Umbilical Cord Blood vs. Bone Marrow for Tendon Repair
Tendon injuries are tough to heal because tendons have low blood supply and often form weak scar tissue instead of regenerating fully. This has led researchers to explore the use of mesenchymal stem cells (MSCs) for better healing. Two main sources of MSCs are umbilical cord blood (UCB-MSCs) and bone marrow (BM-MSCs).
Here’s the key takeaway: UCB-MSCs outperform BM-MSCs in tendon repair. They are easier to collect (no surgery needed), grow faster, and maintain their regenerative abilities regardless of age. BM-MSCs, while effective, decline in quality as donors age and carry risks like ectopic bone formation.
Highlights:
- UCB-MSCs: Collected at birth, maintain "youthful" properties, higher tendon-related protein production, and faster recovery potential.
- BM-MSCs: Require surgical extraction, decline with age, and have a higher risk of complications like abnormal bone formation.
Quick Comparison:
| Feature | UCB-MSCs | BM-MSCs |
|---|---|---|
| Collection Method | Non-surgical, at birth | Surgical aspiration |
| Cell Quality | Maintained regardless of age | Declines with donor age |
| Healing Matrix | Well-organized structure | Disorganized, weaker structure |
| Risk of Bone Formation | Low | Higher |
| Availability | Immediate if banked | Requires weeks of preparation |
Bottom line: UCB-MSCs offer a more efficient and safer option for tendon repair, especially when banked at birth for future use. Understanding the benefits of cord blood banking is essential for families considering these regenerative options. BM-MSCs, while helpful, come with more limitations and risks.
Umbilical Cord Blood MSCs: Properties and Advantages
Umbilical cord blood-derived MSCs stand out for their regenerative potential, largely due to their youthful nature. Since these cells are collected at birth, they remain untouched by environmental factors, diseases, or the aging process that typically impacts adult cells. This gives them a distinct edge in both flexibility and growth capacity. UCB-MSCs show higher proliferation rates and greater viability compared to MSCs derived from bone marrow, enabling faster expansion and extended usability. Their biological youth becomes a foundation for exceptional regenerative capabilities.
Early-Stage Cells with High Growth Potential
The neonatal stage of UCB-MSCs directly contributes to their effectiveness in regenerative medicine. These cells excel in matrix synthesis and can be expanded more efficiently in lab settings. Unlike bone marrow-derived MSCs, which lose potency as they age, UCB-MSCs maintain their neonatal traits. This makes them a valuable resource, especially for families who choose to bank stem cells through services like Americord Registry, which specializes in preserving cord blood, cord tissue, and placental tissue for potential future use. These early-stage cells also play a critical role in promoting structured tendon matrix formation.
Tendon Cell Development and Matrix Formation
UCB-MSCs are particularly adept at facilitating tendon repair, thanks to their ability to generate essential signals like scleraxis and tenascin-C. Research highlights that these cells exhibit the highest median expression of scleraxis, a transcription factor crucial for tendon development, compared to MSCs from other sources like bone marrow or adipose tissue. Additionally, they produce elevated levels of tenascin-C, a glycoprotein that helps organize collagen fibers into the structured matrix essential for tendon strength.
"UC-MSC is superior to other MSCs in differentiating into tendon-like lineage cells and forming a well-organized tendon-like matrix." - PubMed
UCB-MSCs also differentiate into spindle-shaped tenocytes, which are specialized tendon cells with long cytoplasmic extensions - a characteristic feature of functional tendon cells. They produce large amounts of Type I collagen, the primary component of healthy tendon tissue, along with Type III collagen and decorin, a proteoglycan that regulates collagen fibril size. This structured matrix is vital for restoring tendon strength and preventing re-injury by improving mechanical load capacity.
Non-Invasive Collection Process
One of the standout benefits of UCB-MSCs is the ease of their collection. Umbilical cord blood is collected through a non-invasive process that poses no risk to the mother or newborn. This eliminates the donor site morbidity associated with bone marrow extraction, which requires invasive surgery and comes with pain and recovery time.
"The harvesting procedure, including bone marrow aspiration, is invasive. For that reason, allogeneic human umbilical cord blood-derived MSCs (hUCB-MSCs) are being used... due to the simplicity of a single procedure." - Knee Surgery & Related Research
This one-time collection opportunity at birth makes stem cell banking a practical choice for families looking to prepare for future regenerative treatments. Once preserved, these cells can be stored for long-term use, offering a ready resource for potential therapies without the need for further invasive procedures. Additionally, UCB-MSCs have an immune-privileged status, meaning they are less likely to provoke an immune response, which opens the door for potential use in family members beyond the original donor.
