recognition and paving the way for tailored treatment plans.
AI for Pattern Recognition and Outcome Prediction
Convolutional Neural Networks (CNNs) are making waves in stem cell imaging, achieving up to 97.5% accuracy in classifying cell types and forecasting behavior post-transplantation. By analyzing intricate morphological features, these systems enhance established imaging methods like MRI and PET/CT, improving the precision of treatment predictions.
In January 2025, Dr. Dajiang Liu and his team at Penn State College of Medicine introduced the Genetic Progression Score (GPS). This AI model surpasses existing tools by an impressive 25% to 1,000% in predicting disease progression for individuals with early autoimmune symptoms. Dr. Liu highlighted its clinical impact:
"By targeting a more relevant population - people with family history or who are experiencing early symptoms - we can use machine learning to identify patients with the highest risk for disease and then identify suitable therapeutics that may be able to slow down the progression of the disease."
Another breakthrough comes from U-Net architectures, which excel at predicting protein expression from standard imaging. A 2021 study combined U-Net with a conditional generative adversarial network, achieving a correlation coefficient of 0.77 when predicting eight stem cell markers. This method eliminates the need for chemical dyes, enabling real-time monitoring of live cells for up to 48 hours without causing damage.
Personalizing Treatment with AI-Driven Analysis
AI goes beyond identifying patterns - it integrates a wide range of patient data to create highly individualized treatment plans. By combining imaging results with genomic profiles, immune markers, and medical histories, AI helps doctors determine the best newborn stem cell type and dosage for each patient’s unique needs.
Transfer learning addresses the issue of limited patient data by training AI models on large, general datasets before fine-tuning them with smaller clinical datasets. This method boosts predictive accuracy even when only a handful of patient scans are available. For example, in predicting rheumatoid arthritis treatment outcomes, AI models using transfer learning have achieved AUC values between 0.78 and 0.82, indicating reliable performance.
AI also enables label-free imaging, which allows continuous, noninvasive monitoring of cell health. By analyzing raw microscopic images, AI can assess cell viability, detect aging cells that could compromise therapy, and predict differentiation potential. This ensures that only healthy, effective cells are used, reducing the risk of treatment failure before they reach the patient.
Americord Registry: Supporting Regenerative Medicine
Dependable imaging outcomes rely on consistent, high-quality cell sources. Americord Registry plays a vital role in meeting this need through its specialized banking services.
Cord Blood and Tissue Banking for Future Treatments
The effectiveness of imaging-guided stem cell therapies hinges on having standardized, high-quality cell sources. Americord Registry addresses this by banking umbilical cord mesenchymal stem cells (UC-MSCs) and hematopoietic stem cells, both of which are critical in autoimmune disease studies.
UC-MSCs are particularly significant, representing 73.0% of the cell types used in autoimmune disease trials. Their widespread use stems from their ease in allogeneic treatments and their strong immunomodulatory capabilities. Standardized banking ensures compatibility for labeling, which is essential for long-term MRI tracking. For instance, a 2018 Radiology case showed successful monitoring of SPIO-labeled cord blood cells over a period ranging from 24 hours to 33 months post-transplant in a patient with global cerebral ischemia.
Moreover, by providing well-characterized biological materials, cord blood and tissue banking helps improve the precision of AI models used to predict cell behavior post-infusion. This consistency strengthens the reliability of imaging-based predictions.
Exosome Banking: New Treatment Possibilities
Americord is also advancing treatment options through the innovative storage of exosomes.
Exosome preservation supports the development of cell-free therapies, which eliminate many challenges linked to live cell transplants. Exosomes are nanoscale vesicles (30–150 nm) that transport regulatory molecules like miR-21 and miR-146a. These molecules can penetrate tissues more efficiently, offering immediate, ready-to-use treatment options with reduced toxicity risks.
Unlike live cell therapies, which can lead to complications such as cytokine release syndrome and neurotoxicity, exosome-based treatments provide a safer, non-replicative alternative. Advanced imaging techniques, including PET/CT, allow for precise tracking of these vesicles as they distribute throughout the body and interact with the immune system. With femtomolar-range sensitivity (10⁻¹² mol/L), PET/CT offers far greater precision compared to standard MRI tracking, giving clinicians a clearer picture of therapy outcomes.
