Umbilical Cord Stem Cells for Joint Degeneration
Umbilical cord stem cells (hUC-MSCs) are emerging as a promising option for treating joint degeneration, particularly osteoarthritis. These cells, derived from neonatal umbilical cords, can repair cartilage damage, reduce inflammation, and improve joint function. Unlike traditional treatments that only address symptoms, hUC-MSCs target the root cause by regenerating cartilage and suppressing inflammatory markers.
Key Takeaways:
- Source: hUC-MSCs come from umbilical cords, which are collected safely and ethically after childbirth.
- Benefits: They reduce pain, repair cartilage, and suppress inflammation without the need for invasive procedures.
- Clinical Evidence: Studies show significant improvements in pain scores and joint function, with outcomes lasting up to seven years.
- Advantages: Faster cell growth, lower risk of immune rejection, and non-invasive collection compared to other stem cell sources.
These cells offer a forward-looking solution for joint health, with ongoing research exploring their full potential in regenerative medicine.
Properties of Human Umbilical Cord Mesenchymal Stem Cells (hUC-MSCs)
Cell Growth and Differentiation Capabilities
hUC-MSCs are highly efficient when it comes to multiplying and transforming into the specific cell types needed for joint repair. Their proliferation rate stands out - when grown in a bioreactor, they multiply over 1,000 times faster than bone marrow stem cells. Even after extensive lab expansion, they maintain their ability to self-renew, ensuring enough cells can be produced for therapeutic purposes without losing their effectiveness.
These cells don't just transform into new cartilage cells - they also play a critical role in coordinating the repair process. They release trophic factors and extracellular vesicles that encourage existing joint cells to migrate and multiply at the injury site. hUC-MSCs also have anti-apoptotic properties, meaning they help prevent cell death. They achieve this by increasing protective proteins like Bcl-2 and reducing harmful markers such as Bax and Bad, effectively protecting chondrocytes. Additionally, they enhance the expression of key cartilage-forming genes, including SOX9, COL2 (Type II Collagen), and aggrecan. Beyond rebuilding cartilage, hUC-MSCs actively counteract the inflammatory conditions that often hinder joint repair.
Reducing Inflammation and Modulating Immune Response
Chronic inflammation is a major obstacle in joint degeneration, and hUC-MSCs are equipped to tackle this issue. They lower levels of inflammatory markers like IL-1β, TNF-α, and IL-6, while boosting anti-inflammatory agents such as IL-10 and TGF-β1. They also shift macrophages from the pro-inflammatory M1 state to the tissue-healing M2 state, creating a more favorable environment for recovery.
In August 2023, a study published in Nature Scientific Reports demonstrated the impact of hUC-MSCs in a rat model of osteoarthritis. Researchers administered three weekly injections of 2 × 10^6 hUC-MSCs. The results were promising: joint fluid showed reduced levels of IL-1β and TNF-α, decreased chondrocyte death, and overall improved joint conditions. The study concluded:
"UCMSC transplantation appears to improve joint pathology, reduce inflammatory factors, and decrease chondrocyte apoptosis, likely through the involvement of IL-10 and TGF-β1."
hUC-MSCs also modulate immune activity by suppressing T lymphocytes, B lymphocytes, dendritic cells, and natural killer cells. They protect cartilage by reducing matrix-degrading enzymes like ADAMTS-5, which are responsible for breaking down cartilage. By eliminating synovial inflammation, these cells help reduce pain caused by nociceptive hypersensitivity, making them a powerful tool for managing osteoarthritis. Importantly, these benefits are paired with the ethical ease of collecting hUC-MSCs.
Ethical Collection Methods
The collection process for hUC-MSCs is straightforward and entirely safe for both the mother and newborn. It begins after the umbilical cord has been clamped and cut. There are two collection methods: "in utero", which occurs while the placenta is still in the uterus during the third stage of labor, and "ex utero", which happens after the placenta has been delivered. On average, about 110 ml of blood can be harvested from a single placenta by experienced collectors.
