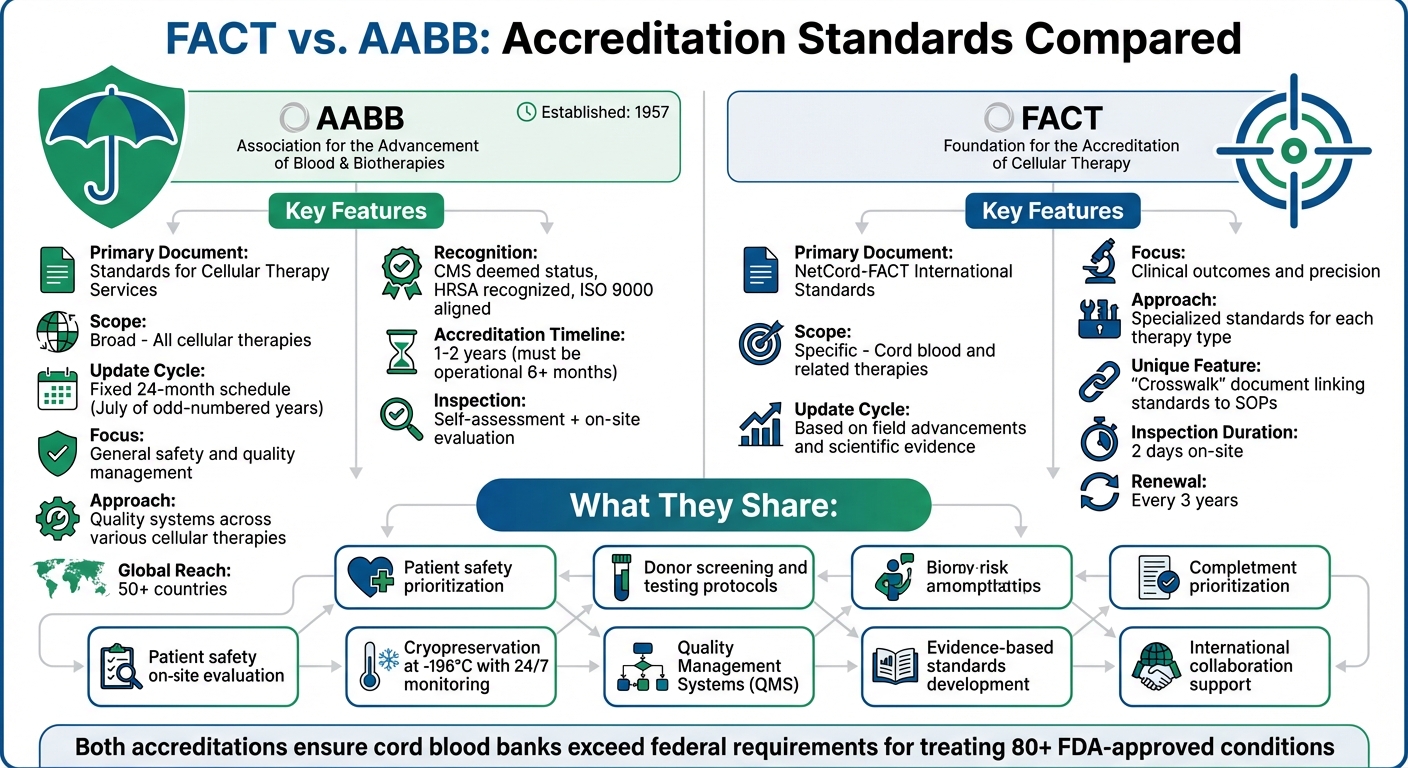
FACT vs. AABB: Accreditation Standards Compared
Accreditation ensures quality in cord blood banking, helping you choose a facility that meets stringent safety and reliability protocols. Two major accrediting bodies in the U.S. are AABB (Association for the Advancement of Blood & Biotherapies) and FACT (Foundation for the Accreditation of Cellular Therapy). Both set high standards, but their focus differs:
- AABB emphasizes broad quality systems across various cellular therapies and updates standards every two years. It’s widely recognized, including by CMS and HRSA.
