What is Wharton’s Jelly? The Hidden Power of Umbilical Cord Tissue
Wharton’s Jelly is a gel-like tissue in the umbilical cord, rich in mesenchymal stem cells (MSCs). These cells have the ability to transform into various tissue types like bone, cartilage, and muscle, making them a promising resource for regenerative medicine. Unlike adult stem cells, MSCs in Wharton’s Jelly are less mature, divide faster, and are less likely to trigger immune responses, making them suitable for potential treatments.
Preserving cord blood, cord tissue, and placenta tissue at birth allows families to store these cells for future medical use. Clinical research has shown their potential in treating conditions like osteoarthritis, spinal cord injuries, and graft-versus-host disease. With advancements in stem cell therapy, banking umbilical cord tissue provides access to emerging treatments that could address various health challenges.
Americord offers specialized processing and storage services for umbilical cord tissue, ensuring long-term viability and flexibility for future use. Their FDA-regulated and AABB-accredited methods provide peace of mind for parents planning for their child’s health.
Biological Properties of Wharton's Jelly
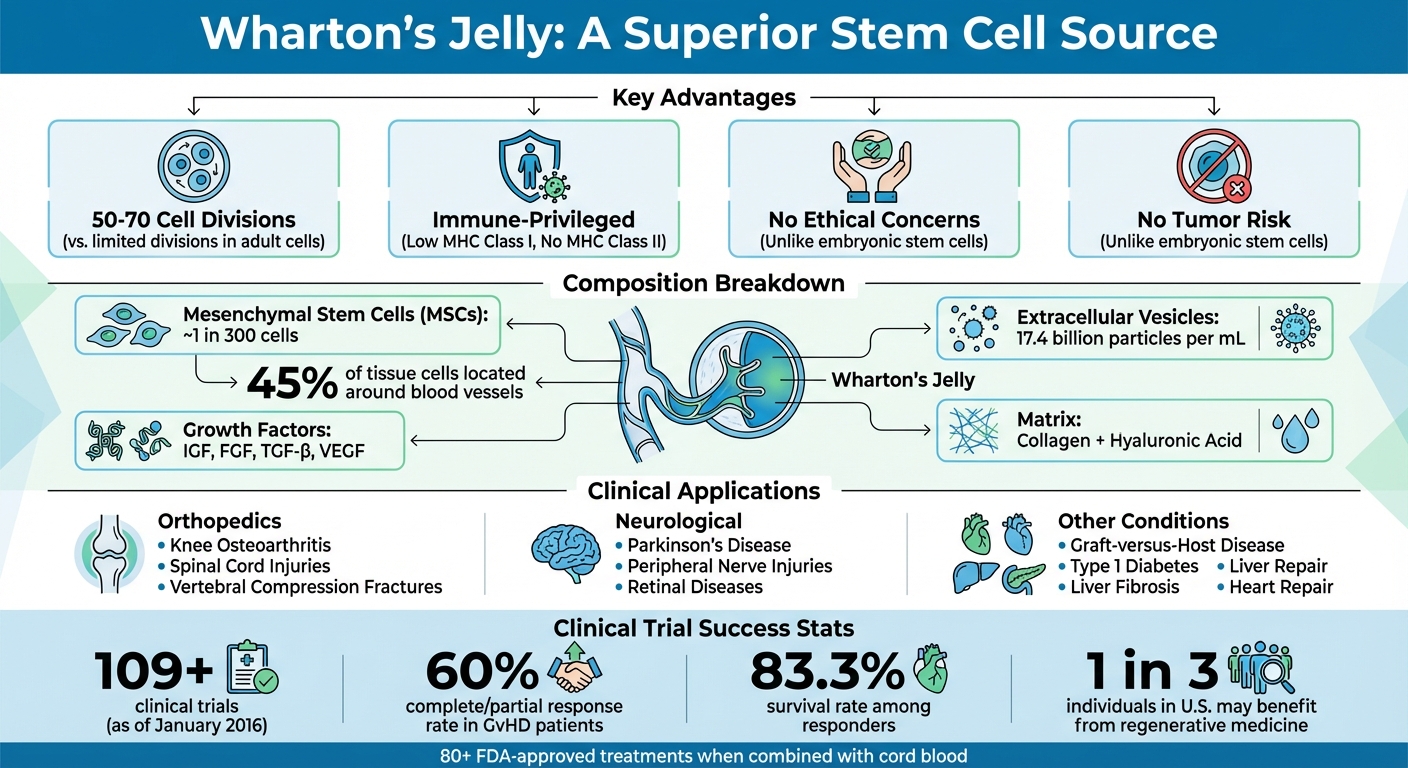
Wharton's Jelly is a fascinating tissue, combining structural elements, specialized cells, and bioactive molecules that play a role in healing and regeneration.