Ethical guidelines ensure that informed consent is obtained before labor begins whenever possible, allowing mothers to make decisions without the influence of pain or medication. Unlike bone marrow collection, which involves an invasive procedure, newborn stem cell collection is completely non-invasive and painless. This simplicity, combined with the high yield of viable cells, supports the growing use of hUC-MSCs in regenerative treatments for joint degeneration.
Clinical Research on hUC-MSCs for Joint Degeneration
Results from Knee Osteoarthritis Studies
Clinical trials have shown promising outcomes for knee osteoarthritis patients treated with hUC-MSCs. Between April 2019 and May 2021, Gangnam Severance Hospital studied 54 patients (60 knees total), comparing allogeneic hUCB-MSC implantation to traditional microdrilling. The hUCB-MSC group was treated with Cartistem®, a mixture containing 0.5 × 10⁷/ml hUCB-MSCs and sodium hyaluronate. After 24 months, patients in this group reported a VAS pain score of 15.21, significantly lower than the 28.57 reported for the microdrilling group. Their IKDC functional score also improved, reaching 58.54 compared to 50.29 in the microdrilling group.
Another multicenter randomized study followed 144 patients with significant cartilage defects over five years. Those treated with hUCB-MSC-HA implantation showed better arthroscopic cartilage grading and greater pain relief than patients undergoing microfracture surgery. Unlike microfracture, which often produces lower-quality fibrous cartilage, hUC-MSCs were able to regenerate cartilage similar to native tissue.
Patient Results and Safety Data
Long-term follow-ups have reinforced these results. In one study tracking nine patients over seven years, histology confirmed hyaline cartilage formation within the first year. MRI scans at three years revealed intact regenerated cartilage, while arthroscopic evaluations using the ICRS CRA scale showed that hUCB-MSC treatment achieved a score of 9.41, outperforming the 7.94 score from microdrilling. Importantly, no bone changes or severe adverse events were reported over the seven-year period.
Safety data from multiple trials highlight that hUC-MSCs are generally well-tolerated. Temporary injection site pain, resolving within 24 hours, is the most common side effect. A study by Matas and colleagues, involving 18 patients who received repeated injections over a year, demonstrated excellent safety outcomes alongside sustained improvements in pain management.
Current Trials and Future Research
Building on these successes, ongoing studies are exploring new uses and refinements for hUC-MSC therapy. Subgroup analyses suggest that hUCB-MSC implantation is particularly effective for restoring cartilage in anterior lesions of the medial femoral condyle. This approach appears especially beneficial for older patients and those with extensive cartilage damage.
"HUC-MSCs possess the ability to alleviate pain, enhance knee joint function, and potentially postpone the need for surgical intervention in both non-surgical and other cases, making them highly deserving of clinical promotion and application."
– Journal of Orthopaedic Surgery and Research
Looking ahead, research is delving into next-generation therapies such as hUC-MSC-derived extracellular vesicles (EVs). These cell-free alternatives could offer similar anti-inflammatory and regenerative benefits while being easier to store and less likely to trigger immune responses. Evidence suggests that cartilage regenerated through hUC-MSC treatment can last between three and seven years, offering patients long-term relief and a better quality of life.