- FACT specializes in clinical outcomes for cord blood banking, with detailed standards tailored to each operational stage.
Both prioritize patient safety, donor screening, and cryopreservation protocols. Accreditation from either body signals a commitment to excellence, ensuring your stored stem cells are managed with care.
Quick Comparison
| Aspect | AABB Standards | FACT Standards |
|---|---|---|
| Primary Document | Standards for Cellular Therapy Services | NetCord-FACT International Standards |
| Scope | Broad: All cellular therapies | Specific: Cord blood and related therapies |
| Update Cycle | Fixed 24-month schedule | Based on field advancements |
| Focus | General safety and quality | Clinical outcomes and precision |
Accreditation matters because it guarantees your cord blood bank exceeds basic regulatory requirements, giving you confidence in your decision.
AABB Accreditation Standards
AABB (Association for the Advancement of Blood & Biotherapies) has been a global leader in quality assessment since 1957. Its mission focuses on ensuring the safety, availability, and effectiveness of transfusion medicine and biotherapies across the globe. By developing peer-reviewed standards, offering accreditation programs, and providing education for professionals and institutions in cellular therapy, AABB sets itself apart from other accreditation models - a distinction that becomes clearer when compared to FACT later.
The accreditation process involves two key phases: a self-assessment through the APEX portal, followed by an on-site evaluation. Most facilities achieve accreditation within one to two years, but they must be operational for at least six months in their chosen activity before applying.
"AABB allows facilities to demonstrate their commitment to quality and safety while raising trust amongst donors, patients, and clients. In addition, having a third-party endorsement differentiates you from competitors and gives you a broader opportunity to increase your overall footprint." - Marc Lewis, Vice President of Operations, Gulf Coast Regional Blood Center
AABB combines quality management principles with technical standards to ensure the highest levels of safety for donors, patients, and staff, while maximizing the benefits of cord blood banking. These standards are updated every two years by expert committees to align with current medical practices and scientific advancements. The latest edition of the Standards for Cellular Therapy Services will take effect on July 1, 2025.
Core Requirements of AABB Standards
AABB standards address the entire process of cord blood banking, starting with maternal screening and infectious disease testing to ensure the safety of the initial materials. The requirements extend to every stage, including product processing, cryopreservation, storage, clinical administration, and monitoring patient outcomes. Unlike certifications that focus solely on technical aspects, AABB also demands documentation of clinical effectiveness.
Facilities are required to maintain written policies, processes, and procedures (PPPs) for all standards and must demonstrate compliance with these during evaluations. Record retention policies are strict - records must be kept for ten years from either the creation date or the product's final disposition, depending on the type of record. Any issues identified during on-site assessments must be addressed with corrective actions before accreditation is granted.
These standards aim to improve existing processes without requiring a complete overhaul of operations. Each set of AABB Standards is valid for two years, with updates for cord blood and cellular therapy services typically introduced on July 1 of odd-numbered years. This rigorous approach has established AABB as a global authority in stem cell banking standards.
AABB's Global Recognition
The precision and thoroughness of AABB standards have made them applicable worldwide. AABB-accredited facilities operate in over 50 countries, and the standards are designed to align with ISO 9000 requirements, making them adaptable to various regional needs. AABB has even provided templates for localized standards used in regions like Latin America, the Caribbean, and Africa.
The organization's influence extends beyond voluntary compliance. AABB holds "deemed status" from the Centers for Medicare and Medicaid Services (CMS), meaning its standards meet or exceed federal regulations. It is also recognized by the Health Resources and Services Administration (HRSA) as an accrediting body for the Bill Young Transplant Program. In California, state law mandates that laboratories adhere to AABB standards, and an AABB assessment can fulfill the state's inspection requirements.