Bone Marrow MSCs: Properties and Limitations
Bone marrow-derived MSCs (BM-MSCs) are a popular focus in tendon repair research. These cells can transform into tenocytes and release growth factors like TGF-β, VEGF, and IGF-1, which play key roles in reducing inflammation and promoting healing. BM-MSCs also produce Type I collagen, crucial for strength, and Type III collagen, important for early stabilization - both vital for tendon recovery. In a clinical study involving 10 patients with Achilles tendinopathy, autologous BM-MSC injections showed an 80% success rate at 24 weeks, with marked improvements in pain and functionality. However, despite their regenerative abilities, BM-MSCs face challenges that can impact their overall effectiveness in tendon repair.
High Cell Numbers but Age-Related Decline
While BM-MSCs initially exist in large numbers, their quality decreases with age. Studies reveal a 70% reduction in tendon-related stem cells in 24-month-old rats compared to 3-month-old ones. This decline affects the cells' ability to differentiate into multiple lineages, reducing their effectiveness for tendon repair in older individuals. Additionally, their potency diminishes during in vitro cell expansion, narrowing their therapeutic potential.
"BM-MSCs suffer from aging and loss of multilineage differentiation potential with in vitro cell passaging." - Pauline Po Yee Lui, Hospital Authority, Hong Kong
Surgical Extraction Requirements
Harvesting BM-MSCs involves surgical aspiration, typically drawing 8 mL of bone marrow from the posterior superior iliac crest under local anesthesia. This procedure carries risks, including donor site pain, infection, and the dilution of cells due to peripheral blood mixing. In a first-in-human trial for Achilles tendinopathy, 3 out of 15 patients (20%) had to be excluded because their MSCs failed to proliferate adequately during the 28 to 43-day culture expansion required before treatment. These challenges, combined with the cells' inherent limitations, can affect long-term tendon repair outcomes.
Tendon Repair Results and Limitations
BM-MSCs are known to enhance collagen production and speed up recovery, but they come with drawbacks. Unlike MSCs from umbilical cord blood (UCB), BM-MSCs carry the risk of ectopic bone formation - the unexpected development of bone-like tissue within the tendon. Inflammatory cytokines like IL-1β, present in injured areas, can cause BM-MSCs to differentiate into osteoblasts (bone cells) instead of tenocytes, leading to abnormal ossification.
"The transplantation of BMSCs into the rabbit tendon defect was reported to form ectopic bone in some tendon samples." - Pauline Po Yee Lui, Hospital Authority, Hong Kong
Animal studies have also shown that BM-MSCs produce larger glycosaminoglycan-rich areas compared to umbilical cord-derived MSCs. This results in a disorganized matrix that weakens the tissue's mechanical properties, making it less capable of handling physical stress effectively.
Side-by-Side Comparison: UCB-MSCs vs. BM-MSCs for Tendon Repair
Examining UCB-MSCs (umbilical cord blood-derived MSCs) alongside BM-MSCs (bone marrow-derived MSCs) sheds light on their respective roles and benefits in tendon repair.