Benefits of Umbilical Cord-Derived Stem Cells Compared to Other Sources
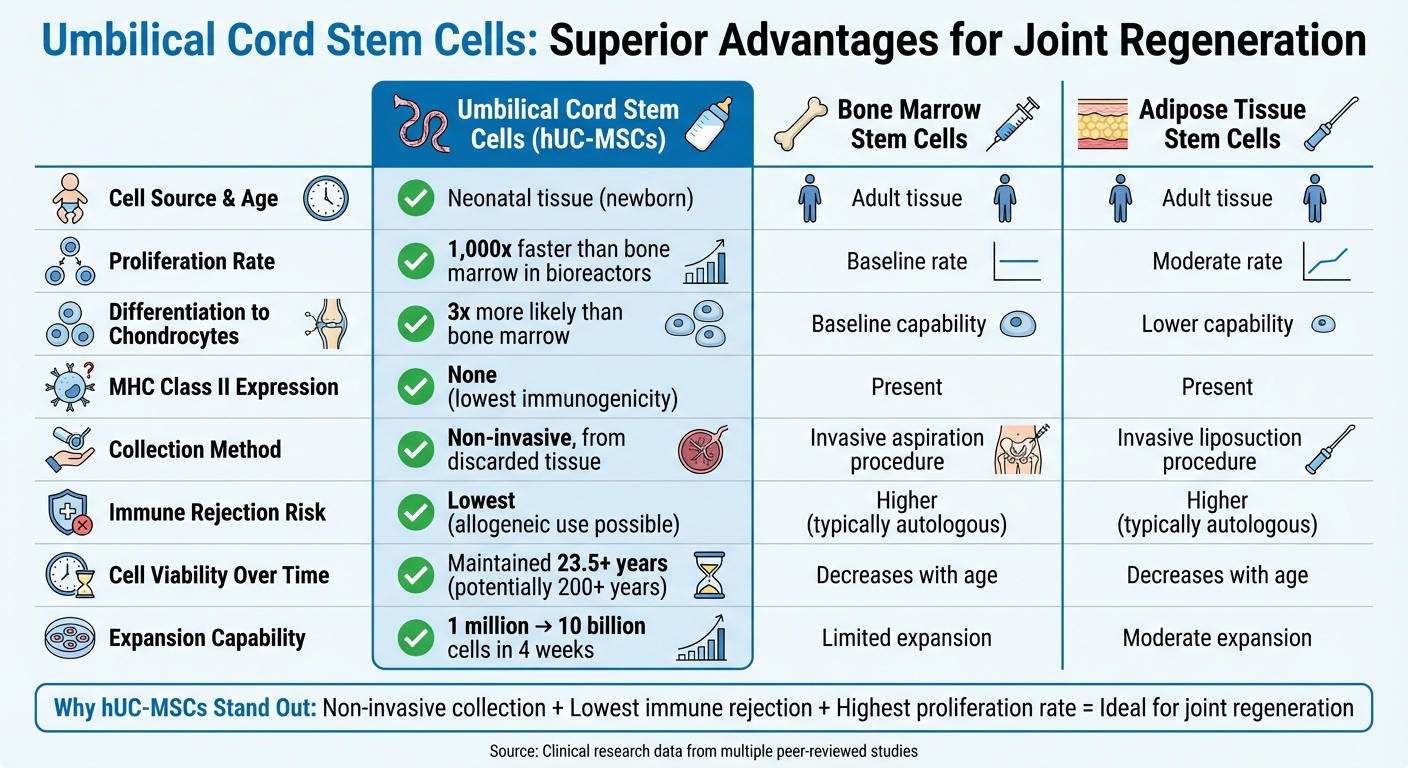
Umbilical Cord Stem Cells vs Other Stem Cell Sources Comparison
Greater Cell Quantity and Viability
Stem cells derived from umbilical cords, sourced from neonatal tissue, stand out for their youth and vitality compared to those obtained from adult bone marrow or adipose tissue. As we age, stem cells in bone marrow and fat naturally diminish in both quantity and quality. However, neonatal cells remain robust and highly viable.
When cultured in specialized bioreactors, umbilical cord stem cells can proliferate at rates over 1,000 times higher than bone marrow stem cells. This remarkable expansion capability ensures that enough cells are available for treatments, eliminating the need for repeated harvesting procedures.
Additionally, these cells exhibit exceptional differentiation potential. Studies reveal that hUC-MSCs are three times more likely than bone marrow stem cells to transform into chondrocytes, the cells responsible for producing collagen. This makes them especially effective for addressing joint degeneration and cartilage damage. Their ability to multiply rapidly pairs well with their low likelihood of triggering immune responses, a point explored further below.