Mesenchymal Stromal Cells (MSCs) in Wharton's Jelly
The standout feature of Wharton's Jelly is its concentration of mesenchymal stromal cells (MSCs), particularly around the umbilical blood vessels. This region contains about 45% of the tissue's cells, with approximately one in every 300 being an MSC - a cell type known for its impressive ability to self-renew.
These WJ-MSCs can transform into various specialized cells, including those forming bone, cartilage, fat, muscle, and even neural-like tissues, highlighting the different roles of stem cells found in birthing tissues. Researchers have found that these cells can be harvested in high numbers, retaining their stability and ability to differentiate when cultured.
The gelatinous matrix of Wharton's Jelly, enriched with collagen and hyaluronic acid, also holds essential growth factors like Insulin Growth Factor (IGF), Fibroblast Growth Factor (FGF), Transforming Growth Factor-beta (TGF-β), and Vascular Endothelial Growth Factor (VEGF). This natural 3D framework not only supports cell attachment but also acts as a reservoir for these critical growth factors.
Anti-Inflammatory and Immune System Effects
One of the remarkable characteristics of WJ-MSCs is their immune-privileged nature. They express low levels of MHC class I molecules, lack MHC class II molecules (like HLA-DR), and do not produce costimulatory molecules such as CD40, CD80, and CD86. This unique profile significantly reduces the likelihood of triggering an immune response after transplantation.
WJ-MSCs actively interact with various immune cells - T-cells, B-cells, Natural Killer (NK) cells, and dendritic cells - to regulate immune responses. They produce high levels of immune-suppressing proteins like HLA-G6, indoleamine-2,3-dioxygenase (IDO), and prostaglandin E2 (PGE2). Additionally, they secrete anti-inflammatory cytokines such as Interleukin-10 (IL-10) and TGF-β.
Clinical evidence highlights these benefits. For instance, in 2011, a 4-year-old boy with severe steroid-resistant Graft-versus-Host Disease (Grade IV) received 3.3 million WJ-MSCs per kilogram of body weight. Within just six days, his bilirubin levels normalized, and his skin rash cleared. After a third infusion of 8.0 million cells per kilogram, all symptoms disappeared, and he remained free of chronic GvHD a year later.
Another study in 2016 by Boruczkowski and colleagues involved 10 patients with steroid-resistant GvHD who received 1 to 3 infusions of WJ-MSCs at a median dose of 1.5 million cells per kilogram. The results were promising: 60% of patients showed a complete or partial response, and 83.3% of those responders survived the follow-up period.
These immune-regulating properties further enhance the ability of WJ-MSCs to promote tissue repair.
Tissue Repair and Regeneration
WJ-MSCs go beyond immune modulation by actively supporting tissue repair through paracrine signaling - the release of growth factors and chemical signals that stimulate the body's natural healing processes.
"The therapeutic potential of MSCs lies not in their ability to directly replace damaged cells, but rather in their capacity to create an environment conducive to healing." - Stem Cell Medical Center
These cells encourage angiogenesis, the formation of new blood vessels, ensuring that injured tissues receive the oxygen and nutrients they need. They also help prevent premature cell death in damaged areas and can migrate to sites of inflammation and injury, thanks to their homing ability.
These biological features make a strong case for preserving umbilical cord tissue. With WJ-MSCs being explored in over 109 clinical trials as of January 2016, their potential for advancing regenerative medicine is immense.
Medical Applications of Wharton's Jelly
Current Uses in Regenerative Medicine
Wharton's Jelly is finding its place in clinical treatments for various challenging medical conditions. In orthopedics, it’s being used to manage knee osteoarthritis through joint injections. For instance, in August 2021, a 27-year-old man with Grade II knee osteoarthritis received a single 2 mL injection of Wharton's Jelly derived from umbilical cord tissue. Under the care of Dr. Ashim Gupta and his team, the patient experienced a 50% reduction in pain and improved joint function within three months.