Regeneration Performance
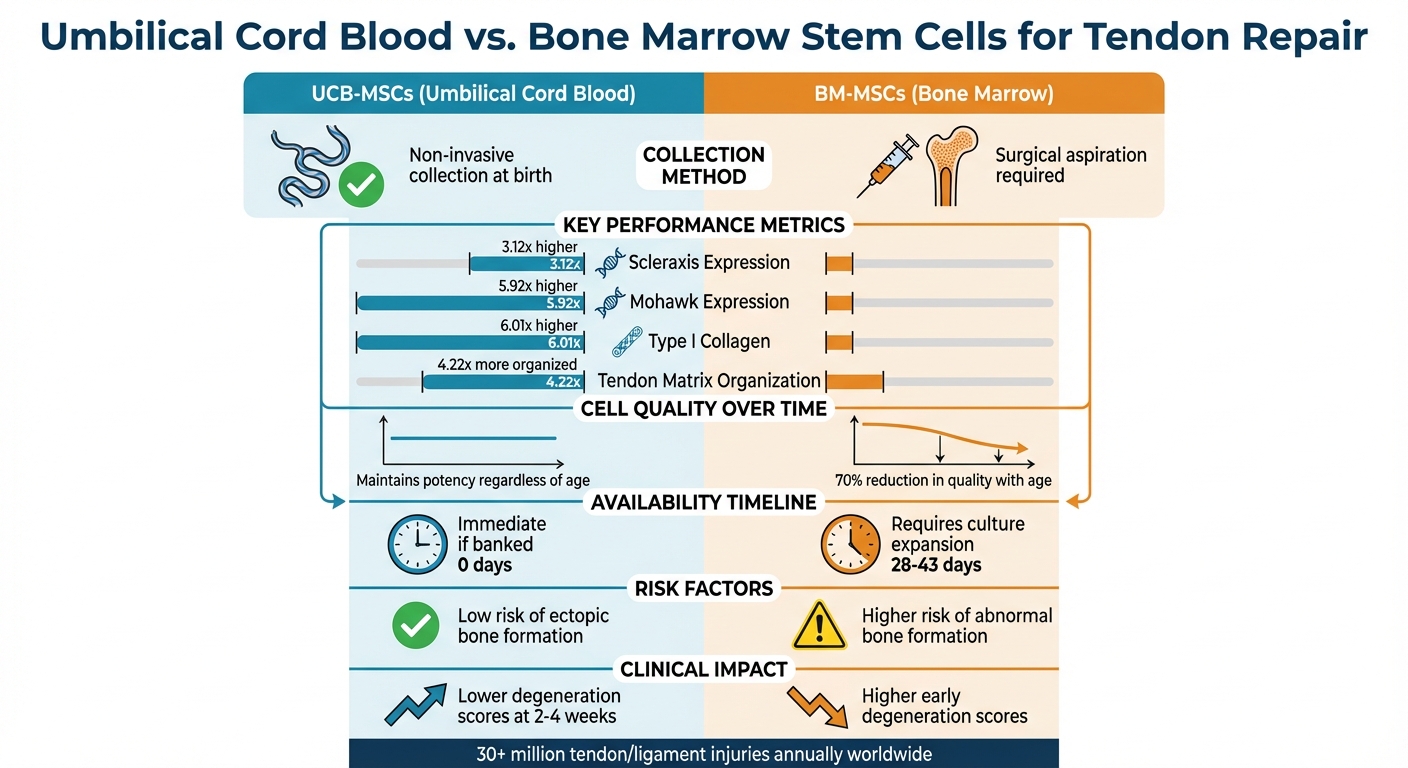
UCB-MSCs demonstrate significantly stronger tenogenic marker expression compared to BM-MSCs. Studies highlight the upregulation of key markers in UCB-MSCs: scleraxis (3.12-fold), mohawk (5.92-fold), type I collagen (6.01-fold), and tenascin-C (1.61-fold) relative to BM-MSCs. These markers are essential for building durable and functional tendon tissue.
Additionally, UCB-MSCs produce a matrix that is 4.22 times more organized than that of BM-MSCs. In contrast, BM-MSCs are more likely to form glycosaminoglycan (GAG)-rich areas, which result in a disorganized matrix with reduced mechanical strength.
"UC-MSC is superior to other MSCs in differentiating into tendon-like lineage cells and forming a well-organized tendon-like matrix under T-3D conditions." - PubMed
These findings suggest that UCB-MSCs may lead to quicker recovery and better long-term results, as explored further below.
Recovery Speed and Long-Term Results
Thanks to their regenerative advantages, UCB-MSCs promote faster and higher-quality healing in the early stages of recovery. Animal studies show lower degeneration scores at 2 and 4 weeks when UCB-MSCs are used, indicating superior early-phase healing. Another advantage is availability - UCB-MSCs can be used immediately if banked, whereas BM-MSCs require 28 to 43 days of culture expansion after extraction.
Over the long term, UCB-MSCs maintain their regenerative capacity for up to 24 months, regardless of the patient’s age. In contrast, BM-MSCs lose effectiveness as donor age increases. Clinical outcomes show that UCB-MSCs provide comparable pain relief and functional improvements, even in older patients or those with larger tendon injuries. The "youthful" properties of UCB-MSCs directly contribute to these advantages, supporting both faster recovery and sustained benefits.
Comparison Table
| Feature | UCB-MSCs | BM-MSCs |
|---|---|---|
| Collection Method | Non-invasive; collected at birth | Invasive surgical aspiration from bone |
| Cell Proliferation | Higher; "young" cells unaffected by donor age | Declines with patient age |
| Scleraxis Expression | 3.12-fold higher | Baseline level |
| Tendon Matrix Formation | 4.22-fold higher; well-organized structure | Lower; prone to GAG-rich, disorganized areas |
| Ectopic Bone Risk | Not typically reported | Risk of bone formation in tendon |
| Early Degeneration (2-4 weeks) | Lower degeneration scores | Higher degeneration scores |
| Availability Timeline | Immediate if banked | Requires 28-43 days for culture expansion |
| Donor Site Morbidity | None | Pain and surgical complications |
Medical Applications and Stem Cell Banking
How Stem Cell Banking Supports Regenerative Medicine
Stem cell banking offers a way to preserve young, healthy cells that can be used for regenerative therapies. By storing cord blood and tissue at birth, families secure cells that haven’t been exposed to aging or environmental damage. These cells are stored in liquid nitrogen at ultra-low temperatures, around -195 °C, preserving their ability to multiply and remain effective for decades.