Reduced Risk of Immune Rejection
Umbilical cord stem cells have a distinct advantage due to their immune-privileged status. They express low levels of MHC class I molecules and lack MHC class II and costimulatory molecules (such as CD40, CD80, and CD86), which are typically responsible for activating T-cells. This unique feature allows them to be used as allogeneic, off-the-shelf treatments, significantly lowering the risk of immune rejection that is common with organ transplants.
Key immunological benefits include:
- Low Immunogenicity: Umbilical cord stem cells have the lowest immune activation potential among stem cell sources.
- No MHC Class II Expression: Unlike bone marrow and adipose-derived cells, hUC-MSCs do not express these molecules.
- Allogeneic Application: They can be used directly from donors without requiring the patient’s own cells, unlike most other sources.
This low risk of rejection enables single-stage surgical procedures, as the cells can be prepared in advance without the need for separate harvesting. Patients also avoid invasive procedures like bone marrow aspiration or liposuction, reducing both surgical risks and donor site complications. Furthermore, hUC-MSCs actively suppress inflammatory cells and encourage M2 macrophage polarization, which helps reduce inflammation and promotes cartilage repair. These combined attributes make them ideal for joint repair and regeneration.
Simple Collection and Storage Process
The collection process for umbilical cord stem cells is simple and non-invasive. These cells are obtained from umbilical cords, which are typically discarded as medical waste after birth. This eliminates the need for painful harvesting procedures and makes the process more patient-friendly.
Once collected, the cells can be cryopreserved in liquid nitrogen for long-term storage. When needed, they can be thawed and used without compromising their viability or effectiveness. This capability has led to the development of clinical resource banks, enabling families to store their newborn’s stem cells for potential future medical needs.
"Wharton's jelly mesenchymal stem cells (WJ-MSCs) have become the first choice for cartilage regeneration engineering owing to their availability and convenience of collection."
– Hanguang Liang, Researcher
From a clinical standpoint, umbilical cord stem cells offer unmatched convenience. They can be prepared as standardized, quality-controlled products, ensuring greater consistency than autologous sources. Patients are spared the need for preliminary harvesting and expansion procedures, making treatments more efficient and readily accessible.
Why Newborn Stem Cell Banking Matters for Future Treatments
Storing Stem Cells for Future Medical Use
Newborn stem cell banking preserves the therapeutic potential of umbilical cord stem cells for future use. These cells, unlike adult stem cells, retain their quality and vitality because they haven't been exposed to environmental toxins or chronic illnesses over time. Stored in liquid nitrogen, these cells remain viable for over 23.5 years and could potentially last for more than 200 years.
What's impressive is their ability to expand. A culture of 1 million human umbilical cord mesenchymal stem cells (hUC-MSCs) can grow to 10 billion cells in just four weeks. This rapid growth means a single collection at birth could provide enough cells for multiple treatments throughout a lifetime. With conditions like osteoarthritis affecting at least 10% of people over 60 worldwide, banking these cells acts as a form of biological insurance, offering a proactive way to prepare for common age-related health challenges.
Americord Registry's Stem Cell Banking Services
Americord Registry specializes in preserving newborn stem cells for future regenerative medicine. Their cord tissue banking focuses on mesenchymal stem cells, which are instrumental in treating joint degeneration. By storing these cells, families ensure they have access to cutting-edge therapies for conditions like joint damage and osteoarthritis.
Americord's CryoMaxx™ Processing uses minimal manipulation techniques to preserve the quality of the stored tissues. Their services include options for banking cord blood, cord tissue, placental tissue, and even newborn exosomes. With an industry-leading $110,000 engraftment guarantee and AABB accreditation, Americord ensures top-notch quality and safety. They also prioritize transparent pricing and personalized support, making the process seamless for families.
Long-Term Health Benefits of Stem Cell Banking for Families
The benefits of stem cell banking extend beyond the donor, offering a genetic match for close family members. This is particularly significant given that 1 in 3 Americans is expected to benefit from regenerative medicine at some point in their lives.