Private insurers, including Aetna, often require AABB accreditation for coverage and reimbursement of specific procedures. This recognition from external entities not only helps facilities attract skilled staff but also fosters trust among donors and patients on a global scale.
FACT Accreditation Standards
FACT focuses exclusively on cellular therapy products and their clinical applications. Unlike AABB, which sets broad technical benchmarks, FACT zeroes in on clinical outcomes. Its standards are developed by international teams of experts, relying on published research and scientific consensus. This clinical focus distinguishes FACT within the accreditation field.
"FACT Standards are the only set of requirements that emphasize the clinical use of cellular therapy products collected and processed with rigorous controls." - FACT
FACT standards address every operational stage, prioritizing reliability in clinical outcomes. These standards apply to cord blood units intended for unrelated allogeneic use, as well as related units for allogeneic or autologous use by donor families. This ensures consistent quality across all cord blood units. This is a fundamental aspect of newborn stem cell banking.
How FACT Accreditation Works
The accreditation process begins with facilities creating a profile in the FACT Accreditation Portal and submitting an Eligibility Application along with a registration fee. Once the application is approved, banks complete a self-assessment and submit their Compliance Application.
A standout feature of FACT's process is the "crosswalk" document - a detailed guide linking each standard to the facility's policies, standard operating procedures (SOPs), and records. This document simplifies the inspection process by helping evaluators quickly locate evidence of compliance. During this phase, banks must also confirm that key personnel, such as Directors and Quality Managers, will be available for the on-site evaluation.
On-site inspections typically last two days, but facilities with multiple locations or off-site storage may require additional time. For international facilities, any documents not in English must be accompanied by translators for each inspector during the evaluation. After the inspection, the Accreditation Committee reviews the findings, and facilities must address any deficiencies before accreditation is granted. Accreditation must be renewed every three years, with a compliance application submitted 11 months before the expiration date. This rigorous process reflects FACT's detailed requirements for cord blood management.
FACT's Cord Blood Requirements
FACT standards encompass every aspect of cord blood banking, including cryopreservation, storage, registry listing, search and selection, release, and distribution. Each phase requires documented procedures and validated processes to maintain quality.
"The major objective of the NetCord FACT Standards is to promote quality medical and laboratory practices throughout all phases of cord blood banking to achieve consistently high-quality cord blood units available to patients for lifesaving treatments." - FACT
Facilities must establish a strong Quality Management System (QMS) that includes meeting records, audits, adverse reaction reports, occurrence reports, and corrective and preventive actions (CAPA). Critical electronic systems must be validated, and equipment must be properly qualified to ensure consistency. FACT regularly updates its standards in collaboration with experts in cell manufacturing and clinical administration, keeping them aligned with global practices and the latest scientific advancements.
How FACT and AABB Standards Differ
Side-by-Side Comparison of Standards
When it comes to cord blood banking, FACT and AABB take distinctly different approaches. AABB employs a broad quality systems approach, applying a single set of standards to all cellular therapy services, including cord blood, hematopoietic progenitor cells, and somatic cells. FACT, on the other hand, developed the NetCord-FACT International Standards, which are tailored specifically for cord blood banking, offering a more specialized set of guidelines.
| Aspect | AABB Standards | FACT Standards |
|---|---|---|
| Primary Document | Standards for Cellular Therapy Services | NetCord-FACT International Standards for Cord Blood Collection, Banking, and Release for Administration |
| Scope | One set of standards for all cellular therapy services | Separate specialized standards for cord blood, immune effector cells, and hematopoietic therapy |
| Update Cycle | Fixed 24-month revision cycle (July of odd-numbered years) | Updated intermittently based on field experience and scientific evidence |
| Operational Focus | General requirements for procurement, processing, storage, and distribution | Detailed requirements for search, selection, and reservation of units |
The update cycles reflect the differing philosophies of these organizations. AABB adheres to a consistent 24-month revision schedule, ensuring global compliance and compatibility with ISO standards. Their standards are even recognized as state law in California. With over six decades of experience, AABB holds the distinction of being the only U.S. program accredited by the International Society for Quality in Healthcare (ISQua). FACT, on the other hand, updates its standards as needed, based on advancements in the field and scientific evidence, allowing for a more adaptive approach.
