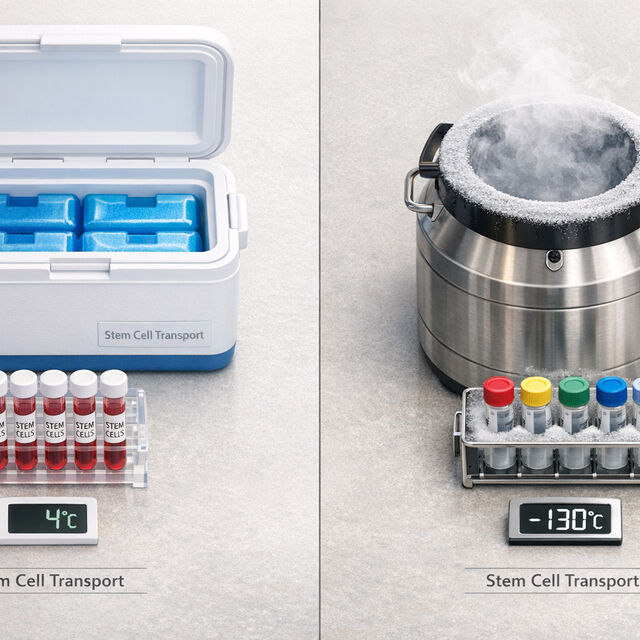
Temperature Guidelines for Stem Cell Transport
Transporting newborn stem cells requires precise temperature control to maintain their viability and prevent damage. Here’s what you need to know:
- Fresh Stem Cells: Must be kept at 4°C (39.2°F) to preserve their viability and avoid clumping. Temperatures above this can reduce cell survival and cause dangerous aggregation.
- Cryopreserved Stem Cells: Require temperatures below −130°C (−202°F), typically maintained with liquid nitrogen vapor. Any warming can damage the cells and lower their effectiveness.
- Risks of Temperature Fluctuations: Higher temperatures can lead to cell death, unwanted differentiation, and aggregation, while warming cryopreserved cells can cause ice crystal formation and osmotic stress.
- Transport Time: Fresh cells have a 72-hour viability window, while cryopreserved cells can be stored for years if kept at ultra-low temperatures.
Proper packaging, insulation, and continuous temperature monitoring are non-negotiable to ensure stem cells remain effective for medical use. Regulatory standards, such as FACT accreditation, mandate strict protocols, including the use of data loggers and validated containers. Whether fresh or frozen, keeping stem cells at the correct temperature is critical for safe transport and successful treatment outcomes.
Why Temperature Control Is Necessary for Stem Cells
Stem cells are incredibly sensitive to temperature changes, making precise control crucial during transport, whether for clinical applications or long-term storage.
How Stem Cells Respond to Temperature Changes
At around 4°C (approximately 39°F), fresh stem cells enter a metabolically inactive state, preserving their integrity in environments with limited nutrients and oxygen. As Na-Hee Yu from the Biomedical Research Institute explains:
"At low temperatures, cells become quiescence which could play a role in increasing cell survival in limited nutrient and oxygen conditions."
When temperatures rise above 4°C, stem cells become metabolically active. At approximately 22°C (72°F), they start secreting extracellular matrix (ECM) components like fibrinogen, collagen I, collagen IV, laminin, and fibronectin. This secretion causes cells to adhere and form aggregates. At body temperature (37°C or 98.6°F), this process accelerates significantly.
For cryopreserved stem cells stored at ultra-low temperatures (below –135°C or approximately –211°F), biological activity is entirely halted. However, any increase in temperature above this threshold can cause ice crystals to form or grow, leading to mechanical damage to cell membranes and internal structures.
Damage Caused by Temperature Changes
Temperature fluctuations can cause severe harm to stem cells during transport. For fresh cells, warming above 4°C increases metabolic activity, quickly depleting the limited nutrients and oxygen in the transport medium. Studies on amniotic fluid stem cells reveal that viability drops from about 85% at 4°C to 62% at 22°C, and falls below 25% at 37°C.
Cell aggregation at higher temperatures introduces additional risks. When clumped cells are injected intravenously, they can adhere to vascular endothelial cells and platelets, obstructing blood flow and potentially causing embolisms in micro-capillaries. Maintaining a single-cell state during transport is critical to avoid these complications and ensure safe vascular injections.
Temperature increases can also prompt unwanted differentiation. The ECM components secreted at warmer temperatures act as differentiation triggers, causing stem cells to transform into other cell types - such as neurons, muscle cells, or epithelial cells. This spontaneous differentiation diminishes their therapeutic value.