Currently, there are over 80 FDA-approved treatments and 300 clinical trials exploring the potential of newborn stem cells. For example, studies on Wharton's Jelly stem cell therapy have shown a 50% reduction in knee osteoarthritis pain, with MRI scans confirming cartilage regeneration lasting at least seven years. By banking these cells at birth, families can access these therapies without the need for invasive procedures later.
"When you bank your baby's stem cells and perinatal tissues, you are giving them a key to unlocking and accessing new treatment options, for their entire life." – Americord Registry
Conclusion
Umbilical cord-derived stem cells (hUC-MSCs) are transforming the treatment of joint degeneration by addressing the root causes - regenerating cartilage and reducing inflammation. With over 60,000 treatments in the U.S. and 100,000 in Europe, their safety and effectiveness are well-established.
These cells offer compelling biological advantages. They expand quickly, possess strong regenerative capabilities, and have low immunogenicity, which reduces the likelihood of rejection. Plus, they are collected from discarded umbilical cord tissue, eliminating the need for invasive procedures.
Clinical studies back up these benefits:
"HUC-MSCs possess the ability to alleviate pain, enhance knee joint function, and potentially postpone the need for surgical intervention... making them highly deserving of clinical promotion and application." - Journal of Orthopaedic Surgery and Research
For families, banking cord blood, tissue, and placenta at birth offers a unique opportunity to safeguard future joint health. With osteoarthritis affecting at least 10% of people over 60 worldwide, preserving these cells ensures access to advanced treatments without the need for invasive procedures later in life. Additionally, banking these cells provides long-term treatment options for both the donor and genetically matched family members. Companies like Americord Registry make this possible, offering families peace of mind and access to cutting-edge therapies.
As research progresses and more clinical trials confirm the long-term benefits, umbilical cord stem cells are set to play a pivotal role in the future of regenerative medicine for joint health.
FAQs
What makes umbilical cord stem cells a promising option for treating joint degeneration?
Umbilical cord stem cells, especially mesenchymal stem cells derived from cord tissue, are drawing interest as a potential treatment for joint degeneration, including osteoarthritis. These cells stand out due to their impressive ability to multiply, low likelihood of immune rejection, and capacity to aid in cartilage repair. Additionally, they release substances that can reduce inflammation and support tissue healing, making them a promising option for addressing joint damage - possibly even reversing it.
Unlike traditional sources like bone marrow or fat tissue, umbilical cord stem cells offer some distinct perks. Collection is simpler and non-invasive, and research shows encouraging results in cartilage regeneration. Their combination of safety and therapeutic potential is paving the way for advancements in regenerative medicine.
What are the risks or side effects of using umbilical cord stem cells for osteoarthritis treatment?
Using human umbilical cord mesenchymal stem cells (hUC-MSCs) to treat osteoarthritis has shown promise in many studies, with some suggesting better outcomes when paired with therapies like hyaluronate. That said, like any medical treatment, there are risks to keep in mind.
Potential side effects include immune reactions, such as graft-versus-host disease (GVHD), though this is rare and typically associated with donor-derived (allogeneic) stem cells. Another concern is the variability in the quality and handling of stem cell products. Poorly processed cells could lead to reduced effectiveness or unexpected side effects.
It's worth noting that, while research into umbilical cord-derived stem cells is advancing, these treatments are not universally approved by the FDA for osteoarthritis. Patients should consult with experienced healthcare providers to weigh the potential risks and benefits before considering this option.
Can umbilical cord stem cells help family members with joint problems?
Umbilical cord stem cells might provide an option for family members dealing with joint issues. Research indicates that these cells have properties that may aid in cartilage repair and help slow joint deterioration. However, whether they can be used for a specific family member depends on factors like medical assessment and cell compatibility.
It's crucial to consult with a healthcare professional to evaluate if stored umbilical cord stem cells could be a viable treatment for joint-related conditions in your family.
The views, statements, and pricing expressed are deemed reliable as of the published date. Articles may not reflect current pricing, offerings, or recent innovations.