How Americord Ensures Safety with Backup Systems
Americord prioritizes the long-term safety of your baby's stem cells by implementing a multi-layered protection system. Here's how they safeguard over 22,000 cryogenically stored units:
- Uninterrupted Power Systems: Backup power setups, including UPS and emergency generators, ensure continuous operations during outages.
- Cryogenic Storage: Stem cells are stored in nitrogen-cooled tanks at -196°C, which don't rely on electricity.
- 24/7 Monitoring: Real-time tracking of temperature, humidity, and equipment performance is paired with automated alerts and remote surveillance.
- Disaster Recovery: A partnership with GenVault enables secure transfer of samples during emergencies.
- Data Security: Digital tracking systems use encryption, firewalls, and redundancy to protect and maintain specimen records.
Americord supports these measures with regular testing, compliance with AABB and FDA standards, and a $110,000 Quality Guarantee. Their systems are designed to ensure your stem cells remain safe and viable for decades of lifetime storage.
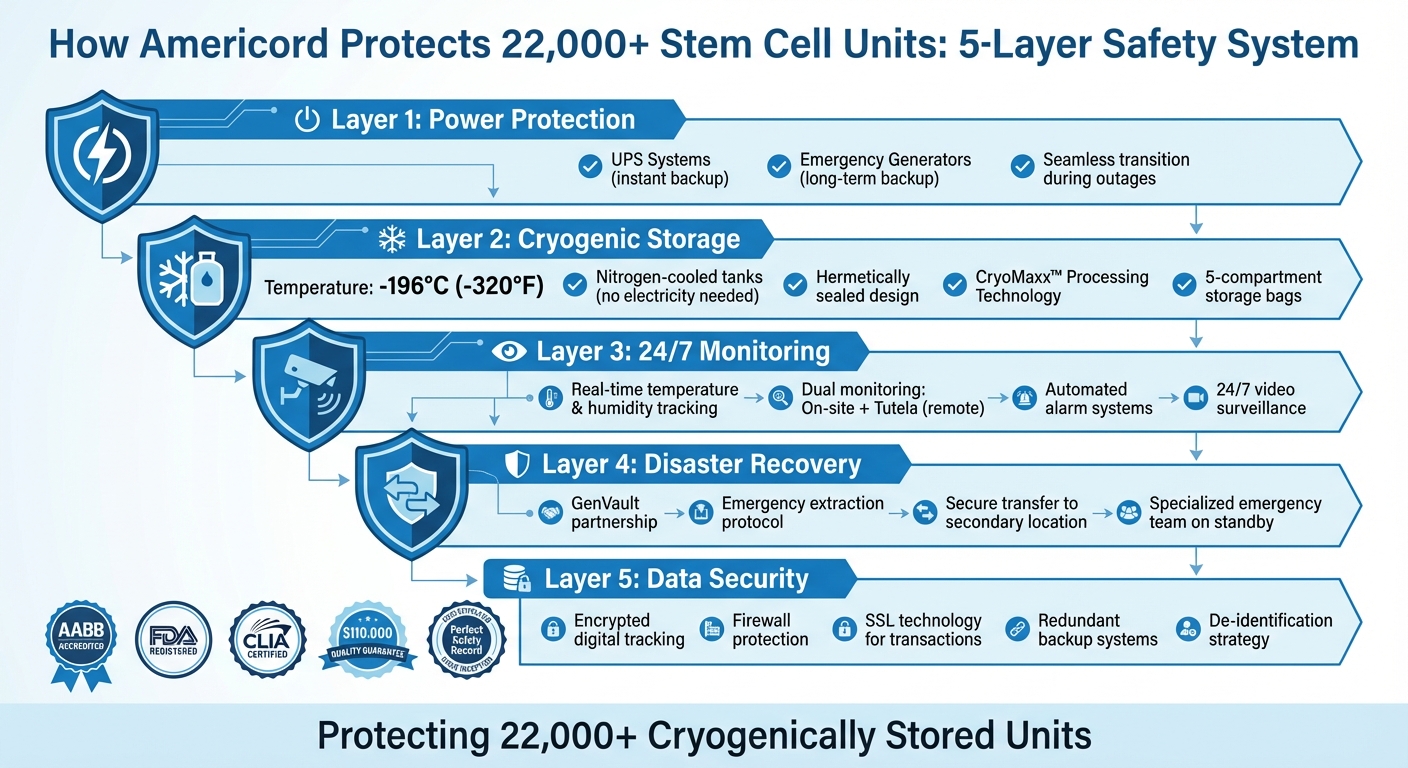
Americord's 5-Layer Stem Cell Safety System with Backup Protection
Power Supply and Battery Backup Systems
Americord's biobanking services and cryogenic storage facilities rely on a multi-layered power system to guarantee uninterrupted operations. This setup ensures that even the slightest power disruptions won't compromise temperature stability or trigger alarms.
