Long-Term Safety of Stem Cell Therapy for Crohn's
Stem cell therapy for Crohn's disease is proving to be safe and effective for long-term use. Research shows that Mesenchymal Stem Cells (MSCs) not only reduce inflammation but also promote healing in severe cases like perianal fistulas. Unlike traditional treatments, MSCs work by rebalancing the immune system without causing systemic risks like infections or cancers.
Key takeaways:
- MSCs reduce inflammation by reprogramming immune cells.
- Long-term studies (up to 4 years) show no therapy-related serious side effects.
- MSCs maintain remission in over 50% of patients, with localized side effects being minimal.
- They offer a safer alternative for patients unresponsive to standard drugs like anti-TNF agents.
Studies confirm MSC therapy's strong safety profile, making it a promising option for patients seeking lasting relief with fewer risks.
Long-Term Study Results on MSC Therapy Safety
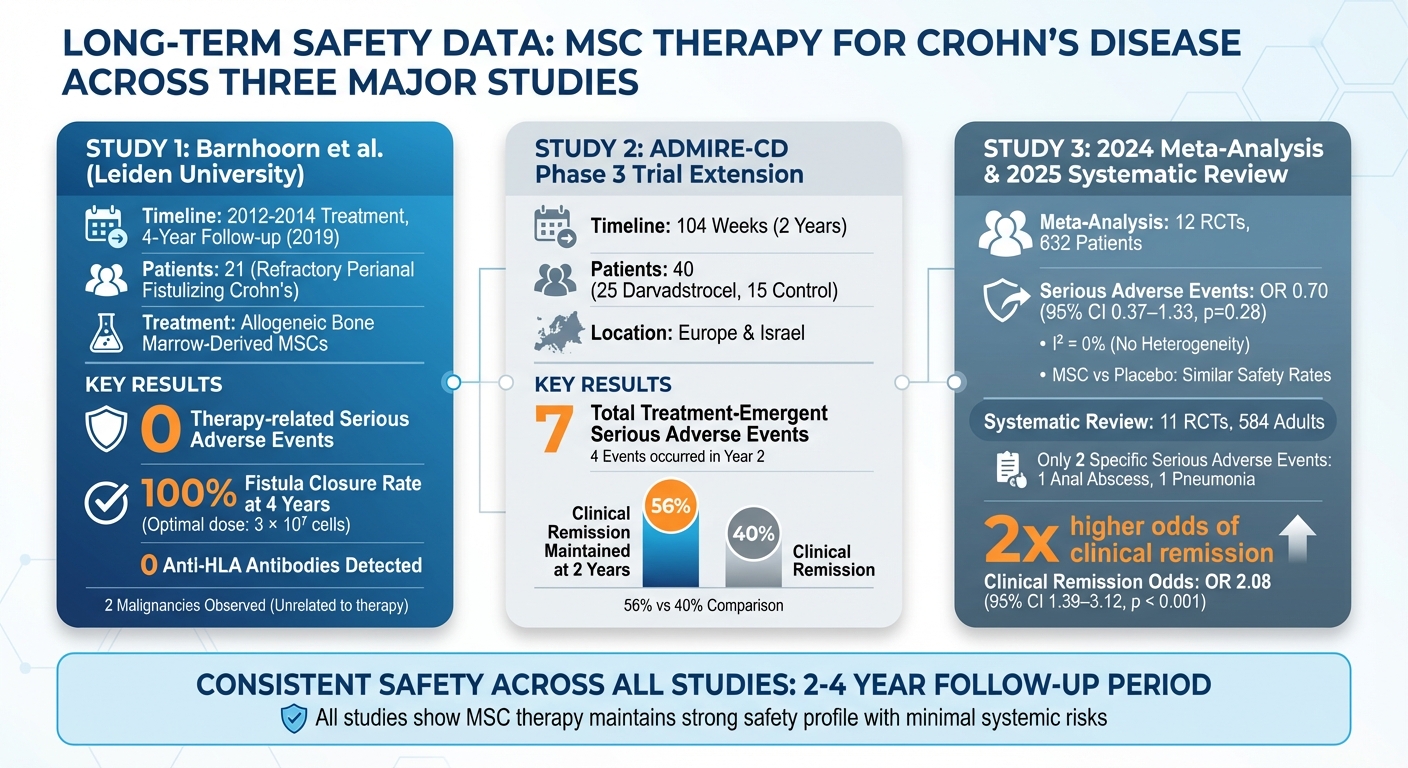
Long-term research continues to confirm the safety of MSC therapy for treating Crohn's disease. Numerous clinical studies have tracked patients for years after receiving MSC therapy, consistently showing a strong safety record. These studies evaluated adverse events over periods ranging from 2 to 4 years.