For injuries like tendon and ligament tears - affecting over 30 million people annually - banked stem cells provide an alternative to procedures like bone marrow extraction. Years down the road, if someone experiences a rotator cuff or Achilles tendon injury, their stored cells are immediately available for treatment. Collecting cord blood is quick, painless, and done right after delivery, avoiding the risks and discomfort of bone marrow aspiration. Since these cells are a perfect genetic match for the child and often a partial match for siblings, the likelihood of rejection in treatments is significantly reduced. This makes stem cell banking a valuable tool for regenerative medicine, especially for orthopedic injuries.
Americord Registry's Preservation Services
Americord Registry offers advanced preservation services to ensure families can access these benefits. Their services include banking cord blood, cord tissue, and placental tissue, as well as exosomes, all processed with CryoMaxx™ technology and stored in 5-compartment vials. This system allows for targeted use of specific portions of the sample without needing to thaw everything at once, making it possible to use the cells for multiple treatments over time.
Cord tissue banking is particularly important for orthopedic treatments, as it contains mesenchymal stem cells (MSCs) and growth factors that aid in tissue repair and reduce inflammation. Americord Registry’s AABB accreditation ensures their storage processes meet strict quality standards, providing peace of mind for families planning for future medical needs.
Conclusion
When comparing umbilical cord blood-derived stem cells (UCB-MSCs) to bone marrow-derived stem cells for tendon repair, UCB-MSCs show stronger regenerative abilities. Research highlights that cord blood cells produce a tendon-like matrix 4.22 times more effectively and significantly increase the expression of key tendon markers, scleraxis (3.12 times) and mohawk (5.92 times). These factors play a critical role in tendon healing and recovery.
Beyond their biological edge, UCB-MSCs come with practical advantages. Cord blood collection is a non-invasive process performed at birth, unlike the more invasive bone marrow extraction. Additionally, while the quality of bone marrow stem cells diminishes with age, cord blood cells maintain their youthful potency when banked. Bone marrow-derived cells also carry a higher risk of complications, such as ectopic bone formation, which is largely avoided with UCB-MSCs. These factors make public vs private cord blood banking a forward-thinking choice for future treatments.
For families planning ahead, storing stem cells at birth ensures access to high-quality, ready-to-use cells for potential tendon repair. With over 30 million tendon and ligament injuries reported globally each year - 30% to 50% of which involve tendons - having banked stem cells available can significantly impact treatment outcomes. This eliminates the need for invasive procedures later in life and ensures timely intervention.
Americord Registry offers advanced preservation services to store these powerful cells for future use. Their CryoMaxx™ processing and 5-compartment vial system allow for flexible, long-term storage of cord blood, cord tissue, and placental tissue. Whether dealing with an Achilles tendon injury or a rotator cuff tear, having these preserved cells on hand provides both medical and logistical advantages that bone marrow-derived cells cannot match.
FAQs
Are UCB-MSC tendon treatments available in the U.S. today?
Yes, UCB-MSC tendon treatments are available in the U.S. and have shown promise in aiding tendon repair while helping to reduce inflammation. They are especially useful for addressing early to moderate injuries, providing a regenerative option for recovery.
Can banked cord blood cells be used for siblings or parents?
Cord blood cells stored in a bank can often be used not just for the baby they came from but also for siblings or even parents. Since these cells have a good chance of being a genetic match, they can be incredibly useful for stem cell treatments within the family. This makes cord blood an important resource for addressing potential health needs and advancing regenerative treatments.
What are the main safety risks of BM-MSCs in tendon repair?
The use of bone marrow-derived mesenchymal stem cells (BM-MSCs) for tendon repair is considered to carry minimal safety risks. Research indicates these cells are generally safe in clinical applications, with rare complications such as infection, immune responses, or unintended tissue growth. Strict regulatory guidelines are in place to reduce risks associated with contamination or improper handling. Although potential long-term concerns, like abnormal cell growth, remain hypothetical, existing evidence supports BM-MSCs as a reliable and safe treatment option when used under appropriate medical supervision.
The views, statements, and pricing expressed are deemed reliable as of the published date. Articles may not reflect current pricing, offerings, or recent innovations.