Spinal cord injuries are another area of exploration. In June 2025, a Phase I clinical trial led by N. Kaplan and colleagues assessed the use of Wharton's Jelly–derived mesenchymal stem cells (WJ-MSCs) in patients with chronic complete spinal cord injuries. Administered through multiple routes, the treatment showed encouraging results after a year. Patients regained sensation and motor function without experiencing serious side effects.
Another example involves osteoporotic vertebral compression fractures. A Phase I/IIa study by Shim and colleagues tested Wharton's Jelly in 20 postmenopausal women aged 50–89. The experimental group received 40 million WJ-MSCs via intramedullary injection on day 0, followed by 200 million cells intravenously on day 7. After 12 months, these women reported significant improvements in pain relief and reduced disability compared to the control group.
These instances highlight the growing potential of Wharton's Jelly in medical research and treatment.
Stem Cell Therapy Research and Clinical Trials
Building on these clinical applications, researchers are delving into other possibilities for Wharton's Jelly. In neurological studies, these cells are being tested for peripheral nerve injuries and neurodegenerative diseases like Parkinson's. Early ophthalmology research in rodent models has also shown promise, with Wharton's Jelly cells helping to rescue photoreceptors and improve vision in retinal diseases.
Work is also underway in metabolic and organ-related conditions. For example, researchers are exploring how Wharton's Jelly cells might aid in beta-cell regeneration for Type 1 diabetes, generate hepatocyte-like cells to address liver fibrosis, and differentiate into cardiomyocytes for heart repair. A 2015 study led by Yan Zhang used Wharton's Jelly cells in a canine model of degenerative disc disease. After injecting 1 million labeled cells into intervertebral discs, researchers observed slower disc height loss and better MRI results over 24 weeks compared to saline-treated controls.
Another promising area involves cell-free therapies using exosomes and extracellular vesicles derived from Wharton's Jelly. These approaches aim to utilize the regenerative properties of Wharton's Jelly without introducing actual cells, potentially simplifying treatment and regulatory processes. Thanks to their immune-privileged status, WJ-MSCs are also being explored for treating graft-versus-host disease through allogeneic transplantation.
Benefits of Preserving Umbilical Cord Tissue
Banking umbilical cord tissue offers a way to preserve mesenchymal stem cells at their peak condition. These cells, sourced from Wharton's Jelly, are ontologically young, meaning they have higher growth potential and age more slowly compared to stem cells derived from adults. They can divide 50–70 times, surpassing the limits seen in most other somatic cells.
Wharton's Jelly also contains a therapeutic mix of growth factors, immunomodulatory cytokines, hyaluronic acid, and extracellular vesicles. This eliminates the need for invasive procedures like bone marrow aspiration and avoids ethical concerns associated with embryonic stem cells.
"Wharton's jelly is a promising and applicable biologic source for orthopedic regenerative application." - Journal of Orthopaedic Surgery and Research
Preserving your baby’s cord tissue offers a proactive step toward potential future medical treatments. The versatility of these cells spans fields such as cardiology, orthopedics, and neurology. With research progressing and clinical trials expanding, cord tissue banking could provide access to personalized treatments that hold immense potential for addressing a wide range of conditions.
Umbilical Cord Tissue Banking with Americord
Americord's Cord Tissue Banking Services
Americord uses a specialized method called CryoMaxx™ Processing to preserve umbilical cord tissue. Unlike methods that isolate only mesenchymal stem cells, this technique keeps the cord tissue as an intact membrane, retaining all multipotent cells, growth factors, and cytokines for potential future use.
The tissue is stored in multiple vials, offering families the flexibility to use the collection for various treatments over time. With manual processing, Americord ensures strict quality control at every step. Once prepared, the tissue is cryopreserved at -196°C in nitrogen-cooled tanks, monitored 24/7 to ensure long-term cell viability. These protocols provide a reliable and future-focused storage solution.