Results from the Barnhoorn et al. Study
From 2012 to 2014, Leiden University Medical Center treated 21 patients with refractory perianal fistulizing Crohn's disease using allogeneic bone marrow-derived MSCs. A follow-up study published in 2019, spanning four years, reported no therapy-related serious adverse events among the patients. Although two malignancies were observed during the follow-up, researchers concluded these were unrelated to the MSC therapy. Importantly, no anti-HLA antibodies were detected, indicating that the allogeneic cells did not provoke an immune response even years post-treatment. In the group receiving the optimal dose (3 × 10⁷ cells), all fistulas remained closed after four years.
"Allogeneic bmMSC therapy for CD-associated perianal fistulas is also in the long-term a safe therapy." - Barnhoorn et al.
Findings from the UK MSC Therapy Study
The ADMIRE-CD phase 3 trial extension monitored 40 patients over 104 weeks across Europe and Israel. This included 25 patients treated with darvadstrocel, an allogeneic adipose-derived MSC therapy, and 15 control patients. By the end of the study, only seven treatment-emergent serious adverse events were reported, with four occurring during the second year. Clinical remission was maintained in 56% of the darvadstrocel group at two years, compared to 40% in the control group.
"Darvadstrocel was well tolerated and clinical remission after treatment with darvadstrocel may be sustained for up to 104 weeks in patients with perianal fistulizing Crohn's disease." - ADMIRE-CD Study
Combined Data from Multiple Clinical Trials
A 2024 meta-analysis reviewed 12 randomized controlled trials involving 632 Crohn's disease patients. The analysis found that serious adverse events occurred at similar rates between MSC therapy and placebo groups, with an OR of 0.70 (95% CI 0.37–1.33, p=0.28) and I² = 0%. Additionally, a 2025 systematic review of 11 randomized controlled trials, covering 584 adults, identified only two specific serious adverse events - one case of anal abscess and one case of pneumonia. MSC therapy, however, more than doubled the odds of achieving clinical remission (OR 2.08, 95% CI 1.39–3.12, p < 0.001). The findings also highlighted the safety and effectiveness of local injections for perianal fistulas, regardless of the stem cell source - be it bone marrow-derived, adipose-derived, or umbilical cord-derived MSCs.
These comprehensive results strengthen the case for MSC therapy as a promising alternative to standard treatments for Crohn's disease.
Safety Profile Compared to Other Treatments
Side Effects and Adverse Events
MSC (Mesenchymal Stem Cell) therapy is primarily associated with localized side effects rather than widespread systemic complications. The most frequently reported issues include proctalgia (rectal pain), anal abscesses, and pain at the procedural site. For patients undergoing autologous MSC treatments, minor complications may arise at the fat collection site, such as seroma (fluid buildup) or discomfort related to liposuction.
Long-term studies have shown no development of anti-HLA antibodies, highlighting MSC therapy's targeted and localized safety profile. These side effects stand in stark contrast to the broader systemic risks often seen with standard treatment options.
MSC Therapy vs. Standard Treatments
While MSC therapy focuses on localized effects, traditional treatments come with more widespread systemic risks. Medications like azathioprine, methotrexate, and anti-TNF agents are known to suppress the immune system, which can lead to serious complications such as opportunistic infections, malignancies, and unpredictable immune responses. This systemic immune suppression increases the risk of severe infections and may even heighten the likelihood of developing certain cancers.
A 2024 umbrella review found that the overall safety profiles of MSC therapy and conventional treatments are comparable, with risk ratios close to 1. However, the key difference lies in the nature of the risks. Conventional therapies often result in systemic immunosuppression, while MSC therapy's effects remain primarily localized.
"Stem cell therapy offers significant progress in treating Crohn's disease... Its safety profile aligns with conventional treatments." – Umbrella Review, 2024
At the two-year mark, MSC therapy showed a healing rate of 41.6%, slightly surpassing Infliximab's 40%. In contrast, standard immunosuppressants demonstrated no healing. This data underscores MSC therapy as a strong long-term alternative, particularly for those seeking options with reduced systemic risks.
Stem Cell Sources for Regenerative Treatments
Cord Tissue-Derived Mesenchymal Stem Cells
Mesenchymal stem cell (MSC) therapy has shown a strong safety profile and long-term benefits, but the source of these cells is crucial for improving outcomes in conditions like Crohn's disease.