Specialized vs. General Approach
These differences extend beyond operational details to the core philosophy of each system.
"AABB Standards for Cellular Therapy Services are designed to be broadly applicable to a range of activities, clinical programs, and cell types." - AABB
AABB’s framework is intentionally broad, applying uniform quality principles across various cell types and therapies. This approach is designed to ensure patient safety while maintaining flexibility for diverse applications.
FACT, however, takes a more focused route, offering detailed guidance for every stage of cord blood banking - from donor management and collection to search, selection, and distribution. Its cord blood-specific requirements address each operational phase with precision.
"The FACT Standards are the only set of requirements that emphasize the clinical use of cellular therapy products collected and processed with rigorous controls." - FACT
This clinical focus is a key differentiator. While AABB prioritizes uniformity and safety across all cellular therapies, FACT integrates clinical outcomes directly into laboratory practices. By emphasizing how collection and processing impact the clinical use of cord blood units, FACT ensures its standards are tightly aligned with the needs of patients and healthcare providers.
What FACT and AABB Standards Have in Common
Shared Safety and Quality Requirements
While FACT and AABB have their unique approaches, they share a strong focus on patient safety and quality assurance. Both organizations establish guidelines that oversee every stage of cord blood banking - from the moment of collection to its clinical release.
One key area of overlap is donor management. Both FACT and AABB emphasize the importance of screening and testing maternal and infant donors to determine their eligibility. This step is critical because the quality of a cord blood unit starts with a thorough evaluation of the donor.
Another shared focus is on storage and preservation protocols. Both organizations enforce strict cryopreservation standards. This includes maintaining liquid nitrogen storage tanks at around -196°C, with 24/7 monitoring systems in place. These systems must log data continuously, and the logs are subject to inspection to ensure the stored units remain viable for future use.
When it comes to processing methods, FACT and AABB require standardized procedures for cell collection, processing, and characterization. These methods are designed to consistently yield high-quality stem cells and their uses. The ultimate goal is to ensure that cord blood units meet established quality benchmarks, making them safe and effective for use in transplant centers worldwide. Both standards also support international collaboration by adhering to shared quality frameworks.
Both organizations base their standards on evidence and develop them with international input. Their guidelines often go beyond federal, state, and national requirements. As AABB explains:
"The Standards cover all aspects of operation from donor selection and testing to product processing, storage, clinical administration, and patient outcomes."
These shared safety and quality measures are the foundation of trust in accredited cord blood banks. Families can rely on this baseline level of protection, knowing their investment in their child’s future health is backed by rigorous standards that Americord Registry and other accredited banks uphold.
Why Accreditation Matters When Choosing a Cord Blood Bank
Accreditation ensures that a cord blood bank exceeds federal and state safety and quality standards. When you decide to preserve your newborn's stem cells, you're making a commitment to a resource that could help treat over 80 FDA-approved conditions. This is an investment in your child's future health, so it's critical to have strong, reliable safeguards in place.
Organizations like FACT and AABB set strict accreditation standards, offering an added layer of oversight. These standards ensure that every step of the banking process - from the initial collection to long-term storage - follows rigorous protocols. Periodic reviews by these accrediting bodies verify that banks maintain best practices, giving you peace of mind when making such an important choice.
How to Verify Accreditation
Verifying a cord blood bank's accreditation is quick and can help you avoid costly mistakes. Start by checking for the official logo on the bank's website. Only facilities with current AABB accreditation are allowed to display the AABB logo, while FACT-accredited banks use a specific "Seal of Accreditation" after completing their licensing requirements.