Uninterrupted Power Supply (UPS) Systems
When the primary power fails, UPS systems immediately take over to power critical equipment like cryogenic tanks, temperature sensors, and alarms. This instant response prevents any fluctuations that could affect temperature regulation or monitoring systems. This level of security is a key part of the cord blood banking benefits families receive when choosing a private bank. By bridging the gap until backup systems activate, UPS systems ensure that essential operations remain consistent and stable.
Emergency Generator Integration
For longer outages, emergency generators automatically kick in to provide continuous power. The transition from UPS to generator power is seamless, keeping the entire facility operational. On-site staff routinely maintain and fine-tune these systems, conducting regular tests to ensure smooth transitions during emergencies. Whether it's a storm, grid failure, or another challenge, this dual-layer setup ensures prolonged electrical disruptions won't impact critical processes, including monitoring and data backups.
Cryogenic Storage with Multiple Freezer Systems
Americord preserves stem cells in nitrogen-cooled tanks kept at an astonishing -196°C (around -320°F). At this temperature, molecular movement and metabolic activity come to a complete standstill, ensuring the cells remain perfectly preserved. Unlike mechanical freezers, these nitrogen tanks don’t rely on electricity, making them a more reliable option for families deciding if cord blood banking is worth it.
Multiple Freezer Configuration
Americord’s facility is equipped with hermetically sealed tanks designed to withstand environmental hazards and even natural disasters. To add another layer of security, Americord has partnered with GenVault to establish a contingency plan for emergencies.
"Through a strategic partnership with GenVault, Americord ensures the immediate extraction and secure transfer of all tanks to a secondary location in the event of a facility-wide threat." - Americord
This system ensures that stem cell samples are not reliant on a single location. Even in extreme situations, the tanks are built to protect their contents, forming a critical part of Americord’s broader emergency response strategy.
CryoMaxx™ Processing Technology
Americord uses its CryoMaxx™ processing method to handle stem cells with precision. For cord blood banking, this involves manual techniques based on the Rubenstein method, while perinatal tissues undergo minimal handling. The process includes controlled-rate freezing, where temperatures drop gradually - 1°C (about 1.8°F) per minute - to avoid ice crystal formation and maintain the highest possible stem cell yield.
Samples are stored in a specially designed 5-compartment bag, which allows up to five treatments from a single collection. This setup not only protects the cells but also maximizes their usability. Together with precise temperature regulation, these measures form a robust system to keep stem cells safe and viable.
24/7 Monitoring and Alarm Response
Americord Registry uses a dual-layer monitoring system - combining on-site alarms with remote surveillance - to keep a constant watch on storage conditions. Each system serves as a backup for the other, ensuring uninterrupted oversight.
Real-Time Monitoring Systems
Specialized sensors continuously track key factors like temperature, humidity, and equipment performance. These sensors send real-time updates to on-site systems and Tutela, a third-party service that provides remote monitoring.
"Temperature sensors and ultra-low freezer probes report data to an interface monitored by Tutela. This third-party service provides remote alerts to key employees if temperatures deviate from the calibrated range." - Americord
Additionally, the facility is equipped with 24/7 remote video surveillance, offering an added layer of security and operational monitoring beyond environmental controls.
Automated Alarm Systems
If any storage conditions fall outside their calibrated range, automated alerts are sent to key personnel through both on-site alarms and Tutela's remote notifications. Lab technicians are on standby to address issues immediately, and an emergency team is prepared to relocate storage tanks if necessary.
"With 24/7 monitoring and a specialized emergency team that is able to move newborn stem cells at a moment's notice, your baby's stem cells are protected from natural catastrophe, national crises, or other potential failure points." - Americord
These systems work hand-in-hand with secure digital backups, which are monitored continuously to ensure accurate sample tracking and data integrity.
Data Backup for Sample Tracking
Protecting digital records is just as important as safeguarding the physical stem cell samples themselves. Americord Registry takes a multi-layered approach to data security, ensuring that specimen tracking remains reliable, accessible, and secure.
Secure Digital Storage
In addition to physical security measures, Americord employs robust digital systems to keep all data safe. Client and tracking information is stored on servers protected by firewalls, effectively blocking unauthorized access or tampering. Advanced encryption protocols, including SSL technology, are used for all payment processes and sensitive data transfers. These measures align with stringent standards set by HIPAA, the FDA, and GDPR.
"All information you provide to us is stored on our secure servers behind firewalls. Any payment transactions will be encrypted using SSL technology." - Americord Registry LLC
To further protect privacy, Americord uses a de-identification strategy. Each specimen is assigned a unique code, and only a small group of authorized employees can map these codes back to client identities. This "blind" storage system ensures that even within the laboratory in Franklin Township, New Jersey, specimens cannot be linked to specific families without proper authorization.