Conclusion
This guide explored how imaging, quality cell banking, and AI integration are shaping the future of regenerative medicine. Clinical imaging has revolutionized stem cell therapies for autoimmune diseases by offering real-time feedback on cell delivery, survival, and integration. This reduces uncertainties in treatment outcomes. For instance, MRI can detect fewer than 1,600 live cells per cubic millimeter, while PET imaging achieves sensitivity levels between 10⁻¹⁰ and 10⁻¹² M. These capabilities allow clinicians to track even small populations of transplanted cells with precision.
Advanced MRI techniques also enable the differentiation of live and dead cells, reducing the risk of false-positive signals. As Ethel J. Ngen from Johns Hopkins University explains:
"Preclinical, translational, and clinical research on cell-based therapies will benefit tremendously from novel imaging approaches that enable the effective monitoring of the delivery, survival, migration, biodistribution, and integration of transplanted cells."
These imaging advancements rely on the availability of high-quality, standardized cell sources. Services like Americord Registry provide essential biological materials - such as cord blood, cord tissue, and exosomes - that are critical for successful therapies. With over 14,831 completed and 8,325 ongoing clinical trials globally for cell-based therapies, the demand for reliable cell sources continues to rise.
AI integration adds another layer of refinement. By analyzing imaging data, AI enhances predictions and allows for personalized treatment adjustments based on patient-specific responses. This blend of advanced imaging, dependable cell banking, and computational analysis is paving the way for a new era in regenerative medicine. It’s a future where treatments can be monitored, validated, and optimized throughout the therapeutic process, giving clinicians the tools to predict and improve outcomes for stem cell therapies in autoimmune diseases.
FAQs
How do MRI and PET/CT scans help improve the success of stem cell therapies for autoimmune diseases?
MRI and PET/CT scans are game-changers when it comes to improving the outcomes of stem cell therapies for autoimmune diseases. These imaging tools give doctors the ability to monitor stem cell delivery, follow their movement through the body, and evaluate their health over time - all without invasive procedures. This real-time feedback allows for adjustments to treatment plans, helping to improve the chances of success.
By showing how stem cells interact with the immune system and damaged tissues, MRI and PET/CT imaging provide the clarity needed for more targeted interventions. This not only boosts the effectiveness of therapies but also helps minimize risks, leading to better care and improved results for patients.
How does artificial intelligence improve predictions and outcomes in stem cell therapies?
Artificial intelligence (AI) is revolutionizing stem cell therapies by improving the way clinical imaging tools like MRI and PET/CT are used to predict and monitor treatment outcomes. These advanced algorithms can process imaging data with incredible accuracy, pinpointing key details like stem cell viability, migration patterns, and the success of cell delivery. For example, AI can identify subtle contrasts in imaging to differentiate between live and dead cells, offering valuable insights into how effective a therapy is.
Another game-changing aspect of AI is its ability to combine data from various imaging techniques. By doing so, it creates a more complete picture of how the body is responding to treatment, enhancing both the precision and safety of preclinical and clinical evaluations. As AI technology continues to evolve, these tools are expected to make therapies even more patient-specific, tailoring treatments to meet individual needs and improving overall effectiveness.
Why are high-quality, standardized stem cell sources essential for imaging-guided therapies?
High-quality, standardized stem cell sources play a key role in the success of imaging-guided therapies by ensuring consistent and reliable results. These cells make it possible to track progress with precision during imaging, assess their viability accurately, and deliver them effectively to the desired areas.
Standardized cells also help minimize risks like immune rejection or unexpected side effects, promoting safer and more predictable outcomes. This is particularly crucial when addressing complex conditions such as autoimmune diseases, where both precision and safety are essential for achieving lasting results.
The views, statements, and pricing expressed are deemed reliable as of the published date. Articles may not reflect current pricing, offerings, or recent innovations.