For further confirmation, visit FACTglobal.org and use their "FACT Accredited Organizations" search tool, or check the AABB website for their registry of accredited facilities. You can also directly request documentation from the bank - ask for a copy of their accreditation certificate. Taking a few minutes to verify this information adds an extra layer of security to your decision.
Americord Registry's AABB Accreditation
Americord Registry holds AABB accreditation, adhering to stringent standards in processing and storing stem cells. The company also offers a $110,000 quality guarantee - the highest engraftment guarantee in the industry. This financial assurance reflects their confidence in their processing and storage protocols.
In addition to accreditation, Americord has garnered a 5/5 consumer rating based on Better Business Bureau reviews. It’s recognized as the "#1 Parent-Rated Cord Blood Bank", with more 5-star reviews across major platforms than any other bank. Coupled with transparent pricing, starting at $3,799 for cord blood banking, and a strong focus on customer satisfaction, Americord demonstrates both regulatory compliance and a commitment to personalized service.
Conclusion
FACT and AABB accreditation standards both play a key role in overseeing cord blood banking, though their focus differs. AABB centers on technical quality management, while FACT places greater importance on clinical outcomes. Despite these distinct approaches, both organizations aim to ensure cord blood units meet strict safety and quality standards across collection, processing, and storage.
Accreditation is crucial because cord blood stem cells are used to treat over 80 FDA-approved conditions. When storing your newborn stem cells, you want confidence that the facility adheres to rigorous, up-to-date protocols. For example, AABB updates its standards every two years through expert committees to keep pace with advancements in the field. Americord Registry, which holds AABB accreditation, underscores its commitment to these high standards with a $110,000 quality guarantee and a 5/5 rating from the Better Business Bureau. This blend of regulatory compliance, financial assurance, and customer trust offers families peace of mind when making decisions about their child’s future health.
Understanding these accreditation details is essential for an informed decision. Opting for an accredited cord blood bank isn’t just about meeting baseline requirements - it’s about selecting a facility that excels in every aspect of stem cell preservation, from collection to long-term storage.
FAQs
What distinguishes FACT and AABB accreditation standards for stem cell banking?
FACT and AABB accreditation standards serve distinct purposes, each with unique focuses and requirements.
FACT places a strong emphasis on clinical outcomes and research. Facilities aiming for this accreditation must store at least 500 units before they can even be inspected. The process involves thorough evaluations to ensure facilities meet its stringent requirements.
On the other hand, AABB concentrates on quality management and technical operations. Its inspections occur every two years, focusing on whether facilities meet specific operational standards necessary to retain accreditation.
Both systems are essential for maintaining the safety and reliability of stem cell banking, but they approach their goals from different angles.
Why does accreditation matter when selecting a cord blood bank?
Picking a cord blood bank is a big decision, and accreditation plays a key role in making the right choice. Accreditation guarantees that the facility adheres to strict quality and safety standards for collecting, processing, and storing stem cells. Organizations like FACT (Foundation for the Accreditation of Cellular Therapy) and AABB (formerly the American Association of Blood Banks) assess these banks to ensure they follow rigorous industry protocols. This helps ensure your family’s stem cells are preserved under the best possible conditions.
Accredited banks are regularly inspected and must prove they maintain strong management systems and deliver reliable clinical outcomes. This means you can trust that the stem cells will remain usable and safe for future medical treatments. By choosing an accredited bank, you reduce risks and ensure that your family’s investment in regenerative medicine is handled with the utmost care.
How can I check if a cord blood bank is accredited by FACT or AABB?
To check if a cord blood bank is accredited by FACT or AABB, you can refer to their official accreditation directories. FACT provides a detailed list of accredited organizations, while AABB follows a strict process involving self-assessments and on-site evaluations every two years. These accreditations confirm that the bank adheres to rigorous standards for safety, quality, and professionalism, ensuring confidence when storing stem cells for potential medical needs.
The views, statements, and pricing expressed are deemed reliable as of the published date. Articles may not reflect current pricing, offerings, or recent innovations.