On top of secure storage, redundant backup systems add another layer of protection for all digital records.
Multiple Data Backup Systems
Americord’s dual-layer tracking system ensures data redundancy. Environmental data from storage tanks is backed up both online and on-site, with updates happening automatically across multiple systems. This setup guarantees that tracking records remain intact and up-to-date.
Even in emergencies, the connection between physical samples and their digital records is preserved. This system ensures that specimens remain traceable under any circumstances. With more than 22,000 cryogenically stored units as of 2023, Americord has maintained a flawless safety record, with no major security or operational issues since its inception.
Testing and Maintenance of Backup Systems
Thorough testing and upkeep are essential parts of Americord's layered safety measures. This section explains how regular inspections and adherence to industry standards help maintain these protections.
Backup systems demand constant attention to ensure reliability. Americord Registry actively tests and maintains its systems, guaranteeing that every safeguard is ready to perform when needed.
Regular System Inspections
Americord employs on-site oversight to make real-time adjustments based on alerts. This hands-on approach allows the team to address potential issues before they escalate.
To maintain precise storage conditions of -196°C for stem cells, specialized lab technicians regularly perform system calibrations as required by AABB standards.
The dual-monitoring system undergoes constant checks. On-site teams monitor localized 24/7 alarms, while a third-party service, Tutela, remotely oversees temperature sensors and ultra-low freezer probes. This layered approach ensures that if one system encounters a problem, the other remains fully operational.
Compliance with Industry Standards
Americord's maintenance protocols adhere to multiple regulatory standards. The facility holds AABB accreditation, FDA registration, and CLIA certification, all of which require detailed documentation and frequent inspections. Martin Smithmyer, Founder and CEO of Americord, emphasized:
"The AABB accreditation reinforces our position as an industry leader and reflects our commitment to the future of regenerative medicine."
Beyond meeting these national standards, the lab also holds state-specific licenses for operation in New York, New Jersey, California, Maryland, and Illinois, each with unique testing and maintenance requirements. These consistent evaluations strengthen Americord’s safety measures. Coupled with cryopreservation tanks designed to resist external environmental factors, these protocols ensure the continuous protection of stored stem cells.
Conclusion
Every component of Americord Registry's system is designed to ensure the safety and security of your stem cell storage. From uninterrupted power supplies and emergency generators to dual-monitoring systems and cryogenic tanks kept at an ultra-low temperature of -196°C, each layer works to protect your family's investment.
With features like CryoMaxx™ processing, 5-compartment storage bags, and a disaster recovery plan supported by GenVault, Americord Registry has upheld a perfect safety record, safeguarding over 22,000 cryogenically stored units since its start.
For families banking cord blood and tissue, these measures provide genuine peace of mind. The $110,000 Cord Blood Quality Guarantee reflects Americord Registry's confidence in its protocols, while AABB accreditation, FDA registration, and CLIA certification highlight its adherence to the highest standards in the field.
"I know that Americord is taking care of my baby's cord blood. We have not needed it yet, but should the need arise, I am confident they will be there every step of the way." - Jessie J., Mother of 3
FAQs
How does Americord ensure the safety of stem cells during emergencies through its partnership with GenVault?
Americord collaborates with GenVault to enhance the safety of stem cell storage, especially during emergencies. GenVault’s advanced biorepository provides climate-controlled, GxP-compliant storage along with secure transport solutions to ensure the integrity of sensitive biological samples like stem cells.
Through this partnership, stem cells are stored in facilities equipped with redundant power systems, real-time monitoring, and stringent environmental controls. This setup protects samples from risks such as power outages or temperature changes. Families can feel confident knowing their stem cells are securely preserved, even in unexpected situations.
How does Americord protect digital records for stem cell tracking?
Americord employs cutting-edge technology and rigorous protocols to maintain the security of digital records used for tracking stem cells. Each stem cell unit is assigned a unique code to ensure precise storage and identification. Access to these codes is tightly controlled, allowing only authorized personnel to retrieve or match samples. This approach prioritizes client confidentiality and privacy.
On top of that, Americord's facility is equipped with robust physical security measures. These include controlled access points and cryo-preservation tanks that are monitored around the clock. Together, these digital and physical safeguards work to protect both the stored stem cells and the associated records.
How does Americord's CryoMaxx™ method ensure the highest quality and usability of stored stem cells?
Americord's CryoMaxx™ method focuses on maintaining the quality and functionality of stem cells through advanced minimal manipulation techniques. This process helps preserve the natural multipotent cells, growth factors, and cytokines present in cord tissue, ensuring their integrity.
To add even more flexibility, the tissue is cryopreserved in multiple vials. This approach allows for easier access and use in future treatments or therapies, ensuring the stem cells remain viable and ready when needed.
The views, statements, and pricing expressed are deemed reliable as of the published date. Articles may not reflect current pricing, offerings, or recent innovations.