Cryopreserved cells face their own challenges. Any warming beyond ultra-low storage temperatures can lead to osmotic stress, increased DMSO toxicity, and mechanical damage. These conditions promote apoptosis and the production of reactive oxygen species. Charles J. Hunt, PhD, highlights the risks:
"Suboptimal cryopreservation has been shown to lead to chromosomal damage and epigenetic changes."
Such damage can result in viability losses of 60–70%, far below the 80% viability threshold needed for clinical success. Maintaining strict temperature control during transport is essential to preserve stem cell viability and their therapeutic potential.
Temperature Requirements for Different Stem Cell Types

Stem Cell Transport Temperature Requirements and Viability Rates
Transporting stem cells requires careful attention to temperature conditions, as different types of stem cells have specific requirements to maintain their integrity. Let’s break down the temperature needs for two main categories: fresh stem cells and cryopreserved stem cells.
Fresh Stem Cells: 4°C (39.2°F)
Fresh stem cells must be transported at 4°C (39.2°F) to remain viable and avoid clumping. While regulatory guidelines allow temperatures between 4°C and 30°C (39.2°F to 86°F), the lower end of this range is crucial for preserving cell quality. For instance, maintaining 4°C ensures cell viability above 80% during transport lasting up to 12 hours.
A study conducted in July 2018 at Kyungpook National University Hospital demonstrated this. Researchers transported 10 million amniotic fluid stem cells at 4°C for 12 hours and found 8.48 million cells still viable - an 84.8% survival rate. In contrast, cells stored at 37°C (98.6°F) during the same period plummeted to just 2.32 million viable cells, a survival rate of only 23%.
Public vs private cord blood banks often differ in their protocols; public banks may extend this timeframe to 48 hours by using insulated shipping kits. Frances Verter, PhD, Founder of Parent's Guide to Cord Blood, highlights the importance of stable temperatures:
"The key to shipping fresh cord blood to the laboratory with maximum cell survival is to keep the temperature stable inside the shipping kit."
Extreme temperatures - comparable to conditions that would harm a child or pet in a parked car - can destroy fresh stem cells. Here’s how temperature impacts cell viability and physical state:
| Temperature | Viability after 12 Hours | Physical State |
|---|---|---|
| 4°C (39.2°F) | >80% | Single-cell suspension (Optimal) |
| 22°C (71.6°F) | ~62% | Moderate aggregation |
| 37°C (98.6°F) | ~23% | Significant aggregation |
For intravenous use, maintaining 4°C is critical. Higher temperatures cause fibrinogen secretion, leading to cell clumping. These clumps can block micro-capillaries and potentially cause embolisms when injected into patients.
Cryopreserved Stem Cells: Below −130°C (−202°F)
Cryopreserved stem cells require ultra-low temperatures to halt all biological activity. They must be stored at below −130°C (−202°F), the glass transition point of aqueous solutions, where metabolic and chemical processes effectively stop. Julie Meneghel and colleagues at Cytiva explain:
"A long-term, practical solution to this difficulty lies in successful cryopreservation that offers secure, stable storage at temperatures below −130°C where metabolic change will not occur."
Most facilities rely on liquid nitrogen vapor storage, maintaining temperatures between −135°C and −196°C (−211°F to −320.8°F). These conditions ensure the cells remain viable for years or even decades. Studies show that bone marrow–derived mesenchymal stem cells stored at −190°C retained 96.0% viability, compared to 81.9% for those stored at −80°C (−112°F). The takeaway? Ultra-low, stable temperatures are non-negotiable for long-term preservation.
While some facilities use −80°C freezers for short-term storage (less than one month), this is not suitable for extended periods. STEMCELL Technologies warns, "cells kept at −80°C will degrade with time and, therefore, it's not recommended for long-term storage." Warming above −130°C can cause osmotic stress, increase DMSO toxicity, and lead to ice crystal formation, all of which activate cell death pathways. This can result in viability losses of 60–70%, far below the 80% threshold required for clinical applications.
Cryopreserved cells are typically transported in dry shippers - specialized containers that maintain liquid nitrogen vapor temperatures without free liquid. These systems are designed to prevent warming events, as even brief temperature fluctuations can lead to chromosomal damage and epigenetic changes, compromising the cells' therapeutic potential.
At Americord Registry, we strictly follow these temperature guidelines to ensure that both fresh and cryopreserved stem cells remain viable and effective during transport.
Packaging and Insulation Methods
Insulated Containers and Cooling Materials
When it comes to transporting stem cells, proper packaging plays a critical role in maintaining their viability. For fresh stem cells, the focus is on using insulation that keeps temperatures stable without relying on active cooling. Public cord blood banks often utilize durable insulated containers that can maintain internal temperatures for up to 48 hours. These containers are designed with high-mass insulation, avoiding the use of ice or gel packs, and keep temperatures within the safe range of 39.2°F to 86°F (4°C to 30°C).
Private banks, on the other hand, tend to use smaller, more portable kits. These kits are paired with expedited medical courier services to minimize risks during transport. Despite their compact size, they’re still built to provide adequate insulation in case of delays or exposure to less-controlled environments.
For cryopreserved stem cells, the requirements are entirely different. These cells need to be kept at ultra-low temperatures, requiring specialized cryogenic shippers like the Arctic Express Transport System. These "dry shippers" use liquid nitrogen vapor to maintain temperatures as low as −320.8°F (−196°C) without the presence of free liquid, which helps prevent any warming events that could damage the cells.
Once the insulation is in place, the containers undergo strict validation processes to ensure their performance under various conditions.
Container Assembly and Validation
Selecting the right insulation materials is only half the battle - ensuring the container performs as expected is equally important. This is where validation testing comes in. Thermo Fisher Scientific advises:
"Ensure that your biological materials are not compromised during transport by testing your shippers with mock material loads in different thermal environments."
Validation involves testing the containers under controlled conditions, often conducted by independent third-party organizations like the International Safe Transit Association (ISTA) or the International Air Transport Association (IATA). This ensures the containers meet global standards rather than relying solely on internal assessments.
Testing must simulate both summer and winter extremes to account for seasonal variations. Additionally, every shipping kit should include continuous temperature monitoring devices, such as data loggers or out-of-range detectors. These devices are not just helpful - they’re mandatory under the 5th edition FACT accreditation standards. They track any temperature deviations during the 48 to 72-hour transport window, providing a secure chain of custody and confirming that the cells remain viable upon delivery. At Americord Registry, we adhere to these rigorous protocols to guarantee the highest standards for stem cell transport and preservation.
Temperature Monitoring and Regulatory Compliance
Temperature Tracking with Data Loggers
Once stem cells are packaged and ready for transport, keeping a close eye on temperature becomes the next crucial step. This is where data loggers come into play. These devices, placed inside shipping containers, record the internal temperature at regular intervals - usually every 15 minutes for fresh stem cell shipments. This creates a detailed thermal history of the journey.
Modern data logging technology goes beyond just temperature. Many sensors also monitor GPS location, shock, and tilt, offering a full picture of the shipment's conditions. Some even use real-time IoT capabilities to send alerts if the temperature starts to drift out of the safe range. These instant notifications allow for quick actions, like adding cooling materials or rerouting, to protect the cells' viability. With the cold chain market for cell and gene therapy expected to grow to $8.06 billion by 2034, at an annual growth rate of 15.64%, these advanced monitoring tools are becoming essential rather than optional.
Beyond preserving cell viability, these measures also ensure compliance with strict regulatory standards.
Regulatory Standards and Documentation Requirements
Temperature monitoring isn’t just a smart precaution - it’s a regulatory necessity. For example, the Foundation for the Accreditation of Cellular Therapy (FACT) requires continuous monitoring under Standard C7.5.2:
"Standard C7.5.2 requires that the temperature inside the outer container be continuously monitored."
The FDA also classifies cord blood processing systems and storage containers as Class II medical devices, which means they’re subject to special controls. For fresh cord blood, temperatures must stay between 39.2°F and 86°F (4°C and 30°C) during transport, and you must have documentation proving these conditions were maintained.
Detailed temperature records are critical for meeting these requirements. Regulatory agencies expect FACT-accredited facilities to keep continuous logs and validation reports in line with Good Distribution Practices (GDP) and Good Manufacturing Practices (GMP). This includes:
- Validation reports confirming container performance
- Standard Operating Procedures (SOPs) outlining temperature protocols
- Logger data from each shipment
At Americord Registry, we maintain these detailed records to ensure every shipment complies with the highest standards. This level of care provides families with peace of mind, knowing their preserved stem cells are safeguarded for future use.
Transport Time and Efficiency Requirements
When it comes to preserving cell viability, timing in transport is just as crucial as temperature control.
72-Hour Viability Window for Fresh Stem Cells
For fresh stem cells, quick transport is non-negotiable. The industry standard allows a 72-hour window from collection to processing or transplantation. Within this timeframe, when stored between 35.6°F and 42.8°F (2°C–6°C), stem cells maintain high viability rates: CD34⁺ cell recovery reaches 92.61%, while total nucleated cell (TNC) recovery is 89.93%.