Why Choose Americord
Americord sets itself apart with its advanced processing techniques and strict adherence to regulatory and quality standards. The company is both FDA-regulated and AABB-accredited, meeting stringent requirements for safety and effectiveness in stem cell banking. Additionally, Americord offers a $110,000 Cord Blood Quality Guarantee, the highest in the industry, to cover the cost of obtaining alternative stem cells if the banked cells fail to engraft.
Americord also emphasizes transparent pricing. As of March 2026, the Complete Family Plan costs $251/month for 24 months (totaling $6,034.15), with a one-time $280 fee for the collection kit and shipping. A limited-time promotion offers 15% off multi-service 20-year plans and 30% off multi-service lifetime plans for orders placed between March 2 and March 31, 2026.
"After shopping around, Americord offers the most value and was extremely customer friendly. This was important to me, being a first-time mother." - Tiffany I., Mother of 1
Planning for Your Baby's Future Health
Americord's advanced processing and biobanking services make it easier to plan ahead for your baby’s health. Banking both cord blood and tissue expands potential treatment options. Cord blood contains hematopoietic stem cells, which are currently used in 80+ FDA-approved treatments for conditions like blood and immune disorders. Meanwhile, cord tissue’s mesenchymal stem cells are being studied in clinical trials for conditions such as Alzheimer’s, Parkinson’s, autism, and orthopedic injuries.
Studies suggest that 1 in 3 individuals in the U.S. could benefit from regenerative medicine at some point in their lives. Using mesenchymal stem cells from cord tissue alongside cord blood can improve transplant success rates and lower the likelihood of Graft-versus-Host Disease. With Americord’s multi-vial storage system, families have the option to access these cells for multiple treatments as research progresses and new therapies emerge.
Conclusion
Wharton's Jelly stands out as the richest source of mesenchymal stem cells (MSCs), offering a powerful combination of hyaluronic acid, growth factors, and extracellular vesicles - averaging an impressive 17.4 billion particles per mL. These cells are known for their ability to support tissue repair and immune system modulation. Unlike embryonic stem cells, Wharton's Jelly MSCs provide regenerative benefits without raising ethical concerns or posing tumor risks.
Ongoing research continues to explore the therapeutic possibilities of Wharton's Jelly. Its immunologically naïve cells are especially valuable, as they lower the likelihood of adverse immune reactions during transplantation. This exciting field highlights the importance of preserving these cells at birth.
"Stem cells from umbilical cord blood are available only right after the birth - highly viable and young. In contrast to other cells of the body, they are fresh and not contaminated by diseases or viruses." - Dr. Mario Lehmann, Medical Production Manager
Americord's CryoMaxx™ Processing ensures that umbilical cord tissue is preserved in its most intact and viable state. With FDA approval and AABB accreditation, this advanced method secures these cells for potential future regenerative treatments. By choosing Americord, families gain peace of mind, knowing they’ve safeguarded a powerful resource for emerging therapies.
Banking both cord blood and tissue expands access to regenerative options and serves as a forward-thinking step toward protecting your child’s health in the years to come.
FAQs
Is Wharton’s Jelly usable for family members too?
Wharton's Jelly is prized for its stem cells and their potential in regeneration. Its main uses lie in regenerative medicine and transplantation, often tailored specifically for the person it was stored for, rather than being intended for use by other family members.
When might cord tissue stem cells actually be needed?
Cord tissue stem cells, located in Wharton’s Jelly, hold promise for regenerative treatments targeting conditions such as rheumatoid arthritis, Type 1 diabetes, multiple sclerosis, and spinal injuries. These mesenchymal stem cells (MSCs) have the ability to transform into different cell types, aiding in tissue repair and reducing inflammation. By preserving these cells at birth through cord tissue banking, families ensure they have access to them for potential therapies, many of which are still being explored in research and clinical trials.
How is cord tissue collected at birth?
Cord tissue is collected through a simple and safe process right after birth. After the umbilical cord is clamped and cut, the Wharton’s Jelly, found inside the cord, is carefully extracted. This jelly-like material is a rich source of mesenchymal stem cells and is preserved for potential medical applications. The collection is done promptly after delivery to maintain the tissue's viability for stem cell banking.
The views, statements, and pricing expressed are deemed reliable as of the published date. Articles may not reflect current pricing, offerings, or recent innovations.