Umbilical cord tissue-derived MSCs (UC-MSCs) offer several advantages. They multiply quickly, have low immunogenicity, and can be collected noninvasively at birth. These cells may also aid in gut healing by activating the Wnt/β-catenin signaling pathway, which supports the regeneration of the intestinal lining.
Studies suggest that UC-MSCs can reprogram macrophages into an anti-inflammatory state through a process called efferocytosis. Although the cells themselves are short-lived, this reprogramming has lasting therapeutic effects that extend well beyond their presence. This unique ability highlights the promise of UC-MSCs in regenerative treatments for Crohn's disease, complementing the therapy's already established safety profile.
The Value of Stem Cell Banking
Preserving these powerful cells is essential for ensuring future treatment options for Crohn's patients. Since cord tissue MSCs must be collected at birth, newborn stem cell banking becomes a critical step. Companies like Americord Registry specialize in newborn stem cell preservation, offering services for cord blood, cord tissue, placental tissue, and exosomes.
Conclusion: Safety Evidence and Future Directions
What Patients and Families Should Know
A 2024 meta-analysis of 12 trials, involving 632 patients, provides strong evidence of the safety of MSC therapy. Serious adverse events were found to be on par with placebo (OR 0.70, 95% CI 0.37–1.33, p = 0.28), and no anti-HLA antibodies were detected over a four-year period. The Barnhoorn et al. study further supports this, reporting no therapy-related serious adverse events even after four years of follow-up.
For patients with refractory Crohn's disease - those who haven't responded to conventional treatments like corticosteroids or anti-TNF agents - MSC therapy presents a safe alternative. The ADMIRE-CD study highlights that 56% of patients treated with darvadstrocel maintained clinical remission at 104 weeks. Importantly, only seven serious adverse events occurred over two years, none of which affected its tolerability. This consistent safety record underscores MSC therapy's role as a promising option in managing Crohn's disease.
With this solid foundation of safety, researchers are now focusing on advancing MSC therapies to improve their effectiveness.
Future Developments in Stem Cell Therapy
Exciting advancements are being made in the field of MSC therapy. One area of focus involves mRNA-engineered MSCs that express CXCR2 receptors, which enhance the cells' ability to migrate to inflamed tissues. Another innovation is the MSC-MATRIX, a bioabsorbable plug containing MSCs. In phase I trials, this system achieved an 83% combined clinical and radiographic remission rate at 24 weeks. Additionally, combination treatments pairing MSCs with biologics, such as Infliximab, are showing longer remission periods compared to traditional therapies alone.
Stem cell banking is also becoming increasingly important. Services like Americord Registry allow families to store cord blood, cord tissue, placental tissue, and exosomes at birth. This ensures access to young, potent stem cells for potential future treatments. Since umbilical cord tissue-derived MSCs must be collected at birth, banking these cells now could be crucial for addressing conditions like Crohn's disease as these therapies continue to evolve into standard care options.
FAQs
Am I a good candidate for MSC therapy?
Your eligibility for mesenchymal stem cell (MSC) therapy for Crohn's disease depends on several factors, including how severe your condition is and how you've responded to previous treatments. Research indicates that MSC therapy may help achieve remission in cases that don't respond to standard treatments and has shown a good safety record over the long term. To find out if MSC therapy is right for you, it’s essential to consult with a healthcare professional. Additionally, Americord Registry offers stem cell banking services, supporting various regenerative therapies.
How long can remission last after MSC treatment?
Remission following mesenchymal stem cell (MSC) treatment has been observed to last up to 3 years or even longer. Research highlights cases where remission was maintained for 3 years, with some follow-up reports showing sustained remission extending as far as 7 years. Current studies continue to support the long-term safety and effectiveness of this treatment approach.
Should I bank my baby’s cord tissue for future Crohn’s treatments?
Banking your baby’s cord tissue could open doors to future treatments, including those for conditions like Crohn’s disease. Mesenchymal stem cells (MSCs) from cord tissue have shown encouraging results in terms of safety and effectiveness, particularly in addressing complications such as perianal fistulas. By preserving cord tissue with a reliable service like Americord Registry, you secure access to these cells, potentially paving the way for advanced regenerative therapies as scientific research continues to evolve.
The views, statements, and pricing expressed are deemed reliable as of the published date. Articles may not reflect current pricing, offerings, or recent innovations.