A study conducted at King Fahad Specialist Hospital (July 2021–February 2022) explored extending storage to 120 hours under controlled conditions. While this extended timeframe resulted in a CD34⁺ recovery of 83.83% and TNC recovery of 76.18%, it also showed a 7.4% greater loss in viability compared to the 72-hour benchmark.
Certain cell types, such as human amniotic fluid stem cells, require even tighter timelines. These cells must be delivered within 12 hours at 39.2°F (4°C) to maintain viability levels above 80%, making rapid shipping absolutely critical.
In contrast, cryopreserved stem cells offer far more flexibility due to their ability to withstand ultra-low temperatures for extended periods.
Extended Transport for Cryopreserved Stem Cells
Cryopreserved stem cells follow a different set of rules. When frozen at temperatures below –238°F (–150°C) - often close to liquid nitrogen levels of –320.8°F (–196°C) - these cells can remain viable for years, even decades. For shorter transport durations, dry ice packaging can maintain temperatures of –94°F (–70°C) for up to 96 hours, giving logistics teams significantly more leeway compared to fresh cell shipments.
However, this advantage comes with its own complexities. Thawing cryopreserved cells leads to an inevitable 35% viability loss. This makes precise temperature control throughout the transport process critical, as any fluctuation could worsen the loss. Dr. David T. Harris from the University of Arizona Health Sciences Biorepository emphasizes the stakes:
"Patient health and patient lives may be at risk if these procedures are done improperly."
With over 5 million cord blood samples stored globally and more than 80% of mesenchymal stem cell submissions relying on cryopreservation, maintaining the ultra-cold chain isn’t just a procedural requirement - it’s essential for patient safety.
Conclusion
Transporting stem cells safely hinges on four key factors: keeping stable temperatures, using tested packaging, continuous monitoring, and following strict regulations. Every step in the process must be carefully controlled to preserve the cells’ viability.
Temperature control is especially critical. Fresh stem cells need to stay between 39°F and 86°F (4°C–30°C), with an ideal range of 59°F to 77°F (15°C–25°C), to remain viable. For cryopreserved cells, temperatures must stay below −130°F (around −90°C) to ensure long-term preservation. Even small deviations can lead to issues like cell aggregation, metabolic stress, or cell death, potentially lowering viability to less than 80%.
Regulatory standards play an equally vital role. From packaging to monitoring, every detail must align with guidelines to maintain viability. The latest FACT accreditation standards (5th edition) now mandate that shipping kits include temperature monitoring or systems to detect out-of-range conditions, ensuring a secure and traceable chain of custody. Additionally, independent validation from organizations like ISTA and IATA confirms that containers can handle real-world challenges such as extreme weather and unexpected delays.
While standard shipping services lack the temperature control needed for cord blood shipments, specialized biobanking services use advanced protocols. These include temperature-regulated transport and priority handling, which not only protect cell viability but also meet the highest clinical standards.
Americord Registry stands out by combining AABB accreditation with proven transport methods and continuous temperature monitoring. Their meticulous process - from collection to storage - ensures your family’s stem cells are preserved for future treatments. When it comes to something as important as your family’s health, choosing a provider with reliable transport and storage practices isn’t just a decision - it’s a necessity.
FAQs
What happens if stem cells warm up during shipping?
If stem cells are exposed to warming during shipping, their ability to survive and function can suffer irreversible damage. This kind of temperature fluctuation can significantly diminish their potential for use in medical treatments, as the cells may no longer perform as intended.
How do shippers keep fresh stem cells near 4°C (39°F) without freezing them?
Shippers rely on insulated containers equipped with advanced features like vacuum-insulated panels, phase-change gel packs, and temperature indicators to keep the internal environment steady at around 4°C (39°F). Paired with trained medical courier services, these measures protect the contents from extreme temperature fluctuations. This meticulous approach ensures that stem cells stay viable and functional throughout transit, all while avoiding freezing.
What should I look for in a temperature monitoring report?
To ensure the integrity of cord blood during transport, a temperature monitoring report must verify that the blood was maintained within the recommended range of 39–86°F (4–30°C). This report should include detailed temperature logs or indicators that confirm stable conditions throughout the journey. If any deviations from this range occurred, they should be documented to assess their potential effect on the viability of the cells.
The views, statements, and pricing expressed are deemed reliable as of the published date. Articles may not reflect current pricing, offerings, or recent